Abstract
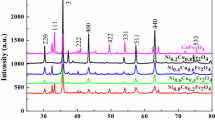
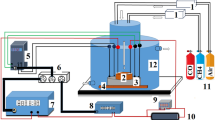
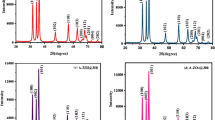
Resistive-based gas sensors have been examined as the most favourable gas sensors for toxic gases and volatile organic compounds owing to their low cost, ease of use, high stability and simple structure. In this work, ammonia gas sensing self-assembled nanostructure material Ni0.5ZnxMg0.5−xFe2O4 (x = 0.00, 0.15, 0.30, 0.45) were successfully synthesized by the modified solution combustion method and examined for the structural, morphological, dielectric and gas sensing properties by X-ray diffraction, field emission scanning electron microscopy, impedance analyzer and the gas sensing setup. The dielectric properties were enhanced by Zn2+ doping. The ammonia vapour sensor based on Ni0.5ZnxMg0.5−xFe2O4 (x = 0.00, 0.15, 0.30, 0.45) shows interesting p-type behaviour gas sensing characteristics at the operational temperature of 300 K. The response to 50 ppm ammonia is 576%, 538%, 423% and 467% for x = 0.00, 0.15, 0.30, 0.45 compositions of Ni0.5ZnxMg0.5−xFe2O4 respectively while as the response and recovery time for all compositions, is in the range of 50–58 s and 64–83 s respectively. Moreover, the ammonia (reducing gas) gas sensing mechanism is also presented. The observed interesting results like sensor response and selectivity propose that the self-assembled Ni0.5ZnxMg0.5−xFe2O4 can be assuredly applied in ammonia vapour detection.















Similar content being viewed by others
Data availability
All data generated or analyzed during the present study are included in this manuscript [No supplementary files].
References
G. Korotcenkov, Metal oxides for solid-state gas sensors: what determines our choice? Mater. Sci. Eng. B 139(1), 1–23 (2007). https://doi.org/10.1016/j.mseb.2007.01.044
R. Nasr Azadani, M. Sabbagh, H. Salehi, A. Cheshmi, A. Raza, B. Kumari, G. Erabi, Sol-gel: uncomplicated, routine and affordable synthesis procedure for utilization of composites in drug delivery: review. J. Compos. Compd. 3(6), 57–70 (2021). https://doi.org/10.52547/jcc.3.1.6
F. Niazvand, P.R. Wagh, E. Khazraei, M. Borzouyan Dastjerdi, C. Patil, I.A. Najar, Application of carbon allotropes composites for targeted cancer therapy drugs: a review. J. Compos. Compd. 3(7), 140–151 (2021). https://doi.org/10.52547/jcc.3.2.7
Z. Shariatinia, Carboxymethyl chitosan: properties and biomedical applications. Int. J. Biol. Macromol. 120(Pt B), 1406–1419 (2018). https://doi.org/10.1016/j.ijbiomac.2018.09.131
B. Fonseca-santos, M. Chorilli, An overview of carboxymethyl derivatives of chitosan: their use as biomaterials and drug delivery systems. Mater. Sci. Eng.: C 77(2017), 1349–1362 (2017). https://doi.org/10.1016/j.msec.2017.03.198
A. Khan, K.A. Alamry, Recent advances of emerging green chitosan-based biomaterials with potential biomedical applications: a review. Carbohydr. Res. 506, 108368 (2021). https://doi.org/10.1016/j.carres.2021.10836
S. Zhang, B. Cheng, Z. Jia, Z. Zhao, X. Jin, Z. Zhao, G. Wu, The art of framework construction: hollow-structured materials toward high-efficiency electromagnetic wave absorption. Adv. Compos. Hybrid Mater. (2022). https://doi.org/10.1007/s42114-022-00514-2
