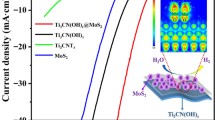
Abstract
Binder-free Mn2O3–MoS2 hybrid composites (HCs) were fabricated using a room temperature kinetic spray process under low-pressure conditions with various weight ratios of Mn2O3-to-MoS2 (1:1, 1:2, and 1:4). The effect of the composition ratio on the electrocatalytic activity of Mn2O3–MoS2 HCs toward the oxygen evolution reaction (OER) in an alkaline medium was investigated. The deposited MoS2 exhibits microrods (MRs) morphology, while pure Mn2O3 exhibits nanoflakes (NFs) morphology. The Mn2O3–MoS2 HCs exhibited NFs-decorated MR morphology. Multilayer heterostructure morphology significantly improves the interfacial synergy between various electroactive species that were verified using various spectroscopic techniques such as micro-Raman and X-ray photoemission spectra. As the MoS2 content in the Mn2O3–MoS2 HCs increased, the interfacial charge transfer kinetics associated with the reduction in the oxidation barrier potential improved. The Mn2O3–MoS2 HCs with a 1:4 ratio demonstrated the optimum combination for OER with the smallest overpotential of 290 mV @ 10 mA cm−2 and Tafel slope of 41 mV dec−1. The long-term OER stability of the fabricated electrocatalysts was verified using chronopotentiometry techniques for 50 h at 50 mA cm−2.
Graphical Abstract














Similar content being viewed by others
References
Abd-Elrahim A, Chun D-M (2021) Heterostructured Mn3O4-2D material nanosheets: one-step vacuum kinetic spray deposition and non-enzymatic H2O2 sensing. Ceram Int 47:35111
Abd-Elrahim A, Chun D-M (2023) Room-temperature coating of Mn3O4–2D material (graphene and MoS2) nanocomposites for improving oxygen evolution reaction kinetics. Mater Res Bull 166:112348
Li P, Zhao R, Chen H, Wang H, Wei P, Huang H, Liu Q, Li T, Shi X, Zhang Y (2019) Recent advances in the development of water oxidation electrocatalysts at mild pH. Small 15:1805103
Bôas NV, Junior JBS, Varanda LC, Machado SAS, Calegaro ML (2019) Bismuth and cerium doped cryptomelane-type manganese dioxide nanorods as bifunctional catalysts for rechargeable alkaline metal-air batteries. Appl Catal B Environ 258:118014
Yan S, Xue Y, Li S, Shao G, Liu Z (2019) Enhanced bifunctional catalytic activity of manganese oxide/perovskite hierarchical core-shell materials by adjusting the interface for metal-air batteries. ACS Appl Mater Interfaces 11:25870
Singu BS, Goda ES, Yoon KR (2021) Carbon Nanotube-Manganese oxide nanorods hybrid composites for high-performance supercapacitor materials. J Ind Eng Chem 97:239
Chen L, Hao C, Zhang Y, Wei Y, Dai L, Cheng J, Zhang H, Ci L (2021) Guest ions pre-intercalation strategy of manganese-oxides for supercapacitor and battery applications. J Energy Chem 60:480
Zhao Q, Song A, Ding S, Qin R, Cui Y, Li S, Pan F (2020) Preintercalation strategy in manganese oxides for electrochemical energy storage: review and prospects. Adv Mater 32:e2002450
Wu B, Xie Y, Meng Y, Qian C, Chen Y, Yuan A, Guo X, Yang H, Wan S, Lin S (2019) Construction of unique heterogeneous cobalt–manganese oxide porous microspheres for the assembly of long-cycle and high-rate lithium ion battery anodes. J Mater Chem A 7:6149
Wu P, Jin X, Qiu Y, Ye D (2021) Recent progress of thermocatalytic and photo/thermocatalytic oxidation for VOCs purification over manganese-based oxide catalysts. Environ Sci Technol 55:4268
Chutia B, Hussain N, Puzari P, Jampaiah D, Bhargava SK, Matus EV, Ismagilov IZ, Kerzhentsev M, Bharali P (2021) Unraveling the role of CeO2 in stabilization of multivalent Mn species on α-MnO2/Mn3O4/CeO2/C surface for enhanced electrocatalysis. Energy Fuels 35:10756
