Abstract
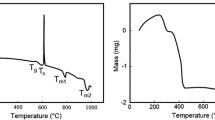
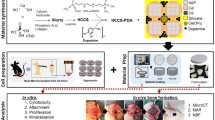
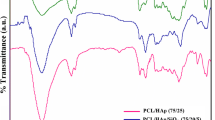
The primary goal of this investigation was to develop a calcium phosphate film hybridized with 1α,25-dihydroxyvitamin D3 for the improvement of osteoconductivity of bone substitutes. The hybrid films (hCaP) were prepared at the different concentrations of 1 × 10−10, 1 × 10−8, and 1 × 10−6 M designated as hCaPL, hCaPM, and hCaPH, respectively. The change of the hormone concentration during the preparation of the hybrid films did not cause significant variations on the physical properties of hCaPs, i.e. surface morphology and roughness. On the other hand, X-ray photon spectroscope (XPS) measurements revealed that the concentration change affected the chemical composition of the hybrid films. Recruitment of osteoblast-like MG-63 cells was considerably improved on hCaPs compared to tissue culture plate (TCP). However, cell proliferation on hCaPs was substantially suppressed and inversely proportional to the hormone concentration used. It was observed that bone-like nodules which consisted of bead-like components and well-developed matrix were rapidly formed on hCaPs. Masson’s trichrome and safranin-O stainings elucidated that the bead-like components were MG-63 cells. Safranin-O staining showed that proteoglycan was produced actively. These results indicate that the cells cultured on hCaPs were strongly stimulated by the hormone to produce proteoglycan which can be considered as an induction of premature bone formation. The number of the nodules was increased with hormone concentration and most pronounced at the hCaPH. Gene expression patterns of alkaline phosphatase (ALP), transforming growth factor-β (TGF-β), and osteopontin (OPN) were strongly modulated by hybridized the hormone. For ALP and OPN, gene expressions were activated earlier on hCaPs than untreated calcium phosphate (CaP) confirming the effect of the hybridization was substantial. The TGF-β gene expression was immediately activated after seeding but difference between samples was not significant suggesting that the gene expression was modulated not by the hormone hybridization but by CaP itself. As a result, hybridization of 1,25(OH)2D3 with CaP can be a potentially strong candidate to promote osteoconductivity of implant materials.









Similar content being viewed by others
References
Finke B, Luethen F, Schroeder K, Mueller P, Bergemann C, Frant M, et al. The effect of positively charged plasma polymerization on initial osteoblastic focal adhesion on titanium surfaces. Biomaterials. 2007;28:4521–34.
Jiang G, Evans ME, Jones IA, Rudd CD, Scotchford CA, Walker GS. Preparation of poly(epsilon-caprolactone)/continuous bioglass fibre composite using monomer transfer moulding for bone implant. Biomaterials. 2005;26:2281–8.
Link D, van den Dolder J, van den Beucken J, Wolke J, Mikos A, Jansen J. Bone response and mechanical strength of rabbit femoral defects filled with injectable CaP cements containing TGF-β1 loaded gelatin microparticles. Biomaterials. 2008;29:675–82.
Borsari V, Fini M, Giavaresi G, Rimondini L, Consolo U, Chiusoli L, et al. Osteointegration of titanium and hydroxyapatite rough surfaces in healthy and compromised cortical and trabecular bone: in vivo comparative study on young, aged, and estrogen-deficient sheep. J Orthop Res. 2007;25:1250–60.
Dong Z, Khor K, Quek C, White T, Cheang P. TEM and STEM analysis on heat-treated and in vitro plasma-sprayed hydroxyapatite/Ti-6Al-4V composite coatings. Biomaterials. 2003;24:97–105.
Shirkhanzadeh M, Azadegan M, Stack V. Fabrication of pure hydroxyapatite and fluoridated-hydroxyapatite coatings by electrocrystallization. Mater Lett. 1994;18:211–4.
Wolke J, van der Waerden J. In vivo dissolution behavior of various RF magnetron-sputtered Ca-P coatings on roughened titanium implants. Biomaterials. 2003;24:2623–9.
Gan L, Wang J, Pilliar R. Evaluating interface strength of calcium phosphate sol-gel-derived thin films to Ti6Al4V substrate. Biomaterials. 2005;26:189–96.
Gross K, Berndt C. Thermal processing of hydroxyapatite for coating production. J Biomed Mater Res A. 1998;39:580–7.
