Abstract
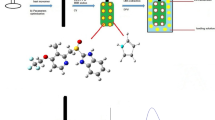
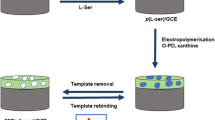
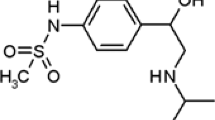
In this research project, a selective and sensitive sensor was electrochemically designed to measure metformin hydrochloride (MET) based on molecularly imprinted polymer (MIP). To prepare the sensor, the polypyrrole was electrochemically synthesized on a pencil graphite electrode (PGE), which modified with silver nanoparticles (AgNPs). Cyclic voltammetry and differential pulse voltammetry techniques were performed to fabricate the sensor and quantitative measurements, respectively. To select the functional monomers, a computational method was used. Some critical factors controlling the performance of the MIP–AgNPs–PGE were optimized using Plackett–Burman design and central composite design methods. Under optimal conditions, a calibration curve was obtained in the range 0.1–1000 µM with a limit of detection of 6.8 nM (S/N = 3, n = 3) and limit of quantitation of 22.8 nM (S/N = 10, n = 3). RSD of 3.9 and 4.1% were obtained for repeatability and reproducibility of the system, respectively. Furthermore, the modified sensor was successfully used for the determination of MET concentration in some real samples.












Similar content being viewed by others
References
F.A. Siddiqui, N. Sher, N. Shafi, S.S. Bahadur, Arab. J. Chem. (2017). https://doi.org/10.1016/j.arabjc.2013.11.035
E.M. Migdadi, A.J. Courtenay, I.A. Tekko, M.T.C. McCrudden, M.-C. Kearney, E. McAlister, H.O. McCarthy, R.F. Donnelly, JCR (2018). https://doi.org/10.1016/j.jconrel.2018.07.009
H.M. Maher, A.E. Abdelrahman, N.Z. Alzoman, H.I. Aljohar, J. Liq. Chromatogr. Relat. Technol. (2019). https://doi.org/10.1080/10826076.2019.1590208
M. Hadi, H. Poorgholi, H. Mostaanzadeh, S. Afr. J. Chem. (2016). https://doi.org/10.17159/0379-4350/2016/v69a16
R. Mirzajani, S. Karimi, Sens. Actuators B (2018). https://doi.org/10.1016/j.snb.2018.05.032
A. Sarafraz-Yazdi, N. Razavi, TrAC Trends Anal. Chem. (2015). https://doi.org/10.1016/j.trac.2015.05.004
M. Sundhoro, S.R. Agnihotra, B. Amberger, K. Augustus, N.D. Khan, A. Barnes, J. BelBruno, L. Mendecki, Food Chem. (2021). https://doi.org/10.1016/j.foodchem.2020.128648
M. Zarejousheghani, P. Rahimi, H. Borsdorf, S. Zimmermann, Y. Joseph, Sensors (2021). https://doi.org/10.3390/s21072406
A. Nezhadali, S. Senobari, M. Mojarab, Talanta (2016). https://doi.org/10.1016/j.talanta.2015.09.016
L. Fang, Y. Miao, D. Wei, Y. Zhang, Y. Zhou, Chemosphere (2021). https://doi.org/10.1016/j.chemosphere.2020.128032
G.F. Abu-Alsoud, C.S. Bottaro, Talanta (2021). https://doi.org/10.1016/j.talanta.2020.121727
J. Wang, R. Liang, W. Qin, Trends Analyt Chem. (2020). https://doi.org/10.1016/j.trac.2020.115980
I. Sadriu, S. Bouden, J. Nicolle, F.I. Podvorica, V. Bertagna, C. Berho, L. Amalric, C. Vautrin-Ul, Talanta (2020). https://doi.org/10.1016/j.talanta.2019.120222
S. Piletsky, F. Canfarotta, A. Poma, A.M. Bossi, S. Piletsky, Trends Biotechnol. (2020). https://doi.org/10.1016/j.tibtech.2019.10.002
A. Nezhadali, G. Bonakdar Ahmadi, J. Food Drug Anal. (2019). https://doi.org/10.1016/j.jfda.2018.05.002
N. Karimian, M.B. Gholivand, F. Taherkhani, J. Electroanal. Chem. (2015). https://doi.org/10.1016/j.jelechem.2014.12.024
S. Suryana, M. Yudi Rosandi, A. Nur Hasanah, Molecules (2021). https://doi.org/10.3390/molecules26071891
A. Nezhadali, M. Mojarab, Anal. Chim. Acta. (2016). https://doi.org/10.1016/j.aca.2016.04.017
A. Nezhadali, S. Sadeghzadeh, J. Electroanal. Chem. (2017). https://doi.org/10.1016/j.jelechem.2017.04.032
A. Nezhadali, Z. Rouki, M. Nezhadali, Talanta (2015). https://doi.org/10.1016/j.talanta.2015.06.082
M.C. Fonseca, C.S. Nascimento Jr., K.B. Borges, Chem. Phys. Lett. (2016). https://doi.org/10.1016/j.cplett.2015.12.061
R. Yu, H. Zhou, M. Li, Q. Song, J. Electroanal. Chem. (2019). https://doi.org/10.1016/j.jelechem.2018.10.043
A. Wong, M.V. Foguel, S. Khan, F.M. de Oliveira, C.R.T. Tarley, M.D.P.T. Sotomayor, Electrochim. Acta. (2015). https://doi.org/10.1016/j.electacta.2015.09.054
A. Nezhadali, R. Shadmehri, Sens. Actuators B (2014). https://doi.org/10.1016/j.snb.2014.04.094
M.I. Blonk, B.V.C. Ander Nagel, L.S. Smit, R.A.A. Mathod, J. Chromatogr. B (2010). https://doi.org/10.1016/j.jchromb.2010.01.037
A. Safavi, N. Maleki, F. Tajabadi, Analyst (2007). https://doi.org/10.1039/B612672C
D. Chen, J. Deng, J. Liang, J. Xie, K. Huang, C. Hu, Anal. Methods (2013). https://doi.org/10.1039/C2AY25897H
A.K. Attia, W.M. Salem, M.A. Mohamed, Acta Chim. Slov. (2015). https://doi.org/10.17344/acsi.2014.950
A.A. Ahmad, B.H. Hameed, A.L. Ahmad, J. Hazard. Mater. (2009). https://doi.org/10.1016/j.jhazmat.2009.05.021
A. Gorska, B. Paczosa-Bator, J. Wyrwa, R. Piech, Electroanalysis (2020). https://doi.org/10.1002/elan.202060012
Y. Altunkaynak, Ö. Yavuz, A. Levent, J. Indian Chem. Soc. 170, 106653 (2020)
Acknowledgements
The authors acknowledge the Payame Noor University (PNU) Research Council for financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nezhadali, A., Khalili, Z. Computer-aided study and multivariate optimization of nanomolar metformin hydrochloride analysis using molecularly imprinted polymer electrochemical sensor based on silver nanoparticles. J Mater Sci: Mater Electron 32, 27171–27183 (2021). https://doi.org/10.1007/s10854-021-07084-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-021-07084-x