Wang H, Sun Y, Li Z, Li H, Guo P (2019) Electrocapacitive behavior of colloidal nanocrystal assemblies of manganese ferrite in multivalent ion electrolytes. Colloids Surf A 572:326
Chai C, Fan C, Liu J, Zhang X, Wang Y, Li R, Duan D, Wang Y (2019) Photoinduced g–C3N4–promoted Mn2+/Mn3+/Mn4+ redox cycles for activation of peroxymonosulfate. J Solid State Chem 277:466
Kuo C-H, Mosa IM, Poyraz AS, Biswas S, El-Sawy AM, Song W, Luo Z, Chen S-Y, Rusling JF, He J (2015) Robust mesoporous manganese oxide catalysts for water oxidation. ACS Catal 5:1693
Wang X, Huang G, Pan Z, Kang S, Ma S, Shen PK, Zhu J (2022) One-pot synthesis of Mn2P-Mn2O3 heterogeneous nanoparticles in a P, N-doped three-dimensional porous carbon framework as a highly efficient bifunctional electrocatalyst for overall water splitting. Chem Eng J 428:131190
Ramírez A, Hillebrand P, Stellmach D, May MM, Bogdanoff P, Fiechter S (2014) Evaluation of MnOx, Mn2O3, and Mn3O4 electrodeposited films for the oxygen evolution reaction of water. J Phys Chem C 118:14073
Wang Y, Hu T, Chen Y, Yuan H, Qiao Y (2020) Crystal facet-dependent activity of α-Mn2O3 for oxygen reduction and oxygen evolution reactions. Int J Hydrogen Energy 45:22744
Kölbach M, Fiechter S, Van De Krol R, Bogdanoff P (2017) Evaluation of electrodeposited α-Mn2O3 as a catalyst for the oxygen evolution reaction. Catal Today 290:2
Dai Y, Men Y, Wang J, Liu S, Li S, Li Y, Wang K, Li Z (2021) Tailoring the morphology and crystal facet of Mn3O4 for highly efficient catalytic combustion of ethanol. Colloids Surf A 627:127216
Hu C, Zhang L, Huang Z, Zhu W, Zhao Z-J, Gong J (2019) Facet-evolution growth of Mn3O4@CoxMn3-xO4 electrocatalysts on Ni foam towards efficient oxygen evolution reaction. J Catal 369:105
Liu P-P, Zheng Y-Q, Zhu H-L, Li T-T (2019) Mn2O3 hollow nanotube arrays on Ni foam as efficient supercapacitors and electrocatalysts for oxygen evolution reaction. ACS Appl Nano Mater 2:744
Wang S, Wang H, Huang C, Ye P, Luo X, Ning J, Zhong Y, Hu Y (2021) Trifunctional electrocatalyst of N-doped graphitic carbon nanosheets encapsulated with CoFe alloy nanocrystals: The key roles of bimetal components and high-content graphitic-N. Appl Catal B 298:120512
Yan L, Xie B, Yang C, Wang Y, Ning J, Zhong Y, Hu Y (2023) Engineering self-supported hydrophobic-aerophilic air cathode with CoS/Fe3S4 nanoparticles embedded in S, N Co-doped carbon plate arrays for long-life rechargeable Zn–Air batteries. Adv Energy Mater 13:2204245
Rebekah A, Anantharaj S, Viswanthan C, Ponpandian N (2020) Zn-substituted MnCo2O4 nanostructure anchored over rGO for boosting the electrocatalytic performance towards methanol oxidation and oxygen evolution reaction (OER). Int J Hydrogen Energy 45:14713
Kaneti YV, Guo Y, Septiani NLW, Iqbal M, Jiang X, Takei T, Yuliarto B, Alothman ZA, Golberg D, Yamauchi Y (2021) Self-templated fabrication of hierarchical hollow manganese-cobalt phosphide yolk-shell spheres for enhanced oxygen evolution reaction. Chem Eng J 405:126580
Yang B, Chang X, Ding X, Ma X, Zhang M (2022) One-dimensional Ni2P/Mn2O3 nanostructures with enhanced oxygen evolution reaction activity. J Colloid Interf Sci 623:196
Xu Q, Qin W, Tian W, Chu JF (2020) A highly efficient Co3V2O8/MoS2/carbon cloth nanocomposite bifunctional electrocatalyst for overall water splitting. Chem Sel 5:14276
Roy D, Malik MS, Chun D-M, Abd-Elrahim AG (2024) Facile one-step deposition of nanosized Ni(OH)2–MoS2 heterostructure electrodes for efficient oxygen evolution reaction. J Phys Chem Solids 186:111836
