Abstract
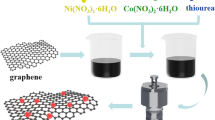
Transition metal chalcogenides have fascinating characteristics are considered as electrode materials for high-performance energy storage devices. Herein, we report the CoS/G nanocomposite was successfully synthesized by a simple one-pot hydrothermal method. The phase formation and morphology of the obtained materials were analyzed by various techniques. The electrochemical properties of the prepared electrode materials were assessed from cyclic voltammetry in a three-electrode system. The obtained cyclic voltammetry curves demonstrate pseudocapacitive behavior for prepared materials due to the synergistic effect between Cobalt and sulfur. The Galvanometric Charge Discharge (GCD) profile of the materials confirms the pseudocapacitive nature and specific capacitance calculated from these curves. The CoS/G nanocomposite delivered high specific capacitance 739.83 Fg−1 compared to the pure CoS nanospheres 390 Fg−1 and prolonged cyclic stability with 91.2% capacity retention after 3000 cycles. Overall, these excellent electrochemical performances indicate the CoS/G nanocomposite as a desirable electrode material for commercial supercapacitor applications.









Similar content being viewed by others
Data availability
Authors can confirm that all relevant data are included in the article.
References
P. Simon, Y. Gogotsi, Materials for electrochemical capacitors. Nat. Mater. 7, 845–854 (2008)
Y. Xiaowei, Z. Junwu, Q. Ling, L. Dan, Bioinspired effective prevention of restacking in multilayered graphene films: towards the next generation of high-performance supercapacitors. Adv. Mater. 23, 2833–2838 (2011). https://doi.org/10.1002/adma.201100261
Z. Haitao, Z. Xiong, Z. Dacheng, S. Xianzhong, He. Lin, W. Changhui, M. Yanwei, One-step electrophoretic deposition of reduced graphene oxide and Ni(OH)2 composite films for controlled syntheses supercapacitor electrodes. J. Phys. Chem. B 117, 1616–1627 (2013). https://doi.org/10.1021/jp305198j
K. Seevakan, A. Manikandan, P. Devendran, Y. Slimani, A. Baykal, T. Alagesan, Structural, magnetic and electrochemical characterizations of Bi2Mo2O9 nanoparticle for supercapacitor application. J. Magn. Magn Mater 486(15), 165254 (2019)
K. Seevakan, A. Manikandan, P. Devendran, Y. Slimani, A. Baykal, T. Alagesan, Structural, morphological and magneto-optical properties of CuMoO4 electrochemical nanocatalyst as supercapacitor electrode. Ceram. Int. 44(16), 20075–20083 (2018)
O.G. Gnonhoue, A. Velazquez-Salazar, É. David, I. Preda, Review of technologies and materials used in high-voltage film capacitors. Polymers 13(5), 766 (2021). https://doi.org/10.3390/polym13050766
A. Abdelkareem, M. Ali, E. Khaled, W. Tabbi, K. Mohammed, S.E. Taha, A. Olabi, Environmental aspects of fuel cells: a review. Sci. Total Environ. 752, 141803 (2020). https://doi.org/10.1016/j.scitotenv.2020.141803
Z. Luojiang, W. Haitao, Z. Xiaoming, T. Yongbing, A review of emerging dual-ion batteries: fundamentals and recent advances. Adv. Func. Mater. 31, 20 (2021). https://doi.org/10.1002/adfm.202010958
J. Xie, Y.C. Lu, A retrospective on lithium-ion batteries. Nat. Commun. 11, 2499 (2020). https://doi.org/10.1038/s41467-020-16259-9
Z. Meng, L. Bo-Quan, Z. Xue-Qiang, H. Jia-Qi, Z. Qiang, A perspective toward practical lithium-sulfur batteries ACS cent. Sci. 6(7), 1095–1104 (2020)
