Abstract
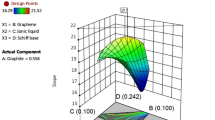
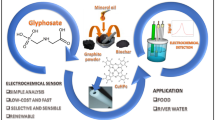
Tetracycline hydrochloride (TC(HCl)) has been extensively used as veterinary drugs and food additives in feed, but the abuse of TC(HCl) poses a great threat to the ecological environment and human health. In this paper, TC(HCl) in aqueous solution was successfully probed by an iron cation exchanged montmorillonite modified glassy carbon electrode (Fe-Mt/GCE) via a direct voltametric method. The electrochemical response sensitivity of Fe-Mt-150/GCE is 27.1 µA µM−1 cm−2. Compared with the traditional electrode, the Fe-Mt/GCE exhibits higher response current intensity in the rapid scanning process, which should be contributed to the larger surface area and more active sites of the modified electrode for the oxidation reaction of TC(HCl). Additionally, it is found that higher scanning corresponds to better signal, implying there are enough active sites in Fe-Mt/GCE for TC(HCl) detection. The machine learning method was further employed to explore the optimum parameters. The predicted data are highly consistent with the experimental data, which were verified by the testing phase in machine learning. The application of machine learning to describe and summarize the data may bring new ideas to the future researches. Montmorillonite could be potentially applied as an electrochemical sensor for antibiotic tetracycline hydrochloride detection due to its low cost and easy fabrication.











Similar content being viewed by others
References
C. Li, L. Zhu, W. Yang, X. He, S. Zhao, X. Zhang, W. Tang, J. Wang, T. Yue, Z. Li, Amino-functionalized Al-MOF for fluorescent detection of tetracyclines in Milk. J. Agric. Food Chem. 67, 1277–1283 (2019)
N. Hassani, A. Baraket, S. Boudjaoui, E. Neto, J. Bausells, N.E. Bari, B. Bouchikhi, A. Elaissari, A. Errachid, N. Zine, Development and application of a novel electrochemical immune sensor for tetracycline screening in honey using a fully integrated electrochemical Bio-MEMS. Biosens. Bioelectron. 130, 330–337 (2019)
T. Wang, Q. Mei, Z. Tao, H. Wu, M. Zhao, S. Wang, Y. Liu, A smart phone-integrated ratio metric fluorescence sensing platform for visual and quantitative point-of-care testing of tetracycline. Biosens. Bioelectron. 148, 111791 (2020)
S. Graslund, B.E. Bengtsson, Chemicals and biological products used in south-east Asian shrimp farming, and their potential impact on the environment—a review. Sci. Total Environ. 280, 93–131 (2001)
L. Ge, H. Li, X. Du, M. Zhu, W. Chen, T. Shi, N. Hao, Q. Liu, K. Wang, Facile one-pot synthesis of visible light-responsive BiPO4/nitrogen doped graphene hydrogel for fabricating label-free photo electrochemical tetracycline aptasensor. Biosens. Bioelectron. 111, 131–137 (2018)
J. Sun, T. Gan, H. Zhu, Y.M. Liu, Direct electrochemical sensing for oxytetracycline in food using a zinc cation-exchanged montmorillonite. Appl. Clay Sci. 101, 598–603 (2014)
J. Wang, R. Cheng, Y. Wang, L. Sun, L. Chen, X. Dai, J. Pan, G. Pan, Y. Yan, Surface-imprinted fluorescence microspheres as ultrasensitive sensor for rapid and effective detection of tetracycline in real biological samples. Sensors Actuators B Chem. 263, 533–542 (2018)
P. Yan, D. Jiang, Y. Tian, L. Xu, J. Qian, H. Li, J. Xia, H. Li, A sensitive signal-on photoelectrochemical sensor for tetracycline determination using visible-light-driven flower-like CN/BiOBr composites. Biosens. Bioelectron. 111, 74–81 (2018)