Loh JY, Yap FM, Ong W-J (2024) 2D/2D heterojunction interface: Engineering of 1T/2H MoS2 coupled with Ti3C2Tx heterostructured electrocatalysts for pH-universal hydrogen evolution. J Mater Sci Technol 179:86
Cao J, Teng F, Zhang Y, Huang J, Liu X, Zhou Q, Zhang C, Guan J (2024) Defect-rich 1T-MoS2 nanosheets towards efficient electrochemical hydrogen evolution by simultaneous phase and defect engineering. Int J Hydrogen Energy 51:529
Abd-Elrahim AG, Chun D-M (2021) Facile one-step deposition of ZnO-graphene nanosheets hybrid photoanodes for enhanced photoelectrochemical water splitting. J Alloy Compd 870:159430
Abd-Elrahim AG, Chun D-M (2020) Fabrication of efficient nanostructured Co3O4-Graphene bifunctional catalysts: Oxygen evolution, hydrogen evolution, and H2O2 sensing. Ceram Int 46:23479
Chun D-M, Ahn S-H (2011) Deposition mechanism of dry sprayed ceramic particles at room temperature using a nano-particle deposition system. Acta Mater 59:2693
Abd-Elrahim AG, Chun D-M (2021) Room-temperature deposition of ZnO-graphene nanocomposite hybrid photocatalysts for improved visible-light-driven degradation of methylene blue. Ceram Int 47:12812
Li Y, Li FM, Meng XY, Li SN, Zeng JH, Chen Y (1913) Ultrathin Co3O4 nanomeshes for the oxygen evolution reaction. Acs Catal 2018:8
Li XM, Hao XG, Abudula A, Guan GQ (2016) Nanostructured catalysts for electrochemical water splitting: current state and prospects. J Mater Chem A 4:11973
Abd-Elrahim A, Chun D-M (2022) One-step mechanical exfoliation and deposition of layered materials (graphite, MoS2, and BN) by vacuum-kinetic spray process. Vacuum 196:110732
Hong F, Yue B, Hirao N, Liu Z, Chen B (2017) Significant improvement in Mn(2)O(3) transition metal oxide electrical conductivity via high pressure. Sci Rep 7:44078
Niu X, Wei H, Tang K, Liu W, Zhao G, Yang Y (2015) Solvothermal synthesis of 1D nanostructured Mn2O3: effect of Ni2+ and Co2+ substitution on the catalytic activity of nanowires. Rsc Adv 5:66271
Abd-Elrahim A, Chun D-M (2022) Kinetically induced one-step heterostructure formation of Co3O4-Ni (OH) 2-graphene ternary nanocomposites to enhance oxygen evolution reactions. J Alloy Compd 906:164159
Abd-Elrahim A, Chun D-M (2022) Room-temperature fabrication of a heterostructure Cu2O@ CuO nanosheet electrocatalyst for non-enzymatic detection of glucose and H2O2. J Electroanal Chem 924:116874
Schlucker S (2014) Surface-enhanced Raman spectroscopy: concepts and chemical applications. Angew Chem Int Edit 53:4756
Arif AFM, Al-Athel KS, Mostaghimi J (2017). In: Hashmi MSJ (ed) Comprehensive materials finishing. Elsevier, Oxford
Frey GL, Tenne R, Matthews MJ, Dresselhaus M, Dresselhaus G (1999) Raman and resonance Raman investigation of MoS2 nanoparticles. Phys Rev B 60:2883
Najmaei S, Liu Z, Ajayan PM, Lou J (2012) Thermal effects on the characteristic Raman spectrum of molybdenum disulfide (MoS2) of varying thicknesses. Appl Phys Lett 100:013106
Chen J, Wang C (1974) Second order Raman spectrum of MoS2. Solid State Commun 14:857
Sekine T, Uchinokura K, Nakashizu T, Matsuura E, Yoshizaki R (1984) Dispersive Raman mode of layered compound 2H-MoS2 under the resonant condition. J Phys Soc Jpn 53:811
Wieting T, Verble J (1971) Infrared and Raman studies of long-wavelength optical phonons in hexagonal Mo S2. Phys Rev B 3:4286
Stacy AM, Hodul DT (1985) Raman spectra of IVB and VIB transition metal disulfides using laser energies near the absorption edges. J Phys Chem Solids 46:405
Chen Z, Lai J, Shek C (2006) Influence of grain size on the vibrational properties in Mn2O3 nanocrystals. J Non-Cryst Solids 352:3285
