Abstract
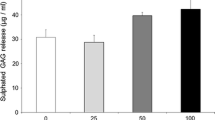
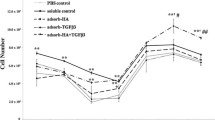
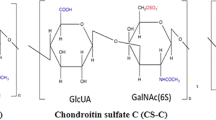
Cartilage extracellular matrix (ECM) is composed primarily of type II collagen (COL II) and large, networks of proteoglycans (PGs) that contain glycosaminoglycans such as hyaluronic acid (HA) and chondroitin sulfate (CS). Since cartilage shows little tendency for self-repair, injuries are kept unhealed for years and can eventually lead to further degeneration. During the past decades, many investigations have pursued techniques to stimulate articular cartilage repair or regeneration. The current study assessed the effects of exogenous glycosaminoglycans (GAGs) including CS-A, CS-B, CS-C, heparan sulfate and HA, administration on human chondrocytes in terms of proliferation and matrix synthesis, while the cells were seeded and grown on the genipin-crosslinked collagen type II (COL II) scaffold. DNA content was measured by Hoechst dye intercalation, matrix deposition was evaluated by DMMB dye. Expression of collagen II and aggrecan mRNAs was assessed by RT-PCR, followed by gel electrophoresis. In a 28-day in vitro culture, administration of 5 μg/ml CS-A, 50 μg/ml CS-B, 50 μg/ml CS-C, 5 μg/ml HS, and 500 kDa HA led to significant increase in biosynthesis rate of PGs. Gene expression of aggrecan and collagen II were upregulated by CS-A, CS-C and HA. These results showed considerable relevance of GAGs to the issue of in vitro/ex vivo neo-cartilage synthesis for tissue engineering and regenerative medical applications.

Similar content being viewed by others
References
Banu N, Tsuchiya T. Markedly different effects of hyaluronic acid, chondroitin sulfate-A on the differentiation of human articular chondrocytes in micromass, 3-D honeycomb rotation cultures. J Biomed Mater Res. 2007;80A:257–67.
Zhao HG, Ma L, Gong YH, Gao CY, Shen JC. A polylactide/fibrin gel composite scaffold for cartilage tissue engineering: fabrication and an in vitro evaluation. J Mater Sci Mater Med. 2009;20:135–43.
Ko CS, Wu CH, Huang HH, Chu IM. Genipin cross-linking of type II collagen-chondroitin sulfate-hyaluronan scaffold for articular cartilage therapy. J Med Biol Eng. 2007;27:7–14.
Ko CS, Huang JP, Huang CW, Chu IM, Type II. collagen-chondroitin sulfate-hyaluronan scaffold cross-linked by genipin for cartilage tissue engineering. J Biosci Bioeng. 2009;107:177–82.
Pieper JS, van der Kraan PM, Hafmans T, Kamp J, Buma P, van Susante JLC, et al. Crosslinked type II collagen matrices: preparation, characterization, and potential for cartilage engineering. Biomaterials. 2002;23:3183–92.
Sung HW, Chen CN, Huang RN, Hsu JC, Chang WH. In vitro surface characterization of a biological patch fixed with a naturally occurring cross-linking agent. Biomaterials. 2000;21:1353–62.
Sung HW, Huang RN, Huang LLH, Tsai CC. In vitro evaluation of cytotoxicity of a naturally occurring crosslinking reagent for biological tissue fixation. J Biomater Sci Polym Ed. 1999;10:63–78.
Couchman JR. Syndecans: proteoglycan regulators of cell-surface microdomains? Nat Rev Mole Cell Biol. 2003;4:926–37.
Park SN, Park JC, Kim HO, Song MJ, Suh H. Characterization of porous collagen/hyaluronic acid scaffold modified by 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide cross-linking. Biomaterials. 2002;23:1205–12.
Pieper JS, Oosterhof A, Dijkstra PJ, Veerkamp JH, van Kuppevelt TH. Preparation and characterization of porous cross-linked collagenous matrices containing bioavailable chondroitin sulphate. Biomaterials. 1999;20:847–58.
Vankemmelbeke MN, Holen I, Wilson AG, Ilic MZ, Handley CJ, Kelner GS, et al. Expression, activity of ADAMTS-5 in synovium. Eur J Biochem. 2001;268:1259–68.
Barbucci R, Lamponi S, Borzacchiello A, Ambrosio A, Fini M, Torricelli P, et al. Hyaluronic acid hydrogel in the treatment of osteoarthritis. Biomaterials. 2002;23:4503–13.
Fioravanti A, Cantarini L, Chellini F, Manca D, Paccagnini E, Marcolongo R, et al. Effect of hyaluronic acid (MW 500–730 kDa) on proteoglycan and nitric oxide production in human osteoarthritic chondrocyte cultures exposed to hydrostatic pressure. OsteoArthr Cartil. 2005;13:688–96.
Enobakhare BO, Bader DL, Lee DA. Quantification of sulfated glycosaminoglycans in chondrocyte/alginate cultures, by use of 1, 9-dimethylmethylene blue. Anal Biochem. 1996;243:89–91.
Lauder RM, Huckerby TN, Brown GM, Bayliss MT, Nieduszynski IA. Age-related changes in the sulphation of the chondroitin sulphate linkage region from human articular cartilage aggrecan. J Biochem. 2001;358:523–8.
Sugahara K, Mikami T, Uyama T, Mizuguchiz S, Nomuraz K, Kitagawa H. Recent advances in the structural biology of chondroitin sulfate, dermatan sulfate. Curr Opin Struct Biol. 2003;13:612–20.
Bernfield M, Gotte M, Park PW, Reizes O, Fitzgerald ML, Lincecum J, et al. Functions of cell surface heparan sulfate proteoglycans. Annu Rev Biochem. 1999;68:729–77.
Yip GW, Ferretti P, Copp AJ. Heparan sulphate proteoglycans, spinal neurulation in the mouse embryo. Development. 2002;129:2109–19.
Allemann F, Mizuno S, Eid K, Yates KE, Zaleske D, Glowacki J. Effects of hyaluronan on engineered articular cartilage extracellular matrix gene expression in 3-dimensional collagen scaffolds. J Biomed Mater Res. 2001;55:13–9.
Kamada H, Masuda K, Souza ALD, Lenz ME, Pietryla D, Otten L, et al. Age-related differences in the accumulation and size of hyaluronan in alginate culture. Arch Biochem Biophy. 2002;408:192–9.
Acknowledgment
This research is supported by Ministry of Economic Affairs, Taiwan (Technology Development Program for Academia 91-EC-17-A-17-S1-0009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, CH., Ko, CS., Huang, JW. et al. Effects of exogenous glycosaminoglycans on human chondrocytes cultivated on type II collagen scaffolds. J Mater Sci: Mater Med 21, 725–729 (2010). https://doi.org/10.1007/s10856-009-3889-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-009-3889-8