H.W. Jang, A. Zareidoost, M. Moradi, A. Abuchenari, A. Bakhtiari, R. Pouriamanesh, B. Malekpouri, A.J. Rad, D. Rahban, Photosensitive nanocomposites: environmental and biological applications. J. Compos. Compd. 2(2), 50–60 (2020). https://doi.org/10.29252/jcc.2.1.7
M. Qayoom, K.A. Shah, A.H. Pandit et al., Dielectric and electrical studies on iron oxide (α-Fe2O3) nanoparticles synthesized by modified solution combustion reaction for microwave applications. J. Electroceram. 45, 7–14 (2020). https://doi.org/10.1007/s10832-020-00219-2
J. Daraei, Production and characterization of PCL (polycaprolactone) coated TCP/nanoBG composite scaffolds by sponge foam method for orthopedic applications. J. Compos. Compd. 2(2), 44–49 (2020). https://doi.org/10.29252/jcc.2.1.6
M.A. Ahmed, E. Ateia, S. El-Dek, Rare earth doping effect on the structural and electrical properties of Mg–Ti ferrite. Mater. Lett. 57, 4256–4266 (2003). https://doi.org/10.1016/S0167-577X(03)00300-8
T. Meaz, S. Attia, A. Ata, Effect of tetravalent titanium ions substitution on the dielectric properties of Co–Zn ferrites. J. Magn. Magn. Mater. 257, 296–305 (2003). https://doi.org/10.1016/S0304-8853(02)01212-X
R.S. Yadav, I. Kuřitka, J. Vilcakova, J. Havlica, L. Kalina, P. Urbánek, M. Machovsky, M. Masař, M. Holek, Influence of La3+ on structural, magnetic, dielectric, and electrical and modulus spectroscopic characteristics of single phase CoFe2−xLaxO4 nanoparticles. J. Mater. Sci.: Mater. Electron. 28, 9139–9154 (2017). https://doi.org/10.1007/s10854-017-6648-5
B.P. Jacob, S. Thankachan, S. Xavier, E.M. Mohammed, Effect of Tb3+ substitution on structural, electrical and magnetic properties of sol–gel synthesized nanocrystalline nickel ferrite. J. Alloys Compd. 578, 314–319 (2013)
V.A. Adole, T.B. Pawar, P.B. Koli, B.S. Jagdale, Exploration of catalytic performance of nano-La2O3 as an efficient catalyst for dihydropyrimidinone/thione synthesis and gas sensing. J. Nanostruct. Chem. 9, 61–76 (2019). https://doi.org/10.1007/s40097-019-0298-5
P. Rao, R.V. Godbole, S. Bhagwat, Nanocrystalline Pd:NiFe2O4 thin films: a selective ethanol gas sensor. J. Magn. Magn. Mater. (2016). https://doi.org/10.1016/j.jmmm.2016.05.021
A. Sutka, G. Mezinskis, A. Lusis, M. Stingaciu, Gas sensing properties of Zn-doped p-type nickel ferrite. Sens. Actuators B 171–172, 354–360 (2012). https://doi.org/10.1016/j.snb.2012.04.059
M.A. Amer, M. El Hiti, Mössbauer and X-ray studies for Ni0.2ZnxMg0.8−xFe2O4 ferrites. J. Magn. Magn. Mater. 234, 118 (2001)
D. Maity, K. Rajavel, R.T. Kumar, Polyvinyl alcohol wrapped multiwall carbon nanotube (MWCNTs) network on fabrics for wearable room temperature ethanol sensor. Sens. Actuators B 261, 297–306 (2018). https://doi.org/10.1016/j.snb.2018.01.152
S. Pishar et al., Effect of synthesis method and morphology on the enhanced CO2 sensing properties of magnesium ferrite MgFe2O4. Ceram. Int. 44(15), 18578–18584 (2018). https://doi.org/10.1016/j.ceramint.2018.07.082
M.N. Akhtar, A. Rahman, A.B. Sulong, M.A. Khan, Structural, spectral, dielectric and magnetic properties of Ni0.5MgxZn0.5−xFe2O4 nanosized ferrites for microwave absorption and high frequency applications. Ceram. Int. 43(2016), 4357–4365 (2016)
F.A. Hezam, N.O. Khalifa, O. Nur, M.A. Mustafa, Synthesis and magnetic properties of Ni0.5MgxZn0.5−xFe2O4 (0.0 ≤ x ≤ 0.5) nanocrystalline spinel ferrites. Mater. Chem. Phys. (2020). https://doi.org/10.1016/j.matchemphys.2020.123770