Luo Y, Deng Y-Q, Mao W, Yang X-J, Zhu K, Xu J, Han Y-F (2012) Probing the surface structure of α-Mn2O3 nanocrystals during CO oxidation by operando Raman spectroscopy. J Phys Chem C 116:20975
Xu J, Deng Y-Q, Luo Y, Mao W, Yang X-J, Han Y-F (2013) Operando Raman spectroscopy and kinetic study of low-temperature CO oxidation on an α-Mn2O3 nanocatalyst. J Catal 300:225
Stinner C, Prins R, Weber T (2000) Formation, structure, and HDN activity of unsupported molybdenum phosphide. J Catal 191:438
Saha D, Selvaganapathy PR, Kruse P (2020) Peroxide-induced tuning of the conductivity of nanometer-thick MoS2 films for solid-state sensors. ACS Appl Nano Mater 3:10864
Fujita S, Sato H, Motozuka S (2023) Deformation type during the ball milling process: a comparative study of the microstructures formed by ball milling, uniaxial compression, and rolling. Powder Technol 426:118598
Tuschel D (2019) Stress, strain, and Raman spectroscopy. Spectroscopy 34:10
Abd-Elrahim A, Chun D-M (2020) Facile one-step deposition of Co3O4-MoS2 nanocomposites using a vacuum kinetic spray process for non-enzymatic H2O2 sensing. Surf Interfaces 21:100748
Camacho-López MA, Escobar-Alarcón L, Picquart M, Arroyo R, Córdoba G, Haro-Poniatowski E (2011) Micro-Raman study of the m-MoO2 to α-MoO3 transformation induced by cw-laser irradiation. Opt Mater 33:480
Spevack P, Mcintyre N (1992) Thermal reduction of molybdenum trioxide. J Phys Chem 96:9029
Cheng S, Yang L, Chen D, Ji X, Jiang Z-J, Ding D, Liu M (2014) Phase evolution of an alpha MnO2-based electrode for pseudo-capacitors probed by in operando Raman spectroscopy. Nano Energy 9:161
Bernardini S, Bellatreccia F, Della Ventura G, Sodo A (2021) A reliable method for determining the oxidation state of manganese at the microscale in Mn oxides via Raman spectroscopy. Geostand Geoanal Res 45:223
Huang Y, He W, Zhang P, Lu X (2018) Nitrogen-doped MnO2 nanorods as cathodes for high-energy Zn-MnO2 batteries. Funct Mater Lett 11:1840006
Julien C, Massot M, Rangan S, Lemal M, Guyomard D (2002) Study of structural defects in γ-MnO2 by Raman spectroscopy. J Raman Spectrosc 33:223
Cross J, Martin J, Pope L, Koontz S (1990) Atomic oxygen-MoS2 chemical interactions. Surf Coat Technol 42:41
Wang H, Skeldon P, Thompson G (1997) XPS studies of MoS2 formation from ammonium tetrathiomolybdate solutions. Surf Coat Technol 91:200
Zhang J, Wang T, Liu P, Liu S, Dong R, Zhuang X, Chen M, Feng X (2016) Engineering water dissociation sites in MoS2 nanosheets for accelerated electrocatalytic hydrogen production. Energy Environ Sci 9:2789
Yang Y, Zhang K, Lin H, Li X, Chan HC, Yang L, Gao Q (2017) MoS2–Ni3S2 heteronanorods as efficient and stable bifunctional electrocatalysts for overall water splitting. ACS Catal 7:2357
Xiong X, Luo W, Hu X, Chen C, Qie L, Hou D, Huang Y (2015) Flexible membranes of MoS2/C nanofibers by electrospinning as binder-free anodes for high-performance sodium-ion batteries. Sci Rep 5:1
Mcintyre N, Zetaruk D (1977) X-ray photoelectron spectroscopic studies of iron oxides. Anal Chem 49:1521
Oku M, Hirokawa K, Ikeda S (1975) X-ray photoelectron spectroscopy of manganese—oxygen systems. J Electron Spectrosc Relat Phenom 7:465
Zhang X, Li H, Hou F, Yang Y, Dong H, Liu N, Wang Y, Cui L (2017) Synthesis of highly efficient Mn2O3 catalysts for CO oxidation derived from Mn-MIL-100. Appl Surf Sci 411:27
Li Z-Y, Akhtar MS, Bui PT, Yang O-B (2017) Predominance of two dimensional (2D) Mn2O3 nanowalls thin film for high performance electrochemical supercapacitors. Chem Eng J 330:1240
