Abstract
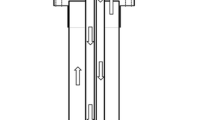
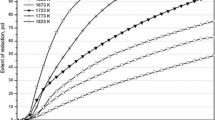
The formation of silicon by reaction between quartz and SiC has been studied in the temperature range of 1600–1900 °C in argon and hydrogen atmospheres. The reaction process was monitored by an infrared gas analyzer, and the reaction products were characterized by LECO, XRD, and SEM. Quartz–SiC reactions with SiO2/SiC molar ratio of 1:1 and 1:2 were studied in a fixed bed reactor in a graphite furnace. The production of silicon from quartz and SiC was strongly affected by temperature, SiO2/SiC molar ratio, and gas atmosphere. The yield of silicon in the reaction at 1900 °C in argon from samples with SiO2/SiC molar ratios of 1:1 and 1:2 reached 32.7 and 44.5 %, respectively. SiO2–SiC reaction at 1900 °C in hydrogen with the SiO2/SiC molar ratio of 1:2 resulted in the silicon yield of 66.7 %. Higher silicon yield in hydrogen was attributed for the involvement of hydrogen in the direct reduction of silica to SiO.









Similar content being viewed by others
References
Schei A, Tuset J, Tveit H (1998) Production of high silicon alloys. Tapir forlag, Trondheim
Schei A, Halvorsen SH (1991) A stoichiometric model of the ferrosilicon process. In: Proceedings from the Kjetil Motzfeldt symposium, Institute of Inorganic Chemistry, NTH, Trondheim, pp 41–56
Filsinger DH, Bourrie DB (1990) Silica to silicon: key carbothermic reactions and kinetics. J Am Ceram Soc 73:1726–1732
Hunt L (1990) Silicon precursors: their manufacture and properties. Handbook of semiconductor silicon technology. William Andrew Publishing, New York
Lee HC, Dhage S, Akhtar MS, Kwak DH, Lee WJ, Kim CY, Yang OB (2010) A simulation study on the direct carbothermal reduction of SiO2 for Si metal. Curr Appl Phys 10:S218–S221
Hakamada M, Fukunaka Y, Oishi T, Nishiyama T, Kusuda H (2010) Carbothermic reduction of amorphous Silica refined from diatomaceous earth. Metall Mater Trans B 41B:350–358
Dal Martello E, Tranell G, Gaal S, Raaness O, Arnberg L (2011) Combined XRD and XRF technique for the quantification of the mass balance in a Si carbothermic production experiment. ISIJ Int 9:1492–1496
Poch W, Dietzel A (1962) Bildung von Siliziumcarbid aus Siliziumoxid und Kohlenstoff. Ber. Deutsch. Ker Ges 39:413–426
Müller MB, Olsen SE, Tuset JK (1972) Heat and mass transfer in the ferrosilicon process. Scand J Metall 1:145–155
Rosenqvist T, Tuset JK (1987) Discussion of “thermodynamics of the Si–C–O system for the production of silicon carbide and metallic silicon. Metall Trans B 18:471–472
Dal Martello E, Tranell G, Gaal S, Raaness OS, Tang K, Arnberg L (2011) Study of pellets and lumps as raw materials in silicon production from quartz and silicon carbide. Metall Mater Trans B 42B:939–950
Kononov R, Ostrovski O, Ganguly S (2008) Carbothermal reduction of manganese oxide in different gas atmosphere. Metall Mater Trans B 39:662–668
Dewan MAR, Zhang G, Ostrovski O (2011) Carbothermal reduction of ilmenite concentrates and synthetic rutile in different gas atmospheres. Miner Process Extr M 120:111–117
Fruehan RJ, Carkin G (2004) Mechanism and rate of reaction of Al2O, Al, and CO vapors with carbon. Metall Mater Trans B 35:617–623
Ostrovski O, Zhang G, Kononov R, Dewan MAR, Li J (2010) Carbothermal solid state reduction of stable metal oxides. Steel Res Int 81:841–846
Li X, Zhang G, Tang K, Ostrovski O, Tronstad R (2015) Carbothermal reduction of quartz in different gas atmospheres. Metall Mater Trans B 46B:1343–1352
Wan X, Zhang G, Ostrovski O, Aral H (2013) Carbothermal reduction of silica in nitrogen and nitrogen–hydrogen mixture. In: Proceedings of the thirteenth international ferroalloys congress, Karaganda, Kazakhstan, pp 739–748
Li X, Zhang G, Tang K, Ostrovski O, Tronstad R (2015) Carbothermal reduction of quartz in methane–hydrogen–argon gas mixture. Metall Mater Trans B. doi:10.1007/s11663-015-0407-x
Chaklader ACD, Roberts AL (1961) Transformation of quartz to cristobalite. J Am Ceram Soc 44:35–41
HSC Chemistry 6.1 (2007) Chemical Reaction and Equilibrium Software with extensive thermochemical database. Outokumpu
Dewan MAR, Zhang G, Ostrovski O (2010) Carbothermal reduction of a primary ilmenite concentrate in different gas atmospheres. Metall Mater Trans B 41B:182–192
Dewan MAR, Zhang G, Ostrovski O (2009) Carbothermal reduction of titania in different gas atmospheres. Metall Mater Trans B 40:62–69
Acknowledgements
This research was supported under the Australian Research Council’s Linkage Projects Funding scheme (Project No. LP100100868). The authors would also like to thank the Mark Wainwright Analytical Centre at the University of New South Wales for LECO analysis, and the Electron Microscopy Centre (EMC) at the University of Wollongong for the electron microscopy characterization.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, X., Zhang, G., Ostrovski, O. et al. Effect of gas atmosphere on the formation of silicon by reaction of SiC and SiO2 . J Mater Sci 51, 876–884 (2016). https://doi.org/10.1007/s10853-015-9413-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-015-9413-2