Abstract
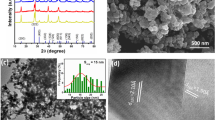
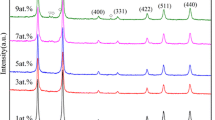
The sol–gel process is a wet chemical methodology that uses compounds in the liquid state as starting reagents. After being transformed into a gel, the reagents can be molded into films, fibers, dense materials, or porous materials. The liquid-state reagents allow different compositions to be mixed, which gives rise to applications in various fields, including catalysis, biomaterials, photonics, and coatings. This study describes a way to produce phosphors based on niobium oxide doped with lanthanides ions by a non-hydrolytic sol–gel methodology for use in solid-state light emission. More specifically, the non-hydrolytic sol–gel route was used to prepare niobium oxide doped with La3+/Eu3+ or La3+/Tb3+ at different concentrations, which was followed by treatment at 550 or 900 °C. The X-ray diffraction (XRD) patterns revealed the presence of niobium oxide in the hexagonal, orthorhombic, and monoclinic phases. The lanthanide concentrations affected the niobium oxide crystalline structure interplanar spacing. The Raman spectra corroborated the XRD observations. The luminescence spectra showed that the niobium oxide matrix doped with lanthanides emitted at different wavelengths; the blue, the green, and the red emissions were ascribed to the matrix, Tb3+, and Eu3+, respectively. The samples doped with La3+ and Tb3+ or Eu3+ presented different colors in the chromaticity diagram. Energy was transferred between the niobium oxide matrix and the lanthanides ions, and the corresponding mechanisms were proposed. In conclusion, the niobium oxide matrix is an excellent host for application as light-emitting devices.














Similar content being viewed by others
References
C. Nico, T. Monteiro, M.P.F. Graça, Prog. Mater. Sci. 80, 1 (2016)
F.T. Aquino, J.L. Ferrari, S.J.L. Ribeiro, A. Ferrier, P. Goldner, R.R. Gonçalves, Opt. Mater. 35, 387 (2013)
F.T. Aquino, R.R. Pereira, J.L. Ferrari, S.J.L. Ribeiro, A. Ferrier, P. Goldner, R.R. Gonçalves, Mater. Chem. Phys. 147, 751 (2014)
M. Palatnikov, O. Shcherbina, N. Sidorov, I. Skab, K. Bormanis, J. Phys. Opt. 13, 207 (2012)
H.P. Rooksby, E.A.D. White, Acta Crystallogr. 16, 888 (1963)
C.R. Matias, E.J. Nassar, M. Verelst, L.A. Rocha, J. Braz. Chem. Soc. 26, 2558 (2015)
F.P. Emmenegger, M.L.A. Robinson, J. Phys. Chem. Solids 29, 1673 (1968)
I. Nowak, M. Ziolek, Chem. Rev. 99, 3603 (1999)
T. Blanquart, J. Niinistö, M. Heikkilä, T. Sajavaara, K. Kukli, E. Puukilainen, C. Xu, W. Hunks, M. Ritala, M. Leskelä, Chem. Mater. 24, 975 (2012)
J. Huang, L. Zhou, Z. Liang, F. Gong, J. Han, R. Wang, J. Rare Earths 28, 356 (2010)
G.S. Freiria, E.J. Nassar, M. Verelst, L.A. Rocha, J. Lumin. 169, 844 (2016)
D. Hreniak, W. Strek, A. Speghini, M. Bettinelli, G. Boulon, Y. Guyot, Appl. Phys. Lett. 88, 161 (2006)
P. Ghigna, S. Pin, A. Speghini, M. Bettinelli, T. Tsuboi, J. Phys. Chem. Solids 71, 400 (2010)
