Abstract
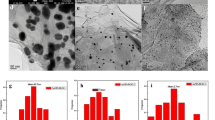
This paper presents a new, simple, accessible and environmentally friendly method to prepare gold-reduced graphene oxide composites (Au/rGO) with gold concentration of 2.5 wt%, 5 wt% and 10 wt%. The Au/rGO materials with low Au concentration present AuNPs of low dimensions and proved good catalytic activity for formic acid (FA) decomposition in aqueous solution. The most of the structural characteristics are not dependent of the gold content such as: large surface area (234–278 m2 g−1), good crystallinity of graphenes, few layers of carbon in the graphene support (5–6 layers), medium density of structural defects in graphene, the presence of remanent oxygenated groups on the graphene surface, very good dispersion of AuNPs on both sides of the graphene sheets. The AuNPs size instead is strongly dependent on the Au concentration: The smallest nanoparticles were obtained for Au2.5/rGO (3–5 nm) and the largest for Au10/rGO (10–14 nm). The position of the energy levels (conduction band and valence band) relative to the Fermi level also changes with the Au content.






Similar content being viewed by others
References
Yang Z, Ren J, Zhang Z, Chen X, Guan G, Qiu L et al (2015) Recent advancement of nanostructured carbon for energy applications. Chem Rev 115:5159–5223
Ye M, Zhang Z, Zhao Y, Qu L (2018) Graphene platforms for smart energy generation and storage. Joule 2:245–268
Das T, Sharma BK, Katiyar AK, Ahn J-H (2018) Graphene-based flexible and wearable electronics. J Semiconduct 39:011007-1–01107-19
Perreault F, Fonseca de Faria A, Elimelech M (2015) Environmental applications of graphene-based nanomaterials. Chem Soc Rev 44:5861–5896
Ambrosi A, Chua CK, Bonanni A, Pumera M (2014) Electrochemistry of graphene and related materials. Chem Rev 114:7150–7188
Zhang C, Nicolosi V (2019) Graphene and MXene-based transparent conductive electrodes and supercapacitors. Energy Storage Mater 16:102–125
Mao HY, Laurent S, Chen W, Akhavan O, Imani M, Ashkarran AA et al (2013) Graphene: promises, facts, opportunities, and challenges in nanomedicine. Chem Rev 113:3407–3424
Reina G, Gonzalez-Dominguez JM, Criado A, Vazquez E, Bianco A, Prato M (2017) Promises, facts and challenges for graphene in biomedical applications. Chem Soc Rev 46:4400–4416
Blanita G, Lazar MD (2013) Review of graphene-supported metal nanoparticles as new and efficient heterogeneous catalysts. Micro Nanosyst 5:138–146
Muhd Julkapli N, Bagheri S (2015) Graphene supported heterogeneous catalysts: An overview. Int J Hydrogen Energy 40:948–979
Yin PT, Shah S, Chhowalla M, Lee K-B (2015) Design, synthesis, and characterization of graphene-nanoparticle hybrid materials for bioapplications. Chem Rev 115:2483–2531
Xu J, Cao Z, Zhang Y, Yuan Z, Lou Z, Xu X et al (2018) A review of functionalized carbon nanotubes and graphene for heavy metal adsorption from water: preparation, application, and mechanism. Chemosphere 195:351–364
Biris AR, Ardelean S, Lazar MD, Dervishi E, Watanabe F, Gosh A et al (2012) Synthesis of few layer graphene over gold nanoclusters supported on MgO. Carbon 50:2252–2263
Zhang Y, Guo H, Weng W, Fu M-L (2017) Surface plasmon resonance, thermal, support and size effect induced photocatalytic activity enhancement of Au/reduced graphene oxide for selective oxidation of benzylic alcohols. Phys Chem Chem Phys 19:31389–31398