K. Binoy, S. Saikiaa, B. Maria, B. Mousumi, T. Joyshil, P. Mayank, B. Dhurbajyoti, A brief review on supercapacitor energy storage devices and utilization of natural carbon resources as their electrode materials. Fuel 282, 118796 (2020). https://doi.org/10.1016/j.fuel.2020.118796
R. Chen, Y. Miao, R.P. Sahu, K.P. Ishwar, Z. Igor, The development of pseudocapacitor electrodes and devices with high active mass loading. Adv. Energy Mater. (2020). https://doi.org/10.1002/aenm.201903848
L.L. Zhang, R. Zhou, X.S. Zhao, Graphene-based materials as supercapacitor electrodes. J. Mater. Chem. 20, 5983–5992 (2010). https://doi.org/10.1039/C000417K
R. Dubey, V. Guruviah, Review of carbon-based electrode materials for supercapacitor energy storage. Ionics 25(4), 1419–1445 (2019). https://doi.org/10.1007/s11581-019-02874-0
X. Zhang, H. Zhang, Z. Lin, M. Yu, X. Lu, Y. Tong, Recent advances and challenges of stretchable supercapacitors based on carbon materials. Sci. China Mater. 59(6), 475–494 (2016). https://doi.org/10.1007/s40843-016-5061-1
I.K. Durga, S.S. Rao, M. Jagadeesh, A.E. Reddy, T. Anitha, H.J. Kim, Synthesis of nanostructured metal sulfides via a hydrothermal method and their use as an electrode material for supercapacitors. New J. Chem. 42, 19183–19192 (2018)
R. Chen, Y. Miao, R.P. Sahu, K.P. Ishwar, I. Zhitomirsky, The development of pseudocapacitor electrodes and devices with high active mass loading. Adv. Energy Mater. (2020). https://doi.org/10.1002/aenm.201903848
V. Augustyn, P. Simon, B. Dunn, Pseudocapacitive oxide materials for high-rate electrochemical energy storage. Energy Environ. Sci. 7(5), 1597–1614 (2014). https://doi.org/10.1039/C3EE44164D
S. Khamlich, Z. Abdullaeva, J.V. Kennedy, M. Maaza, High performance symmetric supercapacitor based on zinc hydroxychloride nanosheets and 3D graphene-nickel foam composite. Appl. Surf. Sci. 405(31), 329–336 (2017)
J. Ge, B. Wang, J. Wang, Q. Zhang, B. Lu, Nature of FeSe2/N-C anode for high performance potassium ion hybrid capacitor. Adv. Energy Mater. 10(4), 1903277 (2020)
Y. Zhu, S. Murali, W. Cai, X. Li, J.W. Suk, J.R. Potts, R.S. Ruoff, Graphene and graphene oxide: synthesis, properties, and applications. Adv. Mater. 22, 3906–3924 (2010). https://doi.org/10.1002/adma.201001068
K. Suenne, Z. Si, H. Yike, A. Muge, J. Yves, C.B. Chabal, H. de Walt, B. Angelo, R. Elisa, Room-temperature metastability of multilayer graphene oxide films. Nat. Mater. 11, 544–549 (2012). https://doi.org/10.1038/nmat3316
W. Sun, G. Gao, Y. Du, K. Zhang, G. Wu, A facile strategy for fabricating hierarchical nanocomposites of V2O5 nanowire arrays on a three-dimensional N-doped graphene aerogel with a synergistic effect for supercapacitors. J. Mater. Chem. A 6, 9938–9947 (2018)
K. Gopalakrishnan, S. Sultan, A. Govindaraj, C.N.R. Rao, Supercapacitors based on composites of PANI with nanosheets of nitrogen doped RGO, BC1.5N, MoS2 and WS2. Nano Energy 12, 52–58 (2015)
B. Xie, Y. Chen, Yu. Mengying, Tu. Sun, Lu. Luhua, T. Xie, Y. Zhang, Wu. Yucheng, Hydrothermal synthesis of layered molybdenum sulfide/N-doped graphene hybrid with enhanced supercapacitor performance. Carbon 99, 35–42 (2016). https://doi.org/10.1016/j.carbon.2015.11.077
R. Balu, A. Dakshanamoorthy, A simple hydrothermal synthesis of cadmium sulfide wrapped on graphene nanocomposite for supercapacitor applications. J. Nanosci. Nanotechnol. 21(12), 5835–5845 (2021). https://doi.org/10.1166/jnn.2021.19503
R. Balu, A. Dakshanamoorthy, Synthesis of wool ball-like copper sulfide nanospheres embedded graphene nanocomposite as electrode for high performance symmetric supercapacitor device. Int. J. Energy Res. 24, 1–15 (2021)