J.M. Dewdney, L. Maes, J.P. Raynaud, F. Blanc, J.P. Scheid, T. Jackson, S. Lens, C. Verschueren, Risk assessment of antibiotic residues of β-lactams and macrolides in food products with regard to their immuno-allergic potential. Food Chem. Toxicol. 29, 477–483 (1991)
J.A.J. Madden, S.F. Plumier, J. Tang, I. Garaiova, N.T. Plumier, M. Herbison, J.O. Hunter, T. Shimada, L. Cheng, T. Shirakawa, Effect of probiotics on preventing disruption of the intestinal microflora following antibiotic therapy: a double-blind, placebo-controlled pilot study. Int. Immunopharmacol. 5, 1091–1097 (2005)
Q. Wang, W.M. Zhao, Optical methods of antibiotic residues detections: a comprehensive review. Sensors Actuators B Chem. 269, 238–256 (2018)
Y.M. Tian, T. Bu, M. Zhang, X.Y. Sun, P. Jia, Q.Z. Wang, Y.N. Liu, F. Bai, S. Zhao, L. Wang, Metal-polydopamine framework based lateral flow assay for high sensitive detection of tetracycline in food samples. Food Chem. 339, 127854 (2021)
M.X. Feng, G.N. Wang, K. Yang, H.Z. Liu, J.P. Wang, Molecularly imprinted polymer-high performance liquid chromatography for the determination of tetracycline drugs in animal derived foods. Food Control 69, 171–176 (2016)
X.M. Peng, W.D. Luo, J.Q. Wu, F.P. Hu, Y.Y. Hu, L. Xu, G.P. Xu, Y. Jian, H.L. Dai, Carbon quantum dots decorated heteroatom co-doped core-shell Fe0@POCN for degradation of tetracycline via multiply synergistic mechanisms. Chemosphere 268, 128806 (2021)
L. Li, L.H. Shi, J. Jia, O. Eltayeb, W.J. Lu, Y.H. Tang, C. Dong, S.M. Shuang, Red fluorescent carbon dots for tetracycline antibiotics and pH discrimination from aggregation-induced emission mechanism. Sensors Actuators B Chem 332, 129513 (2021)
O.A. Abafe, P. Gatyeni, L. Matika, A multi-class multi-residue method for the analysis of polyether ionphores, tetracyclines and sulfonamides in multi-matrices of animal and aquaculture fish tissues by ultra-high performance liquid chromatography tandem mass spectrometry. Food Addit. Contam. Part A 37, 438–450 (2020)
X.D. Zhao, X.J. Li, Y. Li, X.L. Zhang, F.H. Zhai, T.Z. Ren, Y.T. Li, Metagenomic analysis reveals functional genes in soil microbial electrochemical removal of tetracycline. J. Hazard. Mater. 408, 124880 (2021)
P.H. Wu, Y. Li, P. Wu, C. Yu, Microbial fuel cell-driven alkaline thermal hydrolysis for pretreatment of wastewater containing high concentrations of tetracycline in the cathode chamber. J. Environ. Chem. Eng. 9, 104659 (2021)
H. Guo, Y. Su, Y. Shen, Y. Long, W. Li, In situ decoration of Au nanoparticles on carbon nitride using a single-source precursor and its application for the detection of tetracycline. J. Colloid Interface Sci. 536, 646–654 (2019)
Y. Xu, M. Gao, G. Zhang, X. Wang, J. Li, S. Wang, Electrochemically reduced graphene oxide with enhanced electrocatalytic activity toward tetracycline detection. Chin. J. Catal. 36, 1936–1942 (2015)
C. Mousty, Sensors and biosensors based on clay-modified electrodes—new trends. Appl. Clay Sci. 27, 159–177 (2004)
Y. Li, H. Huang, R. Cui, D. Wang, Z. Yin, D. Wang, L. Zheng, J. Zhang, Y. Zhao, H. Yuan, J. Dong, X. Guo, B. Sun, Electrochemical sensor based on graphdiyne is effectively used to determine Cd2+ and Pb2+ in water. Sensors Actuators B Chem. 332, 129519 (2021)
