Abstract
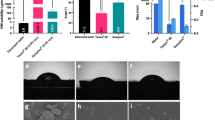
The latex obtained from Hancornia speciosa is used in folk medicine for treatment of several diseases, such as acne, warts, diabetes, gastritis and inflammation. In this work, we describe the biocompatibility assessment and angiogenic properties of H. speciosa latex and its potential application in medicine. The physical–chemical characterization was carried out following different methodologies (CHN elemental analyses; thermogravimetric analyses and Fourier transform infrared spectroscopy). The biocompatibility was evaluated through cytotoxicity and genotoxicity tests in fibroblast mouse cells and the angiogenic properties were evaluated using the chick chorioallantoic membrane (CAM) assay model. The physical–chemical results showed that the structure of Hancornia speciosa latex biomembrane is very similar to that of Hevea brasiliensis (commercially available product). Moreover, the cytotoxicity and genotoxicity assays showed that H. speciosa latex is biocompatible with life systems and can be a good biomaterial for medical applications. The CAM test showed the efficient ability of H. speciosa latex in neovascularization of tissues. The histological analysis was in accordance with the results obtained in the CAM assay. Our data indicate that the latex obtained from H. speciosa and eluted in water showed significant angiogenic activity without any cytotoxic or genotoxic effects on life systems. The same did not occur with H. speciosa latex stabilized with ammonia. Addition of ammonia does not have significant effects on the structure of biomembranes, but showed a smaller cell survival and a significant genotoxicity effect. This study contributes to the understanding of the potentialities of H. speciosa latex as a source of new phytomedicines.






Similar content being viewed by others
References
Bannerman RH. Traditional medicine in modern health care. World Health Forum. 1982;3:8–13.
Lambert J, Srivastava J, Vietmayer N. Medicinal plants—rescuing a global heritage, Technical Paper, 355. Washington: World Bank; 1997.
Smith-Hall C, Larsen HO, Pouliot M. People, plants and health: a conceptual framework for plant consumption. J Ethnobiol Ethnomed. 2012;8:43.
Konno K, Hirayama C, Nakamura M, Tateishi K, Tamura Y, Hattori M, Kohnok K. Papain protects papaya trees from herbivorous insects: role of cysteine proteases in latex. Plant J. 2004;37:370–8.
Gurib-Fakim A. Medicinal plants: traditions of yesterday and drugs of tomorrow. Mol Aspects Med. 2006;27:1–93.
Floriano JF, Mota LSL, Furtado EL, Rossetto VJV, Graeff CFO. Biocompatibility studies of natural rubber latex from different tree clones and collection methods. J Mater Sci Mater Med. 2013;. doi:10.1007/s10856-013-5089-9.
Frade MA, Assis RV, Coutinho-Netto J, Andrade TA, Foss NT. The vegetal biomembrane in the healing of chronic venous ulcers. An Bras Dermatol. 2012;87(1):45–51.
Araújo MM, Massuda ET, Hyppolito MA. Anatomical and functional evaluation of tympanoplasty using a transitory natural latex biomembrane implant from the rubber tree Hevea brasiliensis. Acta Cirúrgica Brasileira. 2012;27(8):566–71.
Martins R, Kinoshita AMO, Carvalho NTA, Guimarães SAC. Comparative study of bone response guides tissue regeneration technique—macroscopic evaluation, Part 1. Full Densitry Sci. 2010;1(3):224–30.
Ereno C, Guimarães SAC, Pasetto S, Herculano RD, Silva CP, Graeff CFO, Tavano O, Baffa O, Kinoshita A. Latex use as an occlusive membrane for guided bone regeneration. J Biomed Mater Res A. 2010;95A:932–9.
Herculano RD, Silva CP, Ereno C, Guimarães SAC, Kinoshita A, Graeff CFO. Natural rubber latex used as drug delivery system in guided bone regeneration (GBR). Mater Res. 2009;12(2):253–6.
Sampaio RB, Mendonca RJ, Simioni AR, Costa RA, Siqueira RC, Correa VM, Tedesco AC, Haddad A, Coutinho Netto J, Jorge R. Rabbit retinal neovascularization induced by latex angiogenic-derived fraction: an experimental model. Curr Eye Res. 2010;35(1):56–62.
Carvalho BR, Reis RM, Coutinho Netto J, Moura MD, Nogueira AA, Ferriani RA. Natural latex (Hevea brasiliensis) mold for neovaginoplasty. Rev Bras Ginecol Obstet. 2008;30(1):31–5.
Balabanian CA, Coutinho-Netto J, Lamano-Carvalho TL, Lacerda SA, Brentegani LG. Biocompatibility of natural latex implanted into dental alveolus of rats. J Oral Sci. 2006;48:201–5.
