Abstract
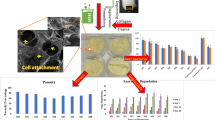
This study aims to develop a novel wound dressing comprising salmon milt DNA (sDNA) and salmon collagen (SC). The sDNA/SC composites were prepared by incubating a mixture of an acidic SC solution, an sDNA solution, and a collagen fibrillogenesis inducing buffer (pH 6.8) containing a crosslinking agent (water-soluble carbodiimide) for gelation, and a subsequent ventilation-drying process to give sDNA/SC films. The conjugation between sDNA and SC were confirmed by sDNA-elution assay and fluorescence microscopy. The sDNA/SC films with various doses of sDNA (sDNA/SC weight ratios of 1:5, 1:10, and 1:20) were used for in vitro cell cultures to evaluate their growth potentials of normal human dermal fibroblasts (NHDF) and normal human epidermal keratinocytes (NHEK). It was found that NHDF proliferation was increased by sDNA conjugation, whereas NHEK proliferation was dose-dependently inhibited. In light of the in vitro results, the appropriate dose of sDNA for in vivo study was determined to be the ratio of 1:10. For the implantation in full-thickness skin defects in rat dorsal region, the sDNA/SC films were reinforced by incorporating them on a porous SC sponge, because the sDNA/SC films exhibited early contraction and inadequate morphologic stability when implanted in vivo. The regenerated tissue in the sDNA/SC sponge group showed similar morphology to native dermis, while the SC sponge group without sDNA showed epithelial overgrowth, indicating that additional sDNA could reduce epidermal overgrowth. Furthermore, blood capillary formation was significantly enhanced in the sDNA/SC sponge group when compared to the SC sponge group. In conclusion, the results suggest that the sDNA/SC composite could be a potential wound dressing for clinical applications.






Similar content being viewed by others
References
S.K. Purna, M. Babu, Burns 26, 54 (2000). doi:10.1016/S0305-4179(99)00103-5
D. Queen, H. Orsted, H. Sanada, G. Sussman, Int. Wound J. 1, 59 (2004). doi:10.1111/j.1742-4801.2004.0009.x
A.F. Falabella, Dermatol. Ther. 19, 317 (2006). doi:10.1111/j.1529-8019.2006.00090.x
J.A. Ramshaw, J.A. Werkmeister, V. Glattauer, Biotechnol. Genet. Eng. Rev. 13, 335 (1996)
C.J. Doillon, F.H. Silver, Biomaterials 7, 3 (1986). doi:10.1016/0142-9612(86)90080-3
M. Ishihara, K. Nakanishi, K. Ono, M. Sato, M. Kikuchi, Y. Saito et al., Biomaterials 23, 833 (2002). doi:10.1016/S0142-9612(01)00189-2
C.H. Lee, A. Singla, Y. Lee, Int. J. Pharm. 221, 1 (2001). doi:10.1016/S0378-5173(01)00691-3
M. Mian, F. Beghe, E. Mian, Int. J. Tissue React. 14(Suppl), 1 (1992)
E. Song, S. Yeon Kim, T. Chun, H.J. Byun, Y.M. Lee, Biomaterials 27, 2951 (2006). doi:10.1016/j.biomaterials.2006.01.015
M.M. Giraud-Guille, L. Besseau, C. Chopin, P. Durand, D. Herbage, Biomaterials 21, 899 (2000). doi:10.1016/S0142-9612(99)00244-6
S. Yunoki, N. Nagai, T. Suzuki, M. Munekata, J. Biosci. Bioeng. 98, 40 (2004)
N. Nagai, K. Mori, Y. Satoh, N. Takahashi, S. Yunoki, K. Tajima et al., J. Biomed. Mater. Res. A 82, 395 (2007). doi:10.1002/jbm.a.31110
N. Nagai, S. Yunoki, Y. Satoh, K. Tajima, M. Munekata, J. Biosci. Bioeng. 98, 493 (2004)
M. Yamada, K. Kato, K. Shindo, M. Nomizu, M. Haruki, N. Sakairi et al., Biomaterials 22, 3121 (2001). doi:10.1016/S0142-9612(01)00061-8
J.J.J.P. Van Den Beucken, M.R.J. Vos, P.C. Thune, T. Hayakawa, T. Fukushima, Y. Okahata et al., Biomaterials 27, 691 (2006). doi:10.1016/j.biomaterials.2005.06.015
W. Liu, S. Sun, Z. Cao, X. Zhang, K. Yao, W.W. Lu et al., Biomaterials 26, 2705 (2005). doi:10.1016/j.biomaterials.2004.07.038
E. Holen, O.A. Bjørge, R. Jonsson, Nutrition 22, 90 (2006). doi:10.1016/j.nut.2006.01.001
T. Fukushima, T. Hayakawa, Y. Inoue, K. Miyazaki, Y. Okahata, Biomaterials 25, 5491 (2004). doi:10.1016/j.biomaterials.2004.01.006
M. Yamada, K. Kato, M. Nomizu, N. Sakairi, K. Ohkawa, H. Yamamoto et al., Chemistry (Easton) 8, 1407 (2002)
N. Nagai, S. Yunoki, T. Suzuki, M. Sakata, K. Tajima, M. Munekata, J. Biosci. Bioeng. 97, 389 (2004)
L.H. Olde Damink, P.J. Dijkstra, M.J. Van Luyn, P.B. Van Wachem, P. Nieuwenhuis, J. Feijen, Biomaterials 17, 765 (1996). doi:10.1016/0142-9612(96)81413-X
K.E. Kaldler, D.F. Holmes, J.A. Trotter, J.A. Chapman, Biochem. J. 316(Pt 1), 1 (1996)
S. Thellung, T. Florio, A. Maragliano, G. Cattarini, G. Schettini, Life Sci. 64, 1661 (1999). doi:10.1016/S0024-3205(99)00104-6
P.W. Cook, N.M. Ashton, M.R. Pittelkow, J. Invest. Dermatol. 104, 976 (1995). doi:10.1111/1523-1747.ep12606228
M.C. Montesinos, A. Desai, J.F. Chen, H. Yee, M.A. Schwarzschild, J.S. Fink et al., Am. J. Pathol. 160, 2009 (2002)
Acknowledgement
Shen thanks the Japan Society for the Promotion of Science (JSPS) for a post-doctoral fellowship for foreign researchers.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, X., Nagai, N., Murata, M. et al. Development of salmon milt DNA/salmon collagen composite for wound dressing. J Mater Sci: Mater Med 19, 3473–3479 (2008). https://doi.org/10.1007/s10856-008-3512-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-008-3512-4