R.R. Pereira, F.T. Aquino, A. Ferrier, P. Goldner, R.R. Gonçalves, J. Lumin. 170, 707 (2016)
K. Laganovska, I. Bite, A. Zolotarjovs, K. Smits, J. Lumin. 203, 358 (2018)
G.S. Freiria, A.L. Ribeiro, M. Verelst, E.J. Nassar, L.A. Rocha, J. Braz. Chem. Soc. 29, 594 (2018)
L. Oliveira, S. Moscardini, E. Molina, E. Nassar, M. Verelst, L. Rocha, Nanotechnology 29, 235204 (2018)
K.J. Ciuffi, O.J. de Lima, H.C. Sacco, E.J. Nassar, J. Non-Cryst. Solids 304, 126 (2002)
H. Schäfer, R. Gruehn, F. Schulte, Angew Chem. Int. Ed. Engl. 5, 40 (1966)
A.L. Viet, M.V. Reddy, R. Jose, B.V.R. Chowdari, S. Ramakrishna, J. Phys. Chem. C 114, 664 (2010)
A.L. Viet, R. Jose, M.V. Reddy, B.V.R. Chowdari, S. Ramakrishna, J. Phys. Chem. C 114, 21795 (2010)
R.A. Rani, A.S. Zoolfakar, A.P. O'Mullane, M.W. Austina, K.K. Zadeh, J. Mater. Chem. A 2, 15683 (2014)
J. Liao, R. Tan, Z. Kuang, C. Cui, Z. Wei, X. Deng, Z. Yan, Y. Feng, F. Li, C. Wang, J. Ma, Chin. Chem. Lett. 29, 1785 (2018)
K.R. Sahu, U. De, Thermochim. Acta 589, 25 (2014)
R. Brayner, F. Bozon-Verduraz, Phys. Chem. Chem. Phys. 5, 1457 (2003)
J.-M. Jehng, I.E. Wachs, Chem. Mater. 3, 100 (1991)
I.S. Cho, S.T. Bae, D.K. Yim, D.W. Kim, K.S. Hong, J. Am. Cer. Soc. 92, 506 (2009)
P.A. Loiko, O.S. Dymshits, I.P. Alekseev, A.A. Zhilin, M.Y. Tsenter, E.V. Vilejshikova, K.V. Bogdanov, X. Mateos, K.V. Yumashev, J. Lumin. 179, 64 (2016)
V. Naresh, S. Budduhu, J. Lumin. 137, 15 (2013)
W.T. Carnall, G.L. Goodman, K.E. Rajnak, E.S. Rana, J. Chem. Phys. 90, 3443 (1989)
P.F.S. Pereira, M.G. Matos, C.M.A. Ferreira, E.H. de Faria, P.S. Calefi, L.A. Rocha, K.J. Ciuffi, E.J. Nassar, J. Lumin. 146, 394 (2014)
G. Blasse, A. Bril, J. Lumin. 3, 109 (1970)
Y. Lu, X. Tang, L. Yan, K. Li, X. Liu, M. Shang, C. Li, J. Lin, J. Phys. Chem. C 117, 21972 (2013)
K. Jack, Video demystified—a handbook for the digital engineer, 4th Edn (2005)
ITU—International Telecommunication Union, Recommendation no. 709—Parameter values for the HDTV standards for production and international programme exchange. (2015)
C. de Mello Donegá, A. Meijerink, G. Blasse, J. Lumin. 62, 189 (1994)
Y. Guoa, J. Lib, J. Sun, J. Lumin. 208, 267 (2019)
Acknowledgements
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-Brasil (CAPES)-Finance Code 001. São Paulo Research Foundation (FAPESP, grant 2015/20298-0 L.A.R, 2018/02387-3 S.B.M., and 2019/02641-0 EJ.N) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Grant 302702/2018-0 L.A.R. and 302668/2017-9 E.J.N.) are also acknowledged. The authors thank Companhia Brasileira de Metalurgia e Mineração (CBMM) for donating NbCl5.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Moscardini, S.B., Sverzut, L., Massarotto, W.L. et al. Multi-color emission from lanthanide ions doped into niobium oxide. J Mater Sci: Mater Electron 31, 5241–5252 (2020). https://doi.org/10.1007/s10854-020-03084-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-020-03084-5