M. Qayoom, G.N. Dar, Crystallite size and compressive lattice strain in NiFe2O4 nanoparticles as calculated in terms of various models: influence of annealing temperature. Int. J. Self-Propag. High-Temp. Synth. 29, 213–219 (2020). https://doi.org/10.3103/S1061386220040111
P. Bindu, S. Thomas, Estimation of lattice strain in ZnO nanoparticles: X-ray peak profile analysis. J. Theor. Appl. Phys. 8, 123–134 (2014). https://doi.org/10.1007/s40094-014-0141-9
X. Wang, M. Qin, F. Fang, B. Jia, H. Wu, X. Qu, A.A. Volinsky, Solution combustion synthesis of nanostructured iron oxides with controllable morphology, composition and electrochemical performance. Ceram. Int. (2017). https://doi.org/10.1016/j.ceramint.2017.12.004
R. Bhat, M. Qayoom, G.N. Dar, B. Want, Structural, dielectric, optical and magnetic studies of dysprosium doped iron oxide nanostructures. Mater. Chem. Phys. 245, 122764 (2020). https://doi.org/10.1016/j.matchemphys.2020.122764
M. Qayoom, R. Bhat, K. Asokan, M.A. Shah, G.N. Dar, Unary doping effect of A2+ (A = Zn Co, Ni) on the structural, electrical and magnetic properties of substituted iron oxide nanostructures. J. Mater. Sci.: Mater. Electron. 31, 8268–8282 (2020). https://doi.org/10.1007/s10854-020-03362-2
E.E. Ateia, G. Abdelatif, M.A. Ahmed, M. Mahmoud, Effect of different Gd3+ ion content on the electric and magnetic properties of lithium antimony ferrite. J. Inorg. Organomet. Polym. 26, 81–90 (2016). https://doi.org/10.1007/s10904-015-0283-5
C.C. Naik, S.K. Gaonkar, I. Furtado, A.V. Salker, Effect of Cu2+ substitution on structural, magnetic and dielectric properties of cobalt ferrite with its enhanced antimicrobial property. J. Mater. Sci.: Mater. Electron. 29, 14746–14761 (2018). https://doi.org/10.1007/s10854-018-9611-1
M.H. Dhaou, S. Hcini, A. Mallah, M.L. Bouazizi, A. Jemni, Structural and complex impedance spectroscopic studies of ferrite nanoparticle. Appl. Phys. A (2016). https://doi.org/10.1007/s00339-016-0652-0
Y.D. Kolekar, L.J. Sanchez, C.V. Ramana, Dielectric relaxations and alternating current conductivity in manganese substituted cobalt ferrite. J. Appl. Phys. 115, 144106–144116 (2014). https://doi.org/10.1063/1.4870232
C. Rayssi, S.E. Kossi, J. Dhahri, K. Khirouni, Frequency and temperature-dependence of dielectric permittivity and electric modulus studies of the solid solution Ca0.85Er0.1Ti1−xCo4x/3O3 (0 ≤ x ≤ 0.1). RSC Adv. (2018). https://doi.org/10.1039/C8RA00794B
P.P. Subha, M.K. Jayaraj, Enhanced room temperature gas sensing properties of low temperature solution processed ZnO/CuO heterojunction. BMC Chem. 13, 4 (2019). https://doi.org/10.1186/s13065-019-0519-5
M. Qayoom, K.A. Shah, A. Firdous, G.N. Dar, Synthesis of sodium acetate oriented Ni(II)-doped iron oxide nanospheres for efficient acetone gas sensing. Sens. Int. 3(2022), 100150 (2022). https://doi.org/10.1016/j.sintl.2021.100150
S.G. Gawas, V.M. Verenkar, Selective sensing of oxidizing gases on Co–Ni–Zn ferrite: mechanism and response characteristics. Mater. Sci. Eng. B 265, 114948 (2021). https://doi.org/10.1016/j.mseb.2020.114948
S.D. Raut, V.V. Awasarmol, B.G. Ghule, S.F. Shaikh, S.K. Gore, R.P. Sharma, P.P. Pawar, R.S. Mane, Enhancement in room-temperature ammonia sensor activity of size-reduced cobalt ferrite nanoparticles on γ-irradiation. Mater. Res. Express (2018). https://doi.org/10.1088/2053-1591/aac99d