S. Hassanpoor, N. Rouhi, Electrochemical sensor for determination of trace amounts of cadmium (II) in environmental water samples based on MnO2/RGO nanocomposite. Int. J. Environ. Anal. Chem. 101, 513–532 (2021)
J. Xu, Y. Chau, Y.K. Lee, Phage-based electrochemical sensors: a review. Micromachines 10, 855 (2019)
J. Xu, C. Zhao, Y. Chau, Y.K. Lee, The synergy of chemical immobilization and electrical orientation of T4 bacteriophage on a micro electrochemical sensor for low-level viable bacteria detection via Differential Pulse Voltammetry. Biosens. Bioelectron. 151, 111914 (2020)
Aaryashree, Y. Takeda, M. Kanai, A. Hatano, Y. Yoshimi, M. Kida: A “single-use” ceramic-based electrochemical sensor chip using molecularly imprinted carbon paste electrode. Sensors 20, 5847 (2020)
Q. Han, C. Wang, P. Liu, G. Zhang, L. Song, Y. Fu, Achieving synergistically enhanced dual-mode electrochemiluminescent and electrochemical drug sensors via a multi-effect porphyrin-based metal-organic framework. Sensors Actuators B Chem 330, 129388 (2021)
A. Walcarius, Electrochemical applications of silica based organic–inorganic hybrid materials. Chem. Mater. 13, 3351–3372 (2001)
Z. Navratilova, P. Kula, Clay modified electrodes: present applications and prospects. Electroanalysis 15, 837–846 (2003)
K. Buruga, H. Song, J. Shang, N. Bolan, J.T. Kalathi, K.H. Kim, A review on functional polymer-clay based nanocomposite membranes for treatment of water. J. Hazard. Mater. 379, 120584 (2019)
J.J. Fan, W. Zhou, Q. Wang, Z.J. Chu, L.Q. Yang, L. Yang, J. Sun, L. Zhao, J.M. Xu, Z.Q. Chen, Structure dependence of water vapor permeation in polymer nanocomposite membranes investigated by positron annihilation lifetime spectroscopy. J. Membr. Sci. 549, 581–587 (2018)
J.Y. Liu, Z. Chen, L. Yao, S.G. Wang, L. Huang, C.J. Dong, L.H. Niu, The 2D platelet confinement effect on the membrane hole structure probed by electrochemical impedance spectroscopy. Electrochem. Commun. 106, 106517 (2019)
R.M. Apetrei, P. Camurlu, The effect of montmorillonite functionlization on the performance of glucose biosensors based on composite montmorillonite/PAN nanofibers. Electrochim. Acta 353, 136484 (2020)
Z. Chen, S.T. Luo, L. Yao, Y. Zhang, Z.D. Lin, S.G. Wang, The inductive effect of montmorillonite/polyether sulfone membrane during the ion diffusion process. Appl. Clay Sci. 203, 106002 (2021)
Y. Xiang, G. Villemure, Electron transport in clay modified electrodes: study of electron transfer between electrochemically oxidized tris(2,2V-bipyridyl)iron cations and clay structural iron(II) sites. Can. J. Chem. 70, 1833–1837 (1992)
J. Xiao, G. Villemure, Preparation, characterization and electrochemistry of synthetic copper clays. Clays Clay Miner. 46, 195–203 (1998)
K. Yao, K. Shimazu, M. Nakata, A. Yamagishi, Clay modified electrodes as studied by the quartz crystal microbalance: adsorption of ruthenium complexes. J. Electroanal. Chem. 442, 235–242 (1998)
K. Yao, K. Shimazu, M. Nakata, A. Yamagishi, Claymodified electrodes as studied by the quartz crystal microbalance: redox processes of ruthenium and iron complexes. J. Electroanal. Chem. 443, 253–261 (1998)
L. Wu, Y. Duan, L. Chen, N. Tang, J. Li, Q. Qian, Q. Wang, G. Lv, Study on controllable assembly of stearic acid within interlayer spacing of montmorillonite and its energy storage performance. Langmuir 35, 5684–5692 (2019)
L.J. Leeson, J.E. Krueger, R.A. Nash, Concerning the structural assignment of the second and third acidity constants of the tetracycline antibiotics. Tetrahedron Lett. 4, 1155–1160 (1963)