Ciapettig G, Stea S, Pizzofenato A, Checchi L, Pelliccaoni GA. A latex membrane, as an alternative in the GTR technique: preliminary report on its biocompatibility. J Mater Sci. 1994;5:647–50.
Ebo DG, Stevens WJ. IgE-mediated natural rubber latex allergy: an update. Acta Clin Belg. 2002;57:58–70.
Yagami T, Haishima Y, Tsuchiya T, Tomitaka-Yagami A, Kano H, Matsumaga K. Proteomic analysis of putative latex allergens. Int Arch Allergy Immunol. 2004;135:3–11.
Akasawa A, Hsieh LS, Lin Y. Serum reactivities to latex proteins (Hevea brasiliensis). J Allergy Clin Immunol. 1995;95:1196–205.
Wagner B, Krebitz M, Buck D, Niggemann B, Yeang HY, Han KH, Scheiner O, Breiteneder H. Cloning, expression, and characterization of recombinant Hev b 3, a Hevea brasiliensis protein associated with latex allergy in patients with spina bifida. J Allergy Clin Immunol. 1999;104:1084–92.
Berthelot K, Lecomte S, Estevez I, Coulary-Salin B, Bentaleb A, Cullin C, Deffieux A, Peruch F. Rubber elongation factor (REF), a major allergen component in Hevea brasiliensis latex has amyloid properties. PLOS One. 2012;7(10):e48065. doi:10.1371/journal.pone.0048065.
Malmonge JA, Camilo EC, Moreno RMB, Mattoso LHC, McMahan CM. Comparative study on technological properties of latex and natural rubber from Harconia speciosa gomes and Hevea brasiliensis. J Appl Polym Sci. 2009;111:2986–91.
Sampaio TS, Nogueira PCL. Volatile components of mangaba fruit (Hancornia speciosa) at three stages of maturity. Food Chem. 2005;95:606–10.
Ferreira HC, Serra CP, Lemos VS, Braga FC, Cortes SF. Nitric oxide-dependent vasodilatation by ethanolic extract of Hancornia speciosa via phosphatidyl-inositol 3-kinase. J Ethnopharmacol. 2007;109:161–4.
Ferreira HC, Serra CP, Endringer DC, Lemos VS, Braga FC, Cortes SF. Endothelium-dependent vasodilatation induced by Hancornia speciosa in rat superior mesenteric artery. Phytomedicine. 2007;14:473–8.
Silva CG, Braga FC, Lima MP, Pesquero JL, Lemos VS, Cortes SF. Hancornia speciosa Gomes induces hypotensive effect through inhibition of ACE and increase on NO. J Ethnopharmacol. 2011;137:709–11.
Moraes TM, Rodrigues CM, Kushima H, Baub TM, Villegas W, Pellizzon CH, Brito A, Hiruma-Lima CA. Hancornia speciosa: indications of gastroprotective, healing and anti-Heliobacter pilori actions. J Ethnopharmacol. 2008;120:161–8.
Macedo M, Ferreira AR. Plantas medicinais usadas no tratamento dermatológico da Bacia do alto Paraguai,Mato Grosso. Revista Brasileira Farmacognosia. 2004;14:40–4.
Ritter MR, Sobierajski GR, Schenkel EP, Mentz LA. Plantas usadas como medicinais no município de Ipê, RS, Brasil. Revista Brasileira Framacognosia. 2002;12:51–62.
Marinho DG, Alviano DS, Matheus ME, Alviano CS, Fernandes PD. The latex obtained from Hancornia speciosa Gomes possesses anti-inflammatory activity. J Ethnopharmacol. 2011;135:530–7.
Carvalho PCL, Soares WS, Ritzinger R, Carvalho JABS. Conservação de fruteiras tropicais com a participação do agricultor. Revista Brasileira de Fruticultura. 2001;23(3):730–4.
Brazilian Institute of Geography and Statistic. Sistema de Recuperação automática. http://www.sidra.ibge.gov.br. Accessed 23 May 2010.
Folkman J. Fundamental concepts of the angiogenic process. Curr Mol Med. 2003;3(7):643–51.
Schultz GS, Sibbad RG, Falanga V, Ayello EA, Dowsett C, Harding K, Romanelli M, Stacey MC, Teot L, Vanscheidt W. Wound bed preparation: a systematic approach to wound management. Wound Repair Regen. 2003;1:S1–28.
Shen JT, Falanga V. Innovative therapies in wound healing. J Cutan Med Surg. 2003;7(3):217–24.
Zisch AH, Lutolf MP, Hubbell JA. Biopolymeric delivery matrices for angiogenic growth factors. Cardiovasc Pathol. 2003;12(6):295–310.
