Abstract
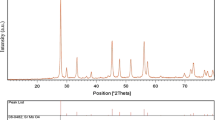
The strontium hexaferrite (SrFe12O19) nanoparticles have been successfully synthesized by co-precipitation route. The effect of various parameters such as calcination temperature and chelating agents were screened to achieve optimum condition. Different chelating agents such as amino acids (proline, alanine, aspartic acid) and surfactants (SDBS, PVP, and EDTA) were used. Compared with the amino acids, the surfactants increase the particle size and the best result was observed for alanine. The SrFe12O19 nanoparticles showed enhanced photocatalytic activity in the degradation of methyl orange under visible light irradiation (λ > 400 nm). The degradation rates of the methyl orange were measured to be as high as 95% in 220 min. The nanoparticles were also characterized by several techniques including FT-IR, XRD, SEM, and VSM. The VSM measurement showed a saturation magnetization value (Ms) of 32 emu/g. The SEM images proposed that the particles are almost spherical with an average particle size of 90 nm.






Similar content being viewed by others
References
J. Safari, F. Azizi, M. Sadeghi, New J. Chem., 39, 1905 (2015)
J. Safari, L. Javadian, RSC Adv. 5, 104973 (2015)
J. Safari, Z. Zarnegar, Ultrason. Sonochem. 21, 1132 (2014)
S. M. Hosseinpour-Mashkani, M. Ramezani, A. Sobhani-Nasab, M. Esmaeili-Zare, J. Mater. Sci. 26, 6086 (2015)
M. Ramezani, A. Sobhani-Nasab, S. M. Hosseinpour-Mashkani, J. Mater. Sci. 26, 4848 (2016)
M. Sadeghi, J. Safari, Z. Zarnegar, RSC Adv. 6, 64749 (2016)
M.N. Ashiq, M.J. Iqbal, I.H. Gul, J. Alloys Compd. 487, 341 (2009)
R.C. Pullar, Prog. Mater. Sci. 57, 1191 (2012)
Y. Bai, W. Zhang, L. Qiao, J. Zhou, J. Adv. Ceram. 1, 100 (2012)
V.V. Pankov, M. Pernet, P. Germi, P. Molard, J. Magn. Magn. Mater. 120, 69 (1993)
M. Stingaciu, M. Topole, P. McGuiness, M. Christensen, Sci. Rep. 5, 14112 (2015)
J. Wanga, Q. Chena, S. Che, J. Magn. Magn. Mater. 280, 281 (2004)
A. Thakur, R.R. Singh, P.B. Barman, Mater. Chem. Phys. 141, 562 (2013)
L. Lechevallier, J.M. Le Breton, A. Morel, P. Tenaud, J. Phys. Condens. Matter. 20, 175203 (2008)
P. Shepherd, K. Mallick, R. Green, J. Mater. Sci. 18, 527 (2007)
C. Surig, K.A. Hempel, D. Bonnenberg, IEEE Trans. Magn. 30, 4092 (1994)
R. B. Jotania, R. B. Khomane, C. C. Chauhan, S. K. Menon, B. D. Kulkarni, J. Magn. Magn. Mater. 320, 1095 (2008)
T. Gonzalez-Carreno, M.P. Morales, C.J. Serna, Mater. Lett. 43, 97 (2000)
R. Muller, J. Magn. Magn. Mater. 120, 61 (1993)
A. Ataie, I.R. Harris, C.B. Ponton, J. Mater. Sci. 30, 1429 (1995)
M. Sadeghi, J. Mater. Sci. 27, 5796 (2016)
A. Sobhani-Nasab, M. Sadeghi, J. Mater. Sci. 27, 7933–7938 (2016)
Acknowledgements
Authors are grateful to council of University of Arak for providing financial support to undertake this work.
Author information
Authors and Affiliations
Corresponding authors
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s10854-017-7115-z.
Rights and permissions
About this article
Cite this article
Mohammadi, K., Sadeghi, M. & Azimirad, R. Facile synthesis of SrFe12O19 nanoparticles and its photocatalyst application. J Mater Sci: Mater Electron 28, 10042–10047 (2017). https://doi.org/10.1007/s10854-017-6763-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-017-6763-3