Abstract
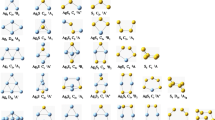
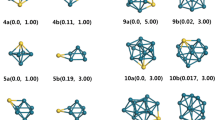
In this work, we have used density functional theory with PW91PW91 functional to analyse the structures, relative stabilities and electronic properties of small bimetallic neutral and charged Au n Al2 (n = 1–5) clusters. The results reveal that doping with two Al atoms can significantly affects the geometries of the ground-state Au n+2 (n = 1–5) clusters. The relative stabilities of the clusters were investigated on the basis of average binding energies, fragmentation energies and second-order difference of energies. The electronic properties are calculated using vertical ionization potential, vertical electron affinity values and these parameters show even–odd alternation phenomenon. The nature of bonding interaction is also investigated for the first time in Al-doped clusters using Bader’s quantum theory of atoms in molecules which indicates the presence of covalent bonding in the studied clusters. The population analysis reveals the transfer of electrons from Al to Au atoms which is responsible for the enhance stability of doped clusters.










Similar content being viewed by others
References
Eachus RS, Marchetti AP, Muenter AA (1999) The photolysis of silver halide imaging materials. Annu Rev Phys Chem 50:117–144
Scaffardi LB, Pellegri N, Ode Sanctis, Tocho JO (2005) Sizing gold nanoparticles by optical extinction spectroscopy. Nanotechnology 16:158–163
Torres MB, Fernández EM, Balbás LC (2008) Theoretical study of oxygen adsorption on pure Au +n+1 and doped MAu +n cationic gold clusters for M = Ti, Fe and n = 3–7. J Phys Chem A 112:6678–6689
Autschbach J, Hess BA, Johansson MP, Neugebauer J, Patzschke M, Pyykkö P, Reiher M, Sundholm D (2004) Properties of WAu12. Phys Chem Chem Phys 6:11–22
Lopez N, Norskov JK (2002) Catalytic CO oxidation by a gold nanoparticle: a density functional study. J Am Chem Soc 124:11262–11263
Haruta M, Kobayashi T, Samo H, Yamada N (1987) Novel gold catalysts for the oxidation of carbon monoxide at a temperature far below 0 °C. Chem Lett 2:405–408
Deka RC, Bhattacharjee D, Chakrabartty AK, Mishra BK (2014) Catalytic oxidation of NO by Au2 − dimers: a DFT study. RSC Adv 4:5399–5404
Chrétien SC, Buratto SK, Metiu H (2007) Catalysis by very small Au clusters. Curr Opin Solid State Mater Sci 11:62–75
Werner J (1999) Chemical aspects of the use of gold clusters in structural biology. J Struct Biol 127:106–112
Gaudry M, Lerme J, Cottancin E, Pellarin M, Vialle JL, Broyer M, Prevel B, Treilleux M, Melinon P (2001) Optical properties of (AuxAg1−x)n clusters embedded in alumina: evolution with size and stoichiometry. Phys Rev B 64:085407
Manzoor D, Krishnamurty S, Pal S (2014) Effect of silicon doping on the reactivity and catalytic activity of gold clusters. J Phys Chem C 118:7501–7507
Liu P, Song K, Zhang D, Liu C (2012) A comparative theoretical study of the catalytic activities of Au2 − and AuAg− dimers for CO oxidation. J Mol Model 18:1809–1818
Deka A, Deka RC (2008) Structural and electronic properties of stable Aun (n = 2–13) clusters: a density functional study. J Mol Struct (Theochem) 870:83–93
Viswanathan M, Sankaran B (2006) A DFT study of the electronic property of gold nanoclusters (Aux, x = 1–12 atoms). Bull Catal Soc India 5:26–32
Shao P, Kuang XY, Zhao YR, Li YF, Wang SJ (2012) Equilibrium geometries, stabilities, and electronic properties of the cationic AunBe+ (n = 1–8) clusters: comparison with pure gold clusters. J Mol Model 18:3553–3562
