Abstract
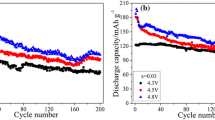
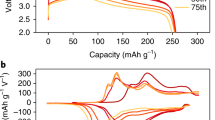
The variations in the structural and chemical features of LiNi0.6Co0.2Mn0.2O2 (LMO2: L = Li, M = Ni0.6Co0.2Mn0.2) materials were investigated via transmission electron microscopy. The LMO2 materials were used as a cathode material in a Li-battery operated at cutoff voltages of 4.15 and 4.45 V, and subjected to as many as 2200 charge–discharge cycles. Charged and discharged materials of LMO2 had Li concentrations of 0 and ~16% at the surface region, respectively, while the c lattice constant varied by up to 2.5%. The spinel phase of Li(Ni0.6Co0.2Mn0.2)2O4 (LM2O4: L = Li, M = Ni0.6Co0.2Mn0.2) appeared reversibly during charging up to 1200 cycles at room temperature; however, the LM2O4 phase was irreversibly maintained when the battery was discharged up to 2200 charge–discharge cycles at the high temperature of 45 °C. In addition, the volume of the lattice cells of the layered LMO2 differed by ~6% from the equivalent of the spinel-type LM2O4 structures. On the other hand, two types of models were suggested for the generation of short-range-ordered (SRO)-type layered structures. In particular, the pseudo-SRO models are applicable to the surface region of the samples discharged, or the inner region of the sample discharged after being cycled like 2200 times at high temperature (45 °C).










Similar content being viewed by others
References
Tarascon JM, Armand M (2001) Issues and challenges facing rechargeable lithium batteries. Nature 414:359–367
Goodenough JB, Park KS (2013) The Li-ion rechargeable battery: a perspective. J Am Chem Soc 135:1167–1176
Thomas MGSR, David WIF, Goodenough JB (1985) Synthesis and structural characterization of the normal spinel Li[Ni2]O4. Mat Res Bull 20:1137–1146
Xia L, Qiu K, Gao Y, He X, Zhou F (2015) High potential performance of cerium-doped LiNi0.5Co0.2Mn0.3O2 cathode material for Li-ion battery. J Mater Sci 50:2914–2920. doi:10.1007/s10853-015-8856-9
Li Z, Zhang D, Yang F (2009) Developments of lithium-ion batteries and challenges of LiFePO4 as one promising cathode material. J Mater Sci 44:2435–2443 doi:10.1007/s10853-009-3316-z
Mohanty D, Kalnaus S, Meisner RA, Safat AS, Li J, Payzant EA, Rhodes K, Wood DL, Daniel C (2013) Structural transformation in a Li1.2Co0.1Mn0.55Ni0.15O2 lithium-ion battery cathode during high-voltage hold. RSC Adv 3:7479–7485
Mukerjee S, Thurston TR, Jisrawi NM, Yang XQ, McBreen J, Daroux ML, Xing XK (1998) Structural evolution of Li x Mn2O4 in lithium-ion battery cells measured in situ using synchrotron X-ray diffraction techniques. J Electrochem Soc 145:466–472
Zeng YW (2008) Investigation of LiNi1/3Co1/3Mn1/3O2 cathode particles after 300 discharge/charge cycling in a lithium-ion battery by analytical TEM. J Power Sources 183:316–324
Nam KW, Bak SM, Hu E et al (2013) Combining in situ synchrotron X-ray diffraction and absorption techniques with transmission electron microscopy to study the origin of thermal instability in overcharged cathode materials for lithium-ion batteries. Adv Funct Mater 23:1047–1063
Bak SM, Hu E, Zhou Y et al (2014) Structural changes and thermal stability of charged LiNi x Mn y Co z O2 cathode materials studied by combined in situ time-resolved XRD and mass spectroscopy. ACS Appl Mater Inter 6:22594–22601