R. Balu, A. Dakshanamoorthy, One-pot preparation of tin sulfide decorated graphene nanocomposite for high performance supercapacitor applications. Inorg. Chem. Commun. 136, 109148 (2022)
C.A. Pandey, S. Ravuri, R. Ramachandran, R. Santhosh, S. Ghosh, S.R. Sitaraman, A.N. Grace, Grace synthesis of NiS–graphene nanocomposites and its electrochemical performance for supercapacitors. Int. J. Nanosci. 17, 1760021 (2018). https://doi.org/10.1142/S0219581X17600213
R. Ramachandran, M. Saranya, P. Kollu, B.P. Raghupathy, S.K. Jeong, A.N. Grace, Solvothermal synthesis of Zinc sulfide decorated Graphene (ZnS/G) nanocomposites for novel supercapacitor electrodes. Electrochim. Acta 178(1), 647–657 (2015)
M. Mao, L. Mei, L. Wu, Q. Li, M. Zhang, Facile synthesis of cobalt sulfide/carbon nanotube shell/core composites for high performance supercapacitors. RSC Adv. 4, 12050–12056 (2014). https://doi.org/10.1039/C4RA00485J
F. Tao, Y.Q. Zhao, G.Q. Zhang, H.L. Li, Electrochemical characterization on cobalt sulfide for electrochemical supercapacitors. Electrochem. Commun. 9, 1282–1287 (2007). https://doi.org/10.1016/j.elecom.2006.11.022.19
K.J. Huang, J.Z. Zhang, G. Shi, Y.M. Shi, One-step hydrothermal synthesis of two-dimensional cobalt sulfide for high-performance supercapacitors. Mater. Lett. 131, 45–48 (2014). https://doi.org/10.1016/j.matlet.2014.05.148
Z. Yang, C.Y. Chen, H.T. Chang, Supercapacitors incorporating hollow cobalt sulfide hexagonal nanosheets. J. Power Sources 196, 7874–7877 (2011). https://doi.org/10.1016/j.jpowsour.2011.03.072
K.J. Huang, J.Z. Zhang, G.W. Shi, Y.M. Liu, One-step hydrothermal synthesis of two-dimensional cobalt sulfide for high-performance supercapacitors. Mater. Lett. 131, 45–48 (2014). https://doi.org/10.1016/j.matlet.2014.05.148
M.M. Shahid, A. Pandikumar, A.M. Golsheikh, N.M. Huang, H.N. Lim, Enhanced electrocatalytic performance of cobalt oxide nanocubes incorporating reduced graphene oxide as a modified platinum electrode for methanol oxidation. RSC Adv. 4, 62793–62801 (2014). https://doi.org/10.1039/C4RA08952A
S.J. Peng, L.L. Li, X.P. Han, W.P. Sun, M. Srinivasan, F.Y. Cheng, Q.Y. Yan, J. Chen, S. Ramakrishna, Cobalt sulfide nanosheet/graphene/carbon nanotube nanocomposites as flexible electrodes for hydrogen evolution. Angew. Chem. Int. Ed. 53, 12594–12599 (2014). https://doi.org/10.1002/anie.201408876
W.S. Hummers Jr., R.E. Offeman, Preparation of graphitic oxide. J. Am. Chem. Soc. 80, 1339–1339 (1958)
E. Sathiyaraj, S. Thirumaran, Structural, morphological and optical properties of iron sulfide, cobalt sulfide, copper sulfide, zinc sulfide and copper-iron sulfide nanoparticles synthesized from single source precursors. Chem. Phys. Lett. (2019). https://doi.org/10.1016/j.cplett.2019.136972
F. Tuinstra, J.L. Koening, Raman spectrum of graphite. J. Chem. Phys. 53, 1126–1130 (1970). https://doi.org/10.1063/1.1674108
C.J. Fu, G.G. Zhao, H.J. Zhang, S. Li, A facile route to controllable synthesis of Fe3O4/graphene composites and their application in lithium-ion batteries. Int. J. Electrochem. Sci. 9, 46–60 (2014)
S. Stankovich, D.A. Dikin, R.D. Piner, K.A. Kohlhaas, A. Kleinhammes, Y.Y. Jia, Y. Wu, S.T. Nguyen, R.S. Ruoff, Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 45, 1558–1565 (2007). https://doi.org/10.1016/j.carbon.2007.02.034
C. Xu, Y. Jing, J.R. He, K.R. Zhou, Y.F. Chen, Q. Li, J. Lin, W.L. Zhang, Self-assembled interwoven CoS2/CNTs/graphene architecture as anode for high-performance lithium ion batteries. J. Alloys Compd. 708, 1178–1183 (2017). https://doi.org/10.1016/j.jallcom.2017.03.099
