Abstract
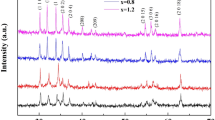
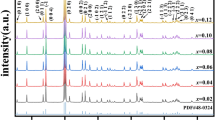
Rod-like α-MnO2 and β-MnO2 were obtained successfully by hydrothermal method under different temperatures. The as-prepared samples have been characterized by XRD, SEM, TG, TEM and HRTEM. The β-MnO2 was obtained under the condition of 150 °C and 220 °C, and the crystallinity of 220 °C would be better, the α-MnO2 was synthesize at 180 °C. The thermal stabilities of manganese oxide were affected by their crystal phases. The microwave absorption properties of rod-like α-MnO2 and β-MnO2 were studied at 2.0–18.0 GHz, and β-MnO2 show much better absorption properties than α-MnO2. The β-MnO2 prepared under the condition of 220 °C presents the optimal microwave absorption properties with the RL values of − 25.5 dB at 14.7 GHz, a thickness of 1.5 mm and an effective bandwidth of 5.0 GHz (13–18.0 GHz). These results indicate that MnO2 were dielectric loss materials, their absorption properties were influenced by the crystal phase, morphological size and crystallinity significantly. Special rod-like morphology and good crystallinity lead to superior impedance matching and moderate attenuation constant, α, and eventually enhanced microwave absorption.







Similar content being viewed by others
References
L. Wang, Z.M. Dang, Appl. Phys. Lett. 87, 282–284 (2005)
W.T. Wang, Q.L. Li, C.B. Chang, Synth. Met. 161, 44–50 (2011)
F.S. Wen, F. Zhang, Z. Liu, J. Phys. Chem. C 115, 14025–14030 (2011)
C.W. Qiang, J.C. Xu, Z.Q. Zhang, L.L. Tian, S.T. Xiao, Y. Liu, P. Xu, J. Alloys Compd. 506, 93–97 (2010)
M.G. Chen, Y. Zhu, H.M. Kou, H. Xu, J.K. Guo, Mater. Des. 32, 3013–3016 (2011)
Y. Liu, Z.Q. Zhang, S.T. Xiao, C.W. Qiang, L.L. Tian, J.C. Xu, Appl. Surf. Sci. 257, 7678–7683 (2011)
X.L. Dong, X.F. Zhang, H. Huang, F. Zuo, Appl. Phys. Lett. 92, 301 (2008)
P. Xu, X.J. Han, C. Wang, D.H. Zhou, Z.S. Lv, A.H. Wen, X.H. Wang, B. Zhang, J. Phys. Chem. B 112, 10443–10448 (2008)
C.K. Cui, Y.C. Du, T.H. Li, X.Y. Zheng, X.H. Wang, X.J. Han, P. Xu, J. Phys. Chem. B 116, 9523–9531 (2012)
G.B. Sun, B.X. Dong, M.H. Cao, B.Q. Wei, C.W. Hu, Chem. Mater. 23, 1587–1593 (2011)
R. Zhao, K. Jia, J.J. Wei, J.X. Pu, X.B. Liu, Mater. Lett. 64, 457–459 (2010)
S.B. Ni, S.M. Lin, Q.T. Pan, F. Yang, K. Huang, D.Y. He, J. Phys. D 42, 055004 (2009)
S.B. Ni, X.L. Sun, X.H. Wang, G. Zhou, F. Yang, J.M. Wang, D.Y. He, Mater. Chem. Phys. 124, 353–358 (2010)