Xu J, Sun Y, Lu M, Wang L, Zhang J, Qian J, Kim EJ (2017) Fabrication of porous Mn2O3 microsheet arrays on nickel foam as high-rate electrodes for supercapacitors. J Alloy Compd 717:108
Wang F, Dai H, Deng J, Bai G, Ji K, Liu Y (2012) Manganese oxides with rod-, wire-, tube-, and flower-like morphologies: highly effective catalysts for the removal of toluene. Environ Sci Technol 46:4034
Lyle H, Singh S, Paolino M, Vinogradov I, Cuk T (2021) The electron-transfer intermediates of the oxygen evolution reaction (OER) as polarons by in situ spectroscopy. Phys Chem Chem Phys 23:24984
Koroteev V, Bulusheva L, Asanov I, Shlyakhova E, Vyalikh D, Okotrub A (2011) Charge transfer in the MoS2/carbon nanotube composite. J Phys Chem C 115:21199
Lin Y-K, Chen R-S, Chou T-C, Lee Y-H, Chen Y-F, Chen K-H, Chen L-C (2016) Thickness-dependent binding energy shift in few-layer MoS2 grown by chemical vapor deposition. ACS Appl Mater Interfaces 8:22637
Rehman J, Fan X, Laref A, Dinh VA, Zheng W (2021) Potential anodic applications of 2D MoS2 for K-ion batteries. J Alloy Compd 865:158782
Qin Y, Zhang Y, Wang J, Zhang J, Zhai Y, Wang H, Li D (2020) Heterogeneous structured Bi2S3/MoS2@ NC nanoclusters: exploring the superior rate performance in sodium/potassium ion batteries. ACS Appl Mater Interfaces 12:42902
Share K, Cohn AP, Carter RE, Pint CL (2016) Mechanism of potassium ion intercalation staging in few layered graphene from in situ Raman spectroscopy. Nanoscale 8:16435
Li F, Zou J, Cao L, Li Z, Gu S, Liu Y, Zhang J, Liu H, Lu Z (2019) In situ study of K+ electrochemical intercalating into MoS2 flakes. J Phys Chem C 123:5067
Cross AD, Morel A, Hollenkamp TF, Donne SW (2011) Chronoamperometric versus galvanostatic preparation of manganese oxides for electrochemical capacitors. J Electrochem Soc 158:A1160
Xie M, Yang L, Ji Y, Wang Z, Ren X, Liu Z, Asiri AM, Xiong X, Sun X (2017) An amorphous Co-carbonate-hydroxide nanowire array for efficient and durable oxygen evolution reaction in carbonate electrolytes. Nanoscale 9:16612
Acknowledgements
This work was supported by the 2024 Research Fund of the University of Ulsan.
Author information
Authors and Affiliations
Contributions
AGA contributed to conceptualization, data curation, methodology, formal analysis, investigation, validation, writing—original draft, and writing—review and editing. DR and MMS contributed to conceptualization, methodology, and investigation. DC contributed to conceptualization, investigation, funding acquisition, project administration, validation, writing—original draft, and writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Not applicable.
Data and code availability
Data will be available upon request.
Additional information
Handling Editor: Christopher Blanford.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
SEM images, EDS spectra of powder; EDS mapping of deposited nanostructured thin films, Raman spectra of micron powder; XPS survey spectra and high-resolution XPS scans of S 2p, Mo 3d, O1s, and Mn 2p of nanostructured films of Mn2O3, MoS2, and Mn2O3–MoS2 NCs; CV plots from to 0.1 V vs. Hg/HgO and the corresponding ECSA; XPS after OER stability test, and OER performance comparison table.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abd-Elrahim, A.G., Roy, D., Malik, M.S. et al. Low-temperature coating of Mn2O3–MoS2 micro-nano-heterostructure anode as an efficient catalyst for water splitting applications. J Mater Sci 59, 7332–7355 (2024). https://doi.org/10.1007/s10853-024-09620-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-024-09620-6