J. Jawad, A.H. Hawari, S. Zaidi, Modeling of forward osmosis process using artificial neural networks (ANN) to predict the permeate flux. Desalination 484, 114427 (2020)
N.A. Darwish, N. Hilal, H. Al-Zoubi, A.W. Mohammad, Neural networks simulation of the filtration of sodium chloride and magnesium chloride solutions using nanofiltration membranes. Chem. Eng. Res. Des. 85, 417–430 (2007)
C. Aguzzi, P. Cerezo, G. Sandri, F. Ferrari, S. Rossi, C. Bonferoni, C. Caramella, C. Viseras, Intercalation of tetracycline into layered clay mineral for drug delivery purposes. Mater. Technol. 29, B96–B99 (2014)
A. Jha, A.C. Garade, M. Shirai, C.V. Rode, Metal cation-exchanged montmorillonite clay as catalysts for hydroxyalkylation reaction. Appl. Clay Sci. 74, 141–146 (2013)
L. Zhang, X. Lu, X. Liu, J. Zhou, H. Zhou, Hydration and mobility of interlayer ions of (Nax, Cay)-montmorillonite: a molecular dynamics study. J. Phys. Chem. C 118, 29811–29821 (2014)
L.V. Govea, H. Steinfind, Thermal stability and magnetic properties of Fe-polyoxocation intercalated montmorillonite. Chem. Mater. 9, 849–856 (1997)
X.M. Sun, Z. Ji, M.X. Xiong, W. Chen, The electrochemical sensor for the determination of tetracycline based on graphene/ L-cysterine composite film. J. Electrochem. Soc. 164, B107 (2017)
S. Negrea, L.A. Diaconu, V. Nicorescu, S. Motoc, C. Orha, F. Manea, Graphene oxide electroreduced onto boron-doped diamond and electrode corated with silver (Ag/GO/BDD) electrode for tetracycline detection in aqueous solution. Nanomaterials 11, 1566 (2021)
Z.Y. Li, C.Q. Liu, V. Sarpong, Z.Y. Gu, Multisegment nanowire/nanoparticle hybrid arrays as electrochemical biosensors for simultaneous detection of antibiotics. Biosens. Bioelectron. 126, 632–639 (2019)
C.M.F. Calixto, P. Cervini, E. Cavalheiro, Determination of tetracycline in environmental water samples at a graphite-polyurethane composite electrode. J. Braz. Chem. Soc. 23, 938–943 (2012)
L. Devkota, L.T. Nguyen, T.T. Vu, B. Piro, Electrochemical determination of tetracycline using AuNP-coated molecularly imprinted over oxidized polypyrrole sensing interface. Electrochim. Acta 270, 535–542 (2018)
D. Vega, L. Agui, A. Gonzalez-Cortes, P. Yanez-Sedeno, J.M. Pingarron, Voltammetry and amperometric detection of tetracyclines at multi-wall carbon nanotube modified electrodes. Anal. Bioanal. Chem. 389, 951–958 (2007)
Y. Zhao, Q. Wang, H. Wang, H. Zhang, X. Sun, T. Bu, Y. Liu, W. Wang, Z. Xu, L. Wang, Europium-based metal-organic framework containing characteristic metal chains: a novel turn-on fluorescence sensor for simultaneous high-performance detection and removal of tetracycline. Sensors Actuators B Chem. 334, 129610 (2021)
Funding
The work was the financial support from the Innovation Science Foundation of Wuhan Institute of Technology (Grant No. CX2020152), Natural Science Foundation of Hubei Province (Grant No. 2020CFB279) and Scientific Research Program of Hubei Province Department of Education (Grant No. B2020052).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Guo, Q., Yang, X., Chen, Z. et al. Low-cost electrochemical sensor based on montmorillonite for antibiotic tetracycline hydrochloride detection. J Mater Sci: Mater Electron 33, 427–442 (2022). https://doi.org/10.1007/s10854-021-07316-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-021-07316-0