Yoon WS, Haas O, Muhammad S et al (2014) In situ soft XAS study on nickel-based layered cathode material at elevated temperatures: a novel approach to study thermal stability. Sci Rep UK 4:6827
Ceder G, Aydinol MK, Kohan AF (1997) Application of first-principles calculations to the design of rechargeable Li-batteries. Comp Mater Sci 8:161–169
Reed J, Ceder G, Van Der Ven A (2001) Layered-to-spinel phase transition in Li x MnO2. Electrochem Solid-State Lett 4:A78–A81
Zhang Y, Wang CY (2009) Cycle-life characterization of automotive lithium-ion batteries with LiNiO2 cathode. J Electrochem Soc 156:A527–A535
Abraham DP, Twesten RD, Balasubramanian M et al (2003) Microscopy and spectroscopy of lithium nickel oxide-based particles used in high power lithium-ion cells. J Electrochem Soc 150:A1450–A1456
Bloom I, Trahey L, Abouimrane A et al (2014) Effect of interface modifications on voltage fade in 0.5Li2MnO3 0.5LiNi0.375Mn0.375Co0.25O2 cathode. J Power Sources 249:509–514
Yabuuchi N, Koyama Y, Nakayama N, Ohzuku T (2005) Solid-state chemistry and electrochemistry of LiCo1/3Ni1/3Mn1/3O2 for advanced lithium-ion batteries II. Preparation and characterization. J Electrochem Soc 152:A1434–A1440
Williams DB, Carter CB (2009) Transmission electron microscopy: a textbook for materials science, 2nd edn. Springer, New York
Wu Ch, Reynolds WTJ, Murayama M (2012) A software tool for automatic analysis of selected area diffraction patterns within Digital Micrograph™. Ultramicroscopy 112:10–14
Egerton RF (2009) Electron energy-loss spectroscopy in the TEM. Rep Prog Phys 72:016502–016527
Cheralathan KK, Kang NY, Park HS, Lee YJ, Choi WC, Ko YS, Park YK (2010) Preparation of spherical LiNi0.80Co0.15Mn0.05O2 lithium-ion cathode material by continuous co-precipitation. J Power Sources 195:1486–1494
Jublot M, Texier M (2014) Sample preparation by focused ion beam micromachining for transmission electron microscopy imaging in front-view. Micron 56:63–67
Kilaas R (2009) Software package Mactempas; software available at http://www.totalresolution.com. Accessed 15 Dec 2015
Van Der Ven A, Marianetti C, Morgan D, Ceder G (2000) Phase transformations and volume changes in spinel Li x Mn2O4. Solid State Ion 135:21–32
Hoppe R, Voigt S, Glaum H, Kissel J, Muller HP, Bernet K (2002) International tables for crystallography: volume A, space group symmetry, 5th edn. Kluwer Academic Publishers, Dordrecht
Amatucci GG, Tarascon JM, Klein LC (1996) CoO2, the end member of the Li x CoO2 solid solution. J Electrochem Soc 143:1114–1123
Li W, Reimer JN, Dahn JR (1993) In situ X-ray diffraction and electrochemical studies of Li1 − x NiO2. Solid State Ion 67:123–130
Cho J, Jung HS, Park YC, Kim GB, Lim HS (2000) Electrochemical properties and thermal stability of LiaNi1 − x CO x O2 cathode materials. J Electrochem Soc 147:15–20
Choi J, Manthiram A (2005) Role of chemical and structural stabilities on the electrochemical properties of layered LiNi1/3Mn1/3Co1/3O2 cathodes. J Electrochem Soc 152:A1714–A1718
Acknowledgement
This study was financially supported by the LG Chemistry Corporation. The authors are also grateful for the use of the TEM facilities at Inha University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yun, HS., Lee, SH., Cho, NH. et al. Transmission electron microscopy studies of structural degradation behavior of LiNi0.6Co0.2Mn0.2O2 cathode materials. J Mater Sci 52, 8377–8390 (2017). https://doi.org/10.1007/s10853-017-1080-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-017-1080-z