Yu X, Huo Y, Yang J, Chang S, Ma Y, Huang W (2013) Reduced graphene oxide supported Au nanoparticles as an efficient catalyst for aerobic oxidation of benzyl alcohol. Appl Surf Sci 280:450–455
Li X, Ma Y, Yang Z, Huang D, Xu S, Wang T et al (2017) In situ preparation of magnetic Ni–Au/graphene nanocomposites with electron-enhanced catalytic performance. J Alloys Compd 706:377–386
Sivaranjan K, Padmaraj O, Santhanalakshmi J (2018) Synthesis of highly active rGO-supported mono and bi-metallic nanocomposites as catalysts for chemoselective hydrogenation of α,β-unsaturated ketone to alcohol. New J Chem 42:1725–1735
Pocklanova R, Rathi AK, Gawande MB, Datta KKR, Ranc V, Cepe K et al (2016) Gold nanoparticle-decorated graphene oxide: synthesis and application in oxidation reactions under benign conditions. J Mol Catal 424:121–127
Pogacean F, Socaci C, Pruneanu S, Biris AR, Coros M, Magerusan L et al (2015) Graphene based nanomaterials as chemical sensors for hydrogen peroxide—a comparison study of their intrinsic peroxidase catalytic behavior. Sens Actuat B: Chem 213:474–483
Wu X, Xing Y, Pierce DT, Zhao JX (2017) One-pot synthesis of reduced graphene oxide/metal (oxide) composites. ACS Appl Mater Interfaces 9:37962–37971
Saikia I, Sonowal S, Pal M, Boruah PK, Das MR, Tamuly C (2016) Biosynthesis of gold decorated reduced graphene oxide and its biological activities. Mater Lett 178:239–242
Wang X, Meng Q, Gao L, Jin Z, Ge J, Liu C, Xing W (2018) Recent progress in hydrogen production from formic acid decomposition. Int J Hydrogen Energy 43:7055–7071
Ojeda M, Iglesia E (2009) Formic acid dehydrogenation on Au-based catalysts at near-ambient temperatures. Angew Chem Int Ed 48:4800–4803
Bulushev DA, Beloshapkin S, Ross JRH (2010) Hydrogen from formic acid decomposition over Pd and Au catalysts. Catal Today 154:7–12
Gazsi A, Bansagi T, Solymosi F (2011) Decomposition and reforming of formic acid on supported Au catalysts: production of CO-free H2. J Phys Chem C 115:15459–15466
Zacharska M, Chuvilin AL, Kriventsov VV, Beloshapkin S, Estrada M, Simakov A et al (2016) Support effect for nanosized Au catalysts in hydrogen production from formic acid decomposition. Catal Sci Technol 6:6853–6860
Bi Q-Y, Du X-L, Liu Y-M, Cao Y, He H-Y, Fan K-N (2012) Efficient subnanometric gold-catalyzed hydrogen generation via formic acid decomposition under ambient conditions. J Am Chem Soc 134:8926–8933
Bi Q-Y, Lin J-D, Liu Y-M, Huang F-Q, Cao Y (2016) Promoted hydrogen generation from formic acid with amines using Au/ZrO2 catalyst. Int J Hydrogen Energy 41:21193–21202
Liu Q, Yang X, Huang Y, Xu S, Su X, Pan X et al (2015) A Schiff base modified gold catalyst for green and efficient H2 production from formic acid. Energy Environ Sci 8:3204–3207
Sridhar M, Ferri D, van Bokhoven JA, Kröcher O (2017) Water-assisted oxygen activation during gold-catalyzed formic acid decomposition under SCR-relevant conditions. J Catal 349:197–207
Bi Q-Y, Lin J-D, Liu Y-M, He H-Y, Huang F-Q, Cao Y (2016) Gold supported on zirconia polymorphs for hydrogen generation from formic acid in base-free aqueous medium. J Power Sources 328:463–471
Hummers WS, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80:1339
He Y, Zhang N, Gong Q, Li Z, Gao J, Qiu H (2012) Metal nanoparticles supported graphene oxide 3D porous monoliths and their excellent catalytic activity. Mater Chem Phys 134:585–589
Li W-W, Kong F-Y, Wang J-Y, Chen Z-D, Fang H-L, Wang W (2015) Facile one-pot and rapid synthesis of surfactant-free Au-reduced graphene oxide nanocomposite for trace arsenic(III) detection. Electrochim Acta 157:183–190