Borenfreund E, Puerner J. Toxicity determined in vitro by morphological alterations and neutral red absorption. Toxicol Lett. 1985;24:119–24.
Singh NP, Stephens RE, Schneider EL. Modifications of alkaline microgel electrophoresis for sensitive detection of DNA damage. Int J Radiat Biol. 1994;66:23–8.
Melo-Reis PR, Andrade LS, Silva CB, Araújo LMM, Pereira MS, Mrué F, Chen-Chen L. Angiogenic activity of Symadenium umbellatum Pax latex. Braz J Biol. 2010;70:189–94.
Parente LML, Andrade MA, Brito LAB, Moura VMBD, Miguel MP, Lino-Junior RS, Tresvenzol LFM, Paula JR, Paulo NM. Angiogenic activity of Calendula officialis flowers L. in rats. Acta Cirúrgica Brasileira. 2011;26(1):19–24.
Polovková J, Klein E, Cibulková Z, Lukes V. FTIR study of gamma-irradiated cis-1,4-polyisoprene. Chem Pap. 2006;60(5):327–32.
ISO 10.993-3: International standard: Biological evaluation of medical devices—part 3. Tests for citotoxicity: in vitro methods ISO 10993-3, 1992.
ISO 10.993-5: International standard: Biological evaluation of medical devices - part 5. Tests for citotoxicity: in vitro methods ISO 10993-5, 1992.
Kekwich R, Bhambri S, Chabane MH, Autegarden JE, Levy DA, Leynadier F. The allergenic properties of fresh and preserved Hevea brasiliensis latex protein preparations. Clin Exp Immunol. 1996;104:337–42.
Dulngali S, Fah CS. Preservative system for field latex. Kuala Lumpur: RRIM, Universiti Malaya, Malaya; 1980.
La Grutta S, Mistrello G, Varin E, Pajno GB, Passalacqua G. Comparison of ammoniated and nonammoniated extracts in children with latex allergy. Allergy. 2003;58:814–8.
Cibulková Z, Polovková J, Luke V, Klein EJ. DSC and FTIR study of the gamma radiation effect on cis-polyisoprene. J Therm Anal Calorim. 2006;84:709–13.
Alam TM, Celina M, Assink RA, Clough RL, Gillen KT. 17O NMR investigation of oxidative degradation in polymers under g-irradiation. Radiat Phys Chem. 2001;60:121–7.
Goyal M, Nagori BP, Sasmal D. Wound healing activity of latex of Euphorbia caducifolia. J Ethnopharmacol. 2012;144(3):786–90.
Mello VJ, Gomes MT, Lemos FO, Delfino JL, Andrade SP, Lopes MT, Salas DE. The gastric ulcer protective and healing role of cystine proteins from Carica candamarcensis. Phytomedicine. 2008;15(4):237–44.
Wiegand C, Hipler UC. Methods for measurement of cell and tissue compatibility including tissue regeneration processes. Krankenaushygiene Interdisziplinar. 2008;3(1):1863–5245.
Zwadlo-Klarwasser G, Görlitz K, Hafemann B, Klee D, Klosterhalfen B. The choriallantoic membrane of the chick embryo as a simple model for the study of angiogenic and inflammatory response to biomaterials. J Mater Sci. 2001;12(3):195–9.
May AE, Seizer P, Gawaz M. Platelets: inflammatory firebugs of vascular walls. Arterioscler Thromb Vasc Biol. 2008;28:5–10.
Jackson JR, Seed MP, Kircher CH, Willoughby DA, Winkler JD. The codependence of angiogenesis and chronic inflammation. FASEB J. 1997;11(6):457–65.
Folkman J, Brem H. Angiogenesis and inflammation. In: Gallin JI, Goldstein IM, Snyderman R, editors. Inflammation: basic principles and clinical correlates. 2nd ed. New York: Raven; 1992. p. 821–39.
Remmers EF, Sano H, Wilder RL. Platelet-derived growth factors and heparin binding (fibroblast) growth factors in the synovial tissue pathology of rheumatoid arthritis. Semin Arthritis Rheum. 1991;21(3):191–9.
Acknowledgments
The authors acknowledge the Brazilian funding agencies MCT/CNPq, FNDCT, CAPES, FINEP, FAPEG, FAPESP and FUNAPEP. Revision of the English language text was carried out by Universidade Estadual de Goiás (UEG).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Almeida, L.M., Floriano, J.F., Ribeiro, T.P. et al. Hancornia speciosa latex for biomedical applications: physical and chemical properties, biocompatibility assessment and angiogenic activity. J Mater Sci: Mater Med 25, 2153–2162 (2014). https://doi.org/10.1007/s10856-014-5255-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-014-5255-8