V. Sunny, P. Kurian, P. Mohanan, P.A. Joy, M.R. Anantharaman, J. Alloys Compd. 489, 297–303 (2010)
C. Singh, S.B. Narang, I.S. Hudiara, K. Sudheendran, K.C.J. Raju, J. Magn Magn Mater. 320, 1657–1665 (2008)
P. Yuan, T. Xia, J. Zhai, J.F. Chen, Mater. Sci. Eng. B 176, 163–166 (2011)
A. Maqsood, K. Khan, J. Alloys Compd. 509, 3393–3397 (2011)
W. Xiao, D.L. Wang, X.W. Lou, J. Phys. Chem. C 114, 1430–1434 (2009)
V.B. Boppana, F. Jiao, Chem. Commun. 47, 8973–8975 (2011)
M.W. Xu, L.B. Kong, W.J. Zhou, H.L. Li, J. Phys. Chem. C 112, 19141–19147 (2007)
X.X. He, M.Y. Yang, P. Ni, Y. Li, Z.H. Liu, Colloid Surf. A 363, 64–70 (2010)
O. Ghodbane, J.L. Pascal, F. Favier, ACS Appl. Mater. Interfaces 1, 1130–1139 (2009)
A.J. Roberts, R.C.T. Slade, J. Mater. Chem. 20, 3221–3226 (2010)
H.Q. Wang, G.F. Yang, Q.Y. Li, X.X. Zhong, F.P. Wang, Z.S. Li, Y.H. Li, New J. Chem. 35, 469–475 (2011)
J.H. Zeng, Y.F. Wang, Y. Yang, J. Zhang, J. Mater. Chem. 20, 10915–10918 (2010)
J.Z. Zhao, Z.L. Tao, J. Liang, J. Chen, Cryst. Growth Des. 8, 2799–2805 (2008)
Y.P. Duan, Y. Yang, M. He, S.H. Liu, X.D. Cui, H.F. Chen, J. Phys. D 41, 1854–1862 (2008)
Y.P. Duan, Z. Jia, J. Hui, S.H. Liu, J. Solid State Chem. 184, 1165–1171 (2011)
J. Hui, Y.P. Duan, L. Zhuo, Z. Jia, S.H. Liu, Phys. B 407, 971–977 (2012)
J. Zhang, Y.P. Duan, S.Q. Li, X.G. Li, S.H. Liu, J. Solid State Chem. 183, 1490–1495 (2010)
Y.P. Duan, H. Ma, X.G. Li, S.H. Liu, Z.J. Ji, Phys. B 405, 1826–1831 (2010)
J.J. Hu, Y.P. Duan, J. Zhang, J. Hui, S.H. Liu, W.P. Li, Phys. B 406, 1950–1955 (2011)
M. Zhou, X. Zhang, J.M. Wei, S.L. Zhao, L. Wang, B.X. Feng, J. Phys. Chem. C 115, 1398–1402 (2013)
D. Yan, S. Cheng, R.F. Zhuo, J.T. Chen, J.J. Feng, H.T. Feng, H.J. Li, Z.G. Wu, J.W. Wang, P.X. Yan, Nanotechnology 20, 105706 (2009)
H.T. Guan, C. Gang, S.B. Zhang, Y.D. Wang, Mater. Chem. Phys. 124, 639–645 (2010)
B. Zhao, G. Shao, B.B. Fan, W.Y. Zhao, R. Zhang, Phys. Chem. Chem. Phys. 17, 6044–6052 (2015)
H.J. Kim, J.B. Lee, Y.M. Kim, M.H. Jung, Z. Jaglicic, P. Umek, J. Dolinsek, Nanoscale Res. Lett. 2, 81–86 (2007)
R. Giovanoli, Thermochim. Acta 234, 303–313 (1994)
Q.W. Li, G.A. Luo, J. Li, X. Xia, J. Mater. Process. Tech 137, 25–29 (2003)
X. Zhang, W.S. Yang, J.J. Yang, D.G. Evans, J. Cryst. Growth 310, 716–722 (2008)
X. Wang, Y.D. Li, Chem. Commun. 7, 764–765 (2002)
M.T. Qiao, X.F. Lei, Y. Ma, L.D. Tian, W.B. Wang, K.K. Su, Q.Y. Zhang, J. Alloys Compd. 693, 432–439 (2017)
