Abstract
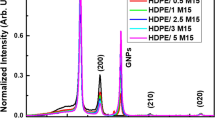
A green biodegradable poly[(butylene succinate)-co-adipate] (PBSA) synthesized by polycondensation reaction is solution-mixed with organically modified layered double hydroxide (m-LDH) to fabricate the PBSA/m-LDH nanocomposite. The crystallization behaviors, morphologies, and microstructures of PBSA and PBSA/m-LDH nanocomposite are studied by differential scanning calorimetry, wide-angle X-ray diffraction (WAXD), transmission electron microscopy (TEM), and small-angle X-ray scattering (SAXS). Both WAXD and TEM results demonstrate that most of the layered materials are exfoliated and randomly distributed in the PBSA matrix. The isothermal crystallization kinetics of neat PBA and PBSA/m-LDH nanocomposite are analyzed using the Avrami equation. With the addition of 3 wt% m-LDH into PBSA, the half-time for crystallization of PBSA/m-LDH nanocomposite decreased by 47–76 % for isothermal crystallization at 48–56 °C compared to that of neat PBSA. The value of σσ e for 3 wt% PBSA/m-LDH nanocomposites is the lowest. The microstructure of PBSA/m-LDH nanocomposites identified by SAXS shows that higher crystal layer thickness is achieved with the introduction of 3 wt% m-LDH. These results indicate that the introduction of 3 wt% m-LDH content into PBSA causes a decrease in surface free energy and thus an increase in the polymer chain mobility.







Similar content being viewed by others
References
Bandyopadhyay J, Sinha Ray S (2010) The quantitative analysis of nano-clay dispersion in polymer nanocomposites by small angle X-ray scattering combined with electron microscopy. Polymer 51:1437–1449
Nikolic MS, Djonlagic J (2001) Synthesis and characterization of biodegradable poly(butylene succinate-co-butylene adipate)s. Polym Degrad Stab 74:263–270
Tserki V, Matzinos P, Pavlidou E, Vachliotis D, Panayiotou C (2006) Biodegradable aliphatic polyesters. Part I. Properties and biodegradation of poly(butylene succinate-co-butylene adipate). Polym Degrad Stab 91:367–376
Ahn BD, Kim SH, Kim YH, Yang JS (2001) Synthesis and characterization of the biodegradable copolymers from succinic acid and adipic acid with 1,4-butanediol. J Appl Polym Sci 82:2808–2826
Han SI, Lim JS, Kim DK, Kim MN, Im SS (2008) In situ polymerized poly(butylene succinate)/silica nanocomposites: physical properties and biodegradation. Polym Degrad Stab 93:889–895
Hennous M, Derriche Z, Privas E, Navard P, Verney V, Leroux F (2013) Lignosulfonate interleaved layered double hydroxide: a novel green organoclay for bio-related polymer. Appl Clay Sci 71:42–48
Bandyopadhyay J, Al-Thabaiti SA, Ray SS, Basahel SN, Mokhtar M (2014) Unique cold-crystallization behavior and kinetics of biodegradable poly[(butylene succinate)-co adipate] nanocomposites: a high speed differential scanning calorimetry study. Macromol Mater Eng 299:939–952
Chen G, Yoon J-S (2005) Nanocomposites of poly[(butylene succinate)-co-(butylene adipate)] (PBSA) and twice-functionalized organoclay. Polym Int 54:939–945
Ojijo V, Malwela T, Sinha Ray S, Sadiku R (2012) Unique isothermal crystallization phenomenon in the ternary blends of biopolymers polylactide and poly[(butylene succinate)-co-adipate] and nano-clay. Polymer 53:505–518
Ikehara T, Kimura H, Qiu Z (2005) Penetrating spherulitic growth in poly(butylene adipate-co-butylene succinate)/poly(ethylene oxide) blends. Macromolecules 38:5104–5108
Ojijo V, Sinha Ray S, Sadiku R (2012) Effect of nanoclay loading on the thermal and mechanical properties of biodegradable polylactide/poly[(butylene succinate)-co-adipate] blend composites. ACS Appl Mater Interfaces 4:2395–2405
Ojijo V, Cele H, Sinha Ray S (2011) Morphology and properties of polymer composites based on biodegradable polylactide/poly[(butylene succinate)-co-adipate] blend and nanoclay. Macromol Mater Eng 296:865–877
