Abstract
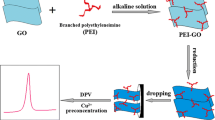
In this paper, a polydopamine-graphene composite gel (PDA-GA) was prepared from graphene oxide by a facile in-situ reduction assembly method using ascorbic acid combined with dopamine (DA). The morphology and microstructure of PDA-GA were characterized by SEM, XRD, FTIR, BET-BJH, and MIP, and the adsorption behavior of PDA-GA for Cu2+ was studied. Furthermore, the response ability of graphene composite gel-modified glassy carbon electrode (PDA-GH-GCE) to trace Cu2+ was investigated by linear sweep anodic stripping voltammetry. Results showed that when the mass ratio of graphene oxide to DA was 1:3, the adsorption effect of PDA-GA for Cu2+ was the best, and the adsorption capacity reached 316 mg/g. PDA-GA-GCE was sensitive to trace Cu2+, and the detection limit could reach 1.6 × 10−7 mol/L.










Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
A. Begum, N. Amin, S. Kaneco et al., Selected elemental composition of the muscle tissue of three species of fish, Tilapia nilotica, Cirrhinamrigala and Clariusbatrachus, from the fresh water Dhanmondi Lake in Bangladesh. Food Chem. 93(3), 439–443 (2005)
A. Cravo, M.J. Bebianno, Bioaccumulation of metals in the soft tissue of Patella aspera: application of metal/shell weight indices. Estuar. Coast. Shelf Sci. 65(3), 571–586 (2005)
H.Y. Song, J.Q. Liu, P. Yin et al., Distribution, enrichment and source of heavy metals in Rizhao offshore area, southeast Shandong Province. Mar. Pollut. Bull. 119(2), 175–180 (2017)
L. Li, S. Wang, X. Shen et al., Ecological risk assessment of heavy metal pollution in the water of China’s coastal shellfish culture areas. Environ. Sci. Pollut. Res. 27(15), 18392–18402 (2020)
M.M. Zhang, P. He, G. Qiao et al., Heavy metal contamination assessment of surface sediments of the Subei Shoal, China: spatial distribution, source apportionment and ecological risk. Chemosphere 223, 211–222 (2019)
J. Tan, L. Zhu, B. Wang, Effects of quercetin on DNA damage induced by copper ion. Int. J. Pharmacol. 3(1), 19–26 (2007)
X.X. Yang, W. Zhang, X.F. Cao et al., Long-term effect of copper with sublethal dose on cytochrome P450 and antioxidant enzyme activities of earthworms. Acta Sci. Circum. 32(3), 745–750 (2012)
P. Ramya, P. Tamilarasi, M. Sukumaran et al., Toxic effect of copper sulphate on biochemical alterations in a estuarine mud crab. Scylla Serrata. 7(5), 15901–15904 (2015)
S. Mukherjee, S. Bhattacharyya, K. Ghosh et al., Sensory development for heavy metal detection: a review on translation from conventional analysis to field-portable sensor. Trends Food Sci. Technol. 109, 674–689 (2021)
S. Huang, S. Lu, C. Huang et al., Sensitive and selective stripping voltammetric determination of copper (II) using a glassy carbon electrode modified with amino-reduced graphene oxide and β-cyclodextrin. Mikrochim. Acta 182(15), 2529–2539 (2015)
Q. Wu, H.M. Bi, X.J. Han, Research progress of electrochemical detection of heavy metal ions. Chin. J. Anal. Chem. 49(3), 330–340 (2021)
X.X. Wang, Y.X. Qi, Y. Shen et al., A ratiometric electrochemical sensor for simultaneous detection of multiple heavy metal ions based on ferrocene-functionalized metal-organic framework. Sens. Actuators B 310, 127756 (2020)
C.M. Quiroa-Montalván, L.E. Gómez-Pineda, L. Álvarez-Contreras et al., Ordered mesoporous carbon decorated with magnetite for the detection of heavy metals by square wave anodic stripping voltammetry. J. Electrochem. Soc. 164(6), B304–B313 (2017)
M. Yun, J.E. Choe, J.M. You et al., High catalytic activity of electrochemically reduced graphene composite toward electrochemical sensing of Orange II. Food Chem. 169, 114–119 (2015)
C. Fei, X. Zhang, Electrochemical sensor for epinephrine based on a glassy carbon electrode modified with graphene/gold nanocomposites. J. Electroanal. Chem. 669, 35–41 (2012)