Yin H, Tang H, Wang D, Gao Y, Tang Z (2012) Facile synthesis of surfactant free Au cluster/graphene hybrids for high performance oxygen reduction reaction. ACS Nano 6:8288–8297
Machado BF, Serp P (2012) Graphene based materials for catalysis. Catal Sci Technol 2:54–75
Lazar MD, Biris AR, Borodi G, Voica C, Watanabe F, Dervishi E et al (2013) Magnesia supported Au and Ag catalysts for the preparation of few-layer graphene–metal nanocomposites: relationship between catalyst structure and the properties of graphene composites. J Mater Sci 48:7409–7421. https://doi.org/10.1007/s10853-013-7556-6
Gregg SB, Vecchio S (2006) Thermogravimetric analysis of synthesis variation effects on CCVD generated multiwalled carbon nanotubes. J Phys Chem B 110:1179–1186
Subrahmanyam KS, Vivekchand SRC, Govindaraj A, Rao CNR (2008) A study of graphene prepared by different methods: characterization, properties and solubilization. J Mater Chem 18:1517–1523
Barman BK, Nanda KK (2013) The dual role of Zn-acid medium for one-step rapid synthesis of M@rGO (M = Au, Pt, Pd and Ag) hybrid nanostructures at room temperature. Chem Commun 49:8949–8951
Li Y, Fan X, Qi J, Ji J, Wang S, Zhang G, Zhang F (2010) Gold nanoparticles–graphene hybrids as active catalysts for Suzuki reaction. Mater Res Bull 45:1413–1418
Biris AR, Pruneanu S, Pogacean F, Lazar MD, Borodi G, Ardelean S et al (2013) Few-layer graphene sheets with embedded gold nanoparticles for electrochemical analysis of adenine. Int J Nanomed 8:1429–1438
Hu Y, Jin J, Wu P, Zhang H, Cai C (2010) Graphene–gold nanostructure composites fabricated by electrodeposition and their electrocatalytic activity toward the oxygen reduction and glucose oxidation. Electrochim Acta 56:491–500
Aruna I, Mehta BR, Malhotra LK, Shivaprasad SM (2008) Size dependence of core and valence binding energies in Pd nanoparticles: interplay of quantum confinement and coordination reduction. J Appl Phys 104(6):064308-1–064308-5
Bond G, Louis C, Thompson D (2007) Catalytic science series: catalysis by gold, vol 6. Imperial College Press, London
Zhu B, Lazar M, Trewyn BG, Angelici RJ (2008) Aerobic oxidation of amines to imines catalysed by bulk gold powder and by alumina-supported gold. J Catal 260:1–6
Singh S, Li S, Carrasquillo-Flores R, Alba-Rubio AC, Dumesic JA, Mavrikakis M (2014) Formic acid decomposition on Au catalysts: DFT, microkinetic modeling, and reaction kinetics experiments. AiChE J 60:1303–1319
Zhu QL, Tsumori N, Xu Q (2014) Sodium hydroxide-assisted growth of uniform Pd nanoparticles on nanoporous carbon MSC-30 for efficient and complete dehydrogenation of formic acid under ambient conditions. Chem Sci 5:195–199
Chen Y, Zhu QL, Tsumori N, Xu Q (2015) Immobilizing highly catalytically active noble metal nanoparticles on reduced graphene oxide: a non-noble metal sacrificial approach. J Am Chem Soc 137:106–109
Acknowledgements
This work was supported by a Grant CNCS-UEFISCDI, under Project Number PN-II-RU-TE-2014-4-1326.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict on interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Grad, O., Mihet, M., Dan, M. et al. Au/reduced graphene oxide composites: eco-friendly preparation method and catalytic applications for formic acid dehydrogenation. J Mater Sci 54, 6991–7004 (2019). https://doi.org/10.1007/s10853-019-03394-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-019-03394-y