Wu CS (2012) Characterization and biodegradability of polyester bioplastic-based green renewable composites from agricultural residues. Polym Degrad Stab 97:64–71
Kale G, Kijchavengkul T, Auras R, Rubino M, Selke SE, Singh SP (2007) Compostability of bioplastic packaging materials: an overview. Macromol Biosci 7:255–277
Shah AA, Hasan F, Hameed A, Ahmed S (2008) Biological degradation of plastics: a comprehensive review. Biotechnol Adv 26:246–265
Chen YA, Chen EC, Wu TM (2015) Organically modified layered zinc phenylphosphonate reinforced stereocomplex-type poly(lactic acid) nanocomposites with highly enhanced mechanical properties and degradability. J Mater Sci 50:7770–7778. doi:10.1007/s10853-015-9348-7
Chen YA, Hang YT, Wu TM (2015) Polymorphism and spherulite morphology of poly(1,4-butylene adipate)/organically-modified layered double hydroxide nanocomposites. J Appl Polym Sci. doi:10.1002/app.42526
Peng H, Han Y, Liu T, Tjiu WC, He C (2010) Morphology and thermal degradation behavior of highly exfoliated CoAl-layered double hydroxide/polycaprolactone nanocomposites prepared by simple solution intercalation. Thermochim Acta 502:1–7
Ciou CY, Li SY, Wu TM (2014) Morphology and degradation behavior of poly(3-hydroxybutyrate-co-3-hydroxyvalerate)/layered double hydroxides composites. Eur Polym J 59:136–143
Chiang MF, Chen EC, Wu TM (2012) Preparation, mechanical properties and thermal stability of poly(l-lactide)/γ-polyglutamate-modified layered double hydroxide nanocomposites. Polym Degrad Stab 97:995–1001
Lonkar SP, Singh RP (2009) Isothermal crystallization and melting behavior of polypropylene/layered double hydroxide nanocomposites. Thermochim Acta 491:63–70
Hm Chen, Chen J, Ln Shao et al (2013) Comparative study of poly (l-lactide) nanocomposites with organic montmorillonite and carbon nanotubes. J Polym Sci Pol Phys 51:183–196
Avrami M (1941) Granulation, phase change, and microstructure kinetics of phase change. III. J Chem Phys 9:177–184
Avrami M (1940) Kinetics of phase change. II transformation-time relations for random distribution of nuclei. J Chem Phys 8:212–224
Alamo RG, Mandelkern L (1991) Crystallization kinetics of random ethylene copolymers. Macromolecules 24:6480–6493
Chen YA, Wu TM (2014) Crystallization kinetics of poly(1,4-butylene adipate) with stereocomplexed poly(lactic acid) serving as a nucleation agent. Ind Eng Chem Res 53:16689–16695
Hoffman JD, Weeks JJ (1962) Melting process and the equilibrium melting temperature of polychlorotrifluoroethylene. J Res Natl Bur Stand A 66:13–28
Hoffman JD (1983) Regime III crystallization in melt-crystallized polymers: the variable cluster model of chain folding. Polymer 24:3–26
Lauritzen JI, Hoffman JD (1973) Extension of theory of growth of chain-folded polymer crystals to large undercoolings. J Appl Phys 44:4340–4352
Lauritzen JI (1973) Effect of a finite substrate length upon polymer crystal lamellar growth rate. J Appl Phys 44:4353–4359
Causin V, Marega C, Saini R, Marigo A, Ferrara A (2007) Crystallization behavior of isotactic polypropylene based nanocomposites. J Therm Anal Cal 90:849–857
Strobl G, Schneider M (1980) Direct evaluation of the electron density correlation function of partially crystalline polymers. J Polym Sci Polym Phys Ed 18:1343–1359
Barbi V, Funari SS, Gehrke R, Scharnagl N, Stribeck N (2003) SAXS and the gas transport in polyether-block-polyamide copolymer membranes. Macromolecules 36:749–758
Acknowledgement
The financial support provided by Ministry of Science and Technology through the Project MOST 104-2212-E-005-089-MY2 is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, YA., Tsai, GS., Chen, EC. et al. Crystallization behaviors and microstructures of poly(butylene succinate-co-adipate)/modified layered double hydroxide nanocomposites. J Mater Sci 51, 4021–4030 (2016). https://doi.org/10.1007/s10853-016-9720-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-016-9720-2