Abstract
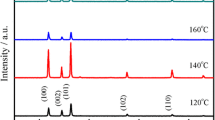
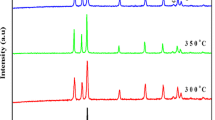
ZnO nanorods were grown by cetyl trimethylammonium bromide assisted hydrothermal technique from a single molecular precursor. The phase and structural analysis were carried out by X-ray diffraction technique and Raman spectroscopy, respectively. The phase and structural analysis has suggested that as prepared nanorods have hexagonal wurzite structure. Morphology of the nanorods was investigated by electron microscopy techniques which showed the formation of well dispersed nanorods of 100 ± 10 nm in diameter and 900 ± 100 nm in length. Optical properties were investigated by photoluminescence spectroscopy. As prepared ZnO nanorods have shown intense room temperature photoluminescence peak in the violet region at 403 nm. Absence of defect mediated green luminescence peak suggests the formation of well crystalline ZnO nanorods without any impurities or structural defects.






Similar content being viewed by others
References
V. Čechalová, A. Kalendová, J. Phys. Chem. Solids 68, 1096 (2007). doi:10.1016/j.jpcs.2006.11.017
A.E. Suliman, Y. Tang, L. Xu, Sol. Energy Mater. Sol. Cells 91, 1658 (2007). doi:10.1016/j.solmat.2007.05.014
K.M. Parida, S.S. Dash, D.P. Das, J. Colloid Interface Sci. 298, 787 (2006). doi:10.1016/j.jcis.2005.12.053
T. Gao, T.H. Wang, App. Phys. A 80, 1451 (2005). doi:10.1007/s00339-004-3075-2
Ü. Özgür, Y.I. Alivov, A. Take, M.A. Reshchikov, S. Doğan, V. Avrutin, S.J. Cho, H. Morkoc, J. Appl. Phys. 98, 041301 (2005). doi:10.1063/1.1992666
B. Liu, H.C. Zeng, J. Am. Chem. Soc. 125, 4430 (2003). doi:10.1021/ja0299452
B. Cheng, E.T. Samulski, Chem. Commun. 8, 986 (2004). doi:10.1039/b316435g
T. Sahoo, S.K. Tripathy, Y.T. Yu, H.K. Ahn, D.C. Shin, I.H. Lee, Mater. Res. Bull. 43, 2060 (2008). doi:10.1016/j.materresbull.2007.09.011
D. Vernardu, G. Kenanakis, S. Couris, E. Koudoumas, E. Kymakis, N. Katsarakis, Thin Solid Films 515, 8764 (2007). doi:10.1016/j.tsf.2007.03.108
R.S. Yadav, A.C. Pandey, S.S. Sanjay, Struct. Chem. 18, 1001 (2007). doi:10.1007/s11224-007-9251-1
X.M. Sun, X. Chen, Z.X. Deng, Y.D. Li, Mater. Chem. Phys. 78, 99 (2002). doi:10.1016/S0254-0584(02)00310-3
Y.H. Ni, X.W. Wei, X. Ma, J.M. Hong, J. Cryst. Growth 283, 48 (2005). doi:10.1016/j.jcrysgro.2005.05.048
J. Xie, P. Li, Y. Wang, Y. Wei, Phys. Status Solidi 205, 1560 (2008). doi:10.1002/pssa.200824115
D. Chu, Y.P. Zeng, D. Jiang, Mater. Lett. 60, 2783 (2006). doi:10.1016/j.matlet.2006.01.089
T.C. Damen, S.P.S. Porto, B. Tell, Phys. Rev. 142, 570 (1966). doi:10.1103/PhysRev.142.570
B.G. Wang, E.W. Shi, W.Z. Zhong, Cryst. Res. Technol. 32, 659 (1997). doi:10.1002/crat.2170320509
S.W. Kim, S. Fujita, F. Fujita, Appl. Phys. Lett. 81, 5036 (2002). doi:10.1063/1.1527690
A. Umar, Y.B. Hahn, Nanotechnology 17, 2174 (2006). doi:10.1088/0957-4484/17/9/016
H.J. Egelhaaf, D. Oelkrug, J. Cryst. Growth 161, 190 (1996). doi:10.1016/0022-0248(95)00634-6
A. Umar, B. Karunagaran, E.K. Suh, Y.B. Hahn, Nanotechnology 17, 4072 (2006). doi:10.1088/0957-4484/17/16/013
Acknowledgement
This work was supported by Post-BK21 program from Ministry of Education and Human-Resource Development.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rai, P., Tripathy, S.K., Park, NH. et al. Synthesis of violet light emitting single crystalline ZnO nanorods by using CTAB-assisted hydrothermal method. J Mater Sci: Mater Electron 20, 967–971 (2009). https://doi.org/10.1007/s10854-008-9816-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-008-9816-9