Nelea V, Pelletier H, Iliescu M, Werckmann J, Craciun V, Mihailescu I, et al. Calcium phosphate thin film processing by pulsed laser deposition and in situ assisted ultraviolet pulsed laser deposition. J Mater Sci: Mater Med. 2002;13:1167–73.
Okumura A, Goto M, Goto T, Yoshinari M, Masuko S, Katsuki T, et al. Substrate affects the initial attachment and subsequent behavior of human osteoblastic cells(Saos-2). Biomaterials. 2001;22:2263–71.
Delmas PD. Biochemical markers of bone turnover for the clinical assessment of metabolic bone disease. Endocrinol Metab Clin North Am. 1990;19:1–18.
Mulkins MA, Manolagas SC, Deftos LJ, Sussman HH. 1α, 25-dihydroxyvitamin D3 increases bone alkaline phosphatase isoenzyme levels in human osteogenic sarcoma cells. J Biol Chem. 1983;258:6219–25.
Franceschi RT, James WM, Zerlauth G. 1α, 25-dihydroxyvitamin D3 specific regulation of growth, morphology, and fibronectin in a human osteosarcoma cell line. J Cell Physiol. 1985;123:401–9.
Franceschi RT, Linson CJ, Peter TC, Romano PR. Regulation of cellular adhesion and fibronectin synthesis by 1 alpha, 25-dihydroxyvitamin D3. J Biol Chem. 1987;262:4165–71.
Lian J, Stewart C, Puchacz E, Mackowiak S, Shalhoub V, Collart D, et al. Structure of the rat osteocalcin gene and regulation of vitamin D-dependent expression. Proc Natl Acad Sci. 1989;86:1143–7.
Bonewald LF, Kester MB, Schwartz Z, Swain LD, Khare A, Johnson TL, et al. Effects of combining transforming growth factor beta and 1α, 25-dihydroxyvitamin D3 on differentiation of a human osteosarcoma (MG-63). J Biol Chem. 1992;267:8943–9.
Boyan BD, Batzer R, Kieswetter K, Liu Y, Cochran DL, Szmuckler-Moncler S, et al. Titanium surface roughness alters responsiveness of MG63 osteoblast-like cells to 1α, 25-(OH)2D3. J Biomed Mater Res. 1998;39:77–85.
Prince CW, Butler WT. 1, 25-dihydroxyvitamin D3 regulates the biosynthesis of osteopontin, a bone-derived cell attachment protein, in clonal osteoblast-like osteosarcoma cells. Coll Relat Res. 1987;7:305–13.
Price PA, Baukol SA. 1, 25-dihydroxyvitamin D3 increases synthesis of the vitamin K-dependent bone protein by osteosarcoma cells. J Biol Chem. 1980;255:11660–3.
Wong GL, Luben RA, Cohn DV. 1, 25-dihydroxycholecalciferol and parathormone: effects on isolated osteoclast-like and osteoblast-like cells. Science. 1977;197:663–5.
Raisz LG, Maina DM, Gworek SC, Dietrich JW, Canalis EM. Hormonal control of bone collagen synthesis in vitro: inhibitory effect of 1-hydroxylated vitamin D metabolites. Endocrinology. 1978;102:731–5.
Kream BK, Rowe D, Smith MD, Maher V, Majeska R. Hormonal regulation of collagen synthesis in a clonal rat osteosarcoma cell line. Endocrinology. 1986;119:1922–8.
Owen TA, Aronow MS, Barone LM, Bettencourt B, Stein GS, Lian JB. Pleiotropic effects of vitamin D on osteoblast gene expression are related to the proliferative and differentiated state of the bone cell phenotype: dependency upon basal levels of gene expression, duration of exposure, and bone matrix competency in normal rat osteoblast cultures. Endocrinology. 1991;128:1496–504.
Ishida H, Bellows CG, Aubin JE, Heersche JN. Characterization of the 1α, 25-(OH)2D3-induced inhibition of bone nodule formation in long-term cultures of fetal rat calvaria cells. Endocrinology. 1993;132:61–6.
Shi Y, Worton L, Esteban L, Baldock P, Fong C, Eisman JE, et al. Effects of continuous activation of vitamin D and Wnt response pathways on osteoblastic proliferation and differentiation. Bone. 2007;41:87–96.
Kim HJ, Kim SH, Kim MS, Lee EJ, Oh HG, Oh WM, et al. Varying Ti-6Al-4V surface roughness induces different early morphologic and molecular responses in MG63 osteoblast-like cells. J Biomed Mater Res A. 2005;74:366–73.