R. Kashyap, R. Kumar, S. Devi, M. Kumar, S. Tyagi, D. Kumar, Ammonia gas sensing performance of nickel ferrite nanoparticles. Mater. Res. Express (2019). https://doi.org/10.1088/2053-1591/ab55b5
U.B. Tumberphale, S.S. Jadhav, S.D. Raut, P.V. Shinde, S. Sangle, S.F. Shaikh, A.M. Al-Enizi, M. Ubaidullah, R.S. Mane, S.K. Gore, Tailoring ammonia gas sensing performance of La3+-doped copper cadmium ferrite nanostructures. Solid State Sci. (2019). https://doi.org/10.1016/j.solidstatesciences.2019.106089
P.B. Koli, K.H. Kapadnis, U.G. Deshpande, Nano-crystalline modified nickel ferrite films: an effective sensor for industrial and environmental gas pollutant detection. J. Nanostruct. Chem. (2019). https://doi.org/10.1007/s40097-019-0300-2
R.R. Powar, V.D. Phadtare, V.G. Parale, S. Pathak, K.R. Sanadi, H.H. Park, D.R. Patil, P.B. Piste, D.N. Zambare, Effect of zinc substitution on magnesium ferrite nanoparticles: structural, electrical, magnetic, and gas-sensing properties. Mater. Sci. Eng. B 262, 114776 (2020). https://doi.org/10.1016/j.mseb.2020.114776
D. Deivatamil, J.A. Mark, T. Raghavan, J.P. Jesuraj, Fabrication of MnFe2O4 and Ni:MnFe2O4 nanoparticles for ammonia gas sensor application. Inorg. Chem. Commun. 123, 108355 (2021). https://doi.org/10.1016/j.inoche.2020.108355
N. Joshi, T. Hayasaka, Y. Liu, H. Liu, O.N. Oliveira, L. Lin, A review on chemiresistive room temperature gas sensors based on metal oxide nanostructures, graphene and 2D transition metal dichalcogenides. Microchim. Acta 185, 213 (2018). https://doi.org/10.1007/s00604-018-2750-5
H.-J. Kim, J.-H. Lee, Highly sensitive and selective gas sensors using p-type oxide semiconductors: overview. Sens. Actuators B 92, 607–627 (2014). https://doi.org/10.1016/j.snb.2013.11.005
S. Thirumalairajan, A novel organic pollutants gas sensing material p-type CuAlO2 microsphere constituted of nanoparticles for environmental remediation. Sens. Actuators B 223, 138–148 (2015). https://doi.org/10.1016/j.snb.2015.09.092
Acknowledgements
This work is supported by the department of science and technology, government of India vides Reference No: DST/TM/WTI/2K16/248 (G) and inter-university accelerator centre (IUAC), New Delhi vide Reference No: IUAC/XIII.7/UFR-63304.
Funding
This work is supported by the DST, New Delhi [Grant No. DST/TM/WTI/2K16/248 (G)], Inter-University Accelerator Centre [Grant No. IUAC/XIII.7/UFR-63304].
Author information
Authors and Affiliations
Contributions
MQ: Conceptualization, Methodology, Writing—original draft. SI: Methodology, Software, Analysis. GFM: Conceptualization, Methodology, Software, Analysis. KAS: Writing—review & editing, Conceptualization, Methodology, Formal analysis, Investigation, Supervision. MQL: Conceptualization, Investigation. RTRK: Writing—review & editing, Conceptualization, Methodology, Experiments. GND: Writing—review & editing, Conceptualization, Methodology, Formal analysis, Investigation, Supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this research article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qayoom, M., Irfan, S., Malik, G.F. et al. Modified solution combustion-grown Zn-doped Ni–Mg ferrite nanostructures for room temperature NH3 sensing. J Mater Sci: Mater Electron 33, 25645–25660 (2022). https://doi.org/10.1007/s10854-022-09261-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-022-09261-y