H. Ahmad, C. Liu, Ultra-thin graphene oxide membrane deposited on highly porous anodized aluminum oxide surface for heavy metal ions preconcentration. J. Hazard. Mater. 415, 125661 (2021)
S. Lee, J. Oh, D. Kim et al., A sensitive electrochemical sensor using an iron oxide/graphene composite for the simultaneous detection of heavy metal ions. Talanta 160, 528–536 (2016)
A. Li, C.J. Pei, Z.Q. Zhu et al., Progress in graphene aerogels. Mod. Chem. Ind. 33(10), 20–23 (2013)
M. Hasanpour, M. Hatami, Application of three dimensional porous aerogels as adsorbent for removal of heavy metal ions from water/wastewater: a review study. Adv. Colloid Interface Sci. 284, 102247 (2020)
V. Suvina, S.M. Krishna, D.H. Nagaraju et al., Polypyrrole-reduced graphene oxide nanocomposite hydrogels: a promising electrode material for the simultaneous detection of multiple heavy metal ions. Mater. Lett. 232, 209–212 (2018)
L. Yihong, Z. Tingyao, H. Rong et al., Synthesis of dopamine-modified graphene aerogels and its adsorption capability for dyes. Ind. Water Treat. 38(10), 67–70 (2018)
T. Li, X. Liu, L. Li et al., Polydopamine-functionalized graphene oxide compounded with polyvinyl alcohol/chitosan hydrogels on the recyclable adsorption of cu(II), Pb(II) and cd(II) from aqueous solution. J. Polym. Res. 26(12), 281 (2019)
S.N. Feng, L.Y. Yu, M.X. Yan et al., Holey nitrogen-doped graphene aerogel for simultaneously electrochemical determination of ascorbic acid, dopamine and uric acid. Talanta 224, 121851 (2021)
X. Yu, P.W. Wu, Y.C. Liu et al., Dopamine and L-arginine tailored fabrication of ultralight nitrogen-doped graphene aerogels for oil spill treatment. J. Fuel Chem. Technol. 45(10), 1230–1235 (2017)
K.L. Zhang, Q.C. Du, C. Yan, Preparation and adsorption property of dopamine reduced graphene oxide aerogel. Mater. Rep. 31(30), 219-221–232 (2017)
M.Z.H. Khan, M. Daizy, C. Tarafder et al., Au-PDA@SiO2 core-shell nanospheres decorated rGO modified electrode for electrochemical sensing of cefotaxime. Sci Rep 9, 19041 (2019)
C. Hou, Q. Zhang, Y. Li et al., P25–graphene hydrogels: room-temperature synthesis and application for removal of methylene blue from aqueous solution. J. Hazard. Mater. 205–206, 229–235 (2012)
X. Mi, G. Huang, W. Xie et al., Preparation of graphene oxide aerogel and its adsorption for Cu2+ ion. Carbon 50(13), 4856–4864 (2012)
J. Zou, F.H. Wang, Y.L. Yang et al., Preparation and adsorptive property of cyclodextrin/graphene oxide composite for Cu2+. China Plast. 33(4), 17–21 (2019)
B.W. Yu, J. Xu, J.H. Liu et al., Adsorption behavior of copper ions on graphene oxide–chitosan aerogel. J. Environ. Chem. Eng. 1(4), 1044–1050 (2013)
L. Pan, Z. Wang, Y. Qi et al., Efficient removal of lead, copper and cadmium ions from water by a porous calcium alginate/graphene oxide composite aerogel. Nanomaterials 8(11), 957 (2018)
J. Xie, J. Li, L. Zhao et al., Fabrication of TiO2-graphene oxide aerogel for the adsorption of copper ions. Nanosci. Nanotechnol. Lett. 6(11), 1018–1023 (2014)
Acknowledgement
The work was supported by National Natural Science Foundation of China (11475017).
Funding
This study was supported by National Natural Science Foundation of China (11475017).
Author information
Authors and Affiliations
Contributions
LY performed the experiment, LY contributed significantly to analysis and manuscript preparation, CH, YB, and HG helped perform the analysis with constructive discussions.
Corresponding author
Ethics declarations
Conflict of interest
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yin, L., Yan, l., Hao, C. et al. Adsorption and trace detection of copper ion by three-dimensional porous graphene composite gel. J Mater Sci: Mater Electron 33, 1966–1976 (2022). https://doi.org/10.1007/s10854-021-07401-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-021-07401-4