Boyan BD, Lohmann CH, Sisk M, Liu Y, Sylvia VL, Cochran DL, et al. Both cyclooxygenase-1 and cyclooxygenase-2 mediate osteoblast response to titanium surface roughness. J Biomed Mater Res. 2001;55:350–9.
Moulder JF, Stickle WF, Sobol PE, Bomben KD (1995) In: Chastain J, King RC, editors. Handbook of x-ray photoelectron spectroscopy. Minnesota: Physical Electronics, Inc.
Kaciulis S, Mattogno G, Pandolfi L, Cavalli M, Gnappi G, Montenero A. XPS study of apatite-based coatings prepared by sol-gel technique. Appl Surf Sci. 1999;151:1–5.
Ide-Ektessabi A, Yamaguchi T, Tanaka Y. RBS and XPS analyses of the composite calcium phosphate coatings for biomedical applications. Nucl Instrum Methods Phys Res B. 2005;241:685–8.
Ferraz MP, Monteiro FJ, Santos JD. CaO-P2O5 glass hydroxyapatite double-layer plasma-sprayed coating: in vitro bioactivity evaluation. J Biomed Mater Res. 1999;45:376–83.
Lohmann CH, Bonewald LF, Sisk MA, Sylvia VL, Cochran DL, Dean DD, et al. Maturation state determines the response of osteogenic cells to surface roughness and 1, 25-dihydroxyvitamin D3. J Bone Miner Res. 2000;15:1169–80.
Matsumoto T, Sowa Y, Ohtani-Fujita N, Tamaki T, Takenaka T, Kuribayashi K, et al. p53-independent induction of WAF1/Cip1 is correlated with osteoblastic differentiation by vitamin D3. Cancer Lett. 1998;129:61–8.
Owen TA, Aronow M, Shalhoub V, Barone LM, Wilming L, Tassinari MS, et al. Progressive development of the rat osteoblast phenotype in vitro: reciprocal relationships in expression of genes associated with osteoblast proliferation and differentiation during formation of the bone extracellular matrix. J Cell Physiol. 1990;143:420–30.
Bonewald LF, Mundy GR. Role of transforming growth factor beta in bone remodeling: a review. Connect Tissue Res. 1989;23:201–8.
Sporn MB, Roberts AB. Peptide growth factors are multifunctional. Nature. 1988;332:217–9.
Sodek J, Ganss B, McKee MD. Osteopontin. Crit Rev Oral Biol Med. 2000;11:279–303.
Irie K, Zalzal S, Ozawa H, Nanci A. Morphological and immunocytochemical characterization of primary osteogenic cell cultures derived from fetal rat cranial tissue. Anat Rec. 1998;252:554–67.
De Bruijn JD, Van Den Brink I, Mendes S, Dekker R, Bovell Y, Van Blitterswijk C. Bone induction by implants coated with cultured osteogenic bone marrow cells. Adv Dent Res. 1999;13:74–81.
Butler WT. The nature and significance of osteopontin. Connect Tissue Res. 1989;23:123–36.
Nagata T, Bellows CG, Kasugai S, Butler WT, Sodek J. Biosynthesis of bone proteins [SPP-1 (secreted phosphoprotein-1, osteopontin), BSP (bone sialoprotein) and SPARC (osteonectin)] in association with mineralized-tissue formation by fetal-rat calvarial cells in culture. Biochem J. 1991;274:513–20.
Oldberg A, Jirskog-Hed B, Axelsson S, Heinegard D. Regulation of bone sialoprotein mRNA by steroid hormones. J Cell Biol. 1989;109:3183–6.
Lynch MP, Stein JL, Stein GS, Lian JB. The influence of type I collagen on the development and maintenance of the osteoblast phenotype in primary and passaged rat calvarial osteoblasts: modification of expression of genes supporting cell growth, adhesion, and extracellular matrix mineralization. Exp Cell Res. 1995;216:35–45.
Beck GR Jr. Inorganic phosphate as a signaling molecule in osteoblast. J Cell Biochem. 2003;90:234–43.
Acknowledgement
This work was supported by the Korea Research Foundation Grant funded by the Korean Government (MOEHRD, Basic Research Promotion Fund) (KRF-2006-311-D00056).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jung, JY., Hong, YJ., Choi, Y.S. et al. A new method for the preparation of bioactive calcium phosphate films hybridized with 1α,25-dihydroxyvitamin D3 . J Mater Sci: Mater Med 20, 2441–2453 (2009). https://doi.org/10.1007/s10856-009-3817-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-009-3817-y