H.L. Lv, X.H. Liang, Y. Cheng, H.Q. Zhang, D.M. Tang, B.S. Zhang, G.B. Ji, Y.W. Du, ACS Appl. Mater. Interfaces 7, 4744–4750 (2015)
B. Zhao, X.Q. Guo, W.Y. Zhao, J.S. Deng, S. Gang, B.B. Fan, Z.Y. Bai, R. Zhang, ACS Appl. Mater. Interfaces 8, 28917–28925 (2016)
X. Xia, Battery Bimon. 34, 411–414 (2004)
H.T. Guan, G. Chen, J. Zhu, Y.D. Wang, J. Alloys Compd. 507, 126–132 (2010)
B. Quan, X.H. Liang, G.B. Ji, J. Lv, S.S. Dai, G.Y. Xu, Y.W. Du, Carbon 129, 310–320 (2018)
H.T. Guan, J.B. Xie, G. Chen, Y.D. Wang, Mater. Chem. Phys. 143, 1061–1068 (2014)
B. Zhao, J.W. Liu, X.Q. Guo, W.Y. Zhao, L.Y. Liang, C. Ma, R. Zhang, Phys. Chem. Chem. Phys. 19, 9128–9136 (2017)
B. Zhao, X.Q. Guo, Y.Y. Zhou, T.T. Su, C. Ma, R. Zhang, CrystEngComm 19, 2178–2186 (2017)
J. Yang, J. Zhang, J.W. Liu, C.Y. Liang, M. Wang, M.M. Liu, R.C. Che, ACS Appl. Mater. Interfaces 5, 7146–7151 (2013)
G.S. Wang, L.Z. Nie, S.H. Yu, RSC Adv. 2, 6216–6221 (2012)
Q.M. Su, G.H. Du, J. Zhang, Y.J. Zhong, B.S. Xu, Y.H. Yang, S. Neupane, W.Z. Li, ACS Nano 8, 3620–3627 (2014)
G.Z. Wang, X.G. Peng, L. Yu, G.P. Wan, S.W. Lin, Q. Yong, J. Mater. Chem. A 3, 2734–2740 (2015)
B. Zhao, W.Y. Zhao, G. Shao, B.B. Fan, R. Zhang, ACS Appl. Mater. Interfaces 7, 12951–12960 (2015)
Y. Cheng, Z.Y. Li, Y. Li, S.S. Dai, G.B. Ji, H.Q. Zhao, J.M. Cao, Y.W. Du, Carbon 127, 643–652 (2018)
B. Quan, X.H. Liang, G.B. Ji, J.N. Ma, P.Y. Ouyang, H. Gong, G.Y. Xu, Y.W. Du, ACS Appl. Mater. Interfaces 9, 9964–9974 (2017)
B. Quan, X.H. Liang, G.B. Ji, Y. Cheng, W. Liu, J.N. Ma, Y.N. Zhang, D.R. Li, G.Y. Xu, J. Alloys Compd. 728, 1065–1075 (2017)
H.L. Lv, Z.H. Yang, P.L. Wang, G.B. Ji, J.Z. Song, L.R. Zheng, H.B. Zeng, Z.C. Xu, Adv. Mater. 30, 1706343 (2018)
Acknowledgements
This work was sponsored by the National Natural Science Foundation of China (NSFC) (Grant No. 51602287), China Postdoctoral Science Foundation Grant (Grant No. 2016M602266) and Natural Science Research Project of Henan Educational Committee (Grant No. 17A430006). The authors would like to thank for the support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Su, T., Zhao, B., Han, F. et al. The effect of hydrothermal temperature on the crystallographic phase of MnO2 and their microwave absorption properties. J Mater Sci: Mater Electron 30, 475–484 (2019). https://doi.org/10.1007/s10854-018-0312-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-018-0312-6