R. Ramachandran, S. Felix, M. Saranya, C. Santhosh, V. Velmurugan, B.P. Ragupathy, S.K. Jeong, A.N. Grace, Synthesis of cobalt sulfide-graphene (CoS/G) nanocomposites for supercapacitor applications. IEEE Trans. Nanotechnol. 12, 985–990 (2013). https://doi.org/10.1109/TNANO.2013.2278287
V. Chabot, D. Higgins, A. Yu, X. Xiao, Z. Chena, J. Zhang, A review of graphene and graphene oxide sponge: Material synthesis and applications to energy and the environment. Energy Environ. Sci. 7, 1564–1596 (2014). https://doi.org/10.1039/C3EE43385D
M. Jianfei, S. Qian, W. Yaqiong, Y. Hongyan, X. Dan, M.M.F. Choi, Facile fabrication of porous CuS nanotubes using well-aligned [Cu(tu)]Cl⋅1/2H2O nanowire precursors as self-sacrificial templates. Cryst. Growth Des. 9, 2546–2548 (2009). https://doi.org/10.1021/cg8006052.27
L. Guojun, C. Lili, W. Yanying, L. Ying, P. Tao, X. He, A novel cobalt tetranitrophthalocyanine/graphene composite assembled by an in situ solvothermal synthesis method as a highly efficient electrocatalyst for the oxygen reduction reaction in alkaline medium. Phys. Chem. Chem. Phys. 31, 13093–13100 (2013). https://doi.org/10.1039/C3CP51577J.29
T.F. Emiru, D.W. Ayele, Controlled synthesis, characterization and reduction of graphene oxide: a convenient method for large scale production. Egypt. J. Basic Appl. Sci. 4(1), 74–79 (2017)
Q. Wang, L. Jiao, H. Du, Y. Si, Y. Wang, H. Yuan, Co3S4 hollow nanospheres grown on graphene as advanced electrode materials for supercapacitors. J. Mater. Chem. 22, 21387–21391 (2012). https://doi.org/10.1039/C2JM34714H
J. Zhu, W. Zhou, Y. Zhou, X. Cheng, J. Yang, Cobalt Sulfide/reduced graphene oxide nanocomposite with enhanced performance for supercapacitors. J. Electron. Mater. 48, 3 (2019). https://doi.org/10.1007/s11664-018-06910-z
S. Muhammad Mehmood, R. Perumal, P. Alagarsamy, L. Hong Ngee, N. Yun Hau, H. Nay Ming, An electrochemical sensing platform based on a reduced graphene oxide-cobalt oxide nanocube@ platinum nanocomposite for nitric oxide detection. J. Mater. Chem. A 3, 14458–14468 (2015). https://doi.org/10.1039/C5TA02608C
K. Subramani, N. Sudhan, R. Divya, M. Sathish, All-solid-state asymmetric supercapacitors based on cobalt hexacyanoferrate-derived CoS and activated carbon. RSC Adv. 7, 6648–6659 (2017). https://doi.org/10.1039/C6RA27331A
R. Balu, S. Sagadevan, A. Dakshanamoorthy, A cost effective, facile hydrothermal approach of zinc sulfide decorated on graphene nanocomposite for supercapacitor applications. J. Nanosci. Nanotechnol. 19, 6987–6994 (2019). https://doi.org/10.1166/jnn.2019.16670
H. Chauhan, M.K. Singh, P. Kumar, S.A. Hashmi, S. Deka, Development of SnS2/rGO nanosheet composite for cost-effective aqueous hybrid supercapacitors. Nanotechnology 28(2), 025401 (2016). https://doi.org/10.1088/1361-6528/28/2/025401
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
RB: Preparation of samples, Collection of data, and Manuscript drafting, AD: Investigation and analysis of data, Writing-review & editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Balu, R., Dakshanamoorthy, A. A facile one-pot hydrothermal synthesis of cobalt sulfide nanospheres integrated with graphene nanocomposite as electrode material for high-performance supercapacitors. J Mater Sci: Mater Electron 33, 10057–10071 (2022). https://doi.org/10.1007/s10854-022-07996-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-022-07996-2