Shao N, Huang W, Mei WN, Wang LS, Wu Q, Zeng XC (2014) Structural evolution of medium-sized gold clusters Au –n (n = 36, 37, 38): appearance of bulk-like face centered cubic fragment. J Phys Chem C 118:6887–6892
Pal R, Wang LM, Huang W, Wang LS, Zeng XC (2009) Structural evolution of doped gold clusters: MAu −x (M = Si, Ge, Sn; x = 5–8). J Am Chem Soc 131:3396
Zhang M, Feng XJ, Zhao LX, He LM, Luo YH (2010) Density-functional investigation of 3d, 4d, 5d impurity doped Au6 clusters. Chin Phys B 19:043103
Lu P, Liu GH, Kuang XY (2014) Probing the structural and electronic properties of bimetallic chromium–gold clusters CrmAun (m + n ≤ 6): comparison with pure chromium and gold clusters. J Mol Model 20:2385
Tafoughalt MA, Sarmah M (2012) Density functional investigation of structural and electronic properties of small bimetallic silver–gold clusters. Phys B 407:2014–2024
Soto JR, Molinaa B, Castro JJ (2014) Nonadiabatic structure instability of planar hexagonal gold cluster cation Au7 and its spectral signature. RSC Adv 4:8157–8164
Li YF, Kuang XY, Wang SJ, Zhao YR (2010) Geometries, stabilities, and electronic properties of small anion Mg-doped gold clusters: a density functional theory study. J Phys Chem A 114:11691–11698
Wang HQ, Kuang XY, Li HF (2010) Density functional study of structural and electronic properties of bimetallic copper–gold clusters: comparison with pure and doped gold clusters. Phys Chem Chem Phys 12:5156–5165
Koyasu K, Naono Y, Akutsu M, Mitsui M, Nakajima A (2006) Photoelectron spectroscopy of binary Au cluster anions with a doped metal atom: AunM− (n = 2–7), M = Pd, Ni, Zn, Cu, and Mg. Chem Phys Lett 422:62–66
Guo JJ, Yang JX, Die D (2008) Ab initio study of small AunY2 (n = 1–4) clusters. Phys B 403:4033–4037
Bhattacharjee D, Mishra BK, Chakrabartty AK, Deka RC (2014) DFT and QTAIM studies on structure and stability of beryllium doped gold clusters. Comput Theor Chem 1034:61–72
Bouwen W, Vanhoutte F, Despa F, Bouckaert S, Neukermans S, Kuhn LT, Weidele H, Lievens P, Silverans RE (1999) Stability effects of AunXm (X = Cu, Al, Y, In) clusters. Chem Phys Lett 314:227–233
Heinebrodt M, Malinowski N, Tast F, Branz W, Billas IML, Martin TP (1999) Bonding character of bimetallic clusters AuX (X = Al, In, Cs). J Chem Phys 110:9915–9921
Wang CJ, Kuang XY, Wang HQ, Li HF, Gu JB, Liu J (2012) Density-functional investigation of the geometries, stabilities, electronic, and magnetic properties of gold cluster anions doped with aluminum: AunAl− (1 ≤ n ≤ 8). Comput Theor Chem 1002:31–36
Zhao LX, Feng XJ, Cao TT, Liang X, Luo YH (2009) Density functional study of Al doped Au clusters. Chin Phys B 18:2709–2718
Majumder C, Kandalam AK, Jena P (2006) Structure and bonding of Au5M (M = Na, Mg, Al, Si, P, and S) clusters. Phys Rev B 74:205437
Accelrys Material Studio 7.0
Rappé AK, Casewit CJ, Colwell KS, Goddard WA III, Skiff WM (1992) UFF, a full periodic table force field for molecular mechanics and molecular dynamics simulations. J Am Chem Soc 114:10024–10035
Antonello S, Arrigoni G, Dainese T, Nardi MD, Parisio G, Perotti L, René A, Venzo A, Maran F (2014) Electron transfer through 3D monolayers on Au25 clusters. ACS Nano 8:2788–2795
Huber SE, Warakulwit C, Limtrakul J, Tsukudad T, Probst M (2012) Thermal stabilization of thin gold nanowires by surfactant-coating: a molecular dynamics study. Nanoscale 4:585–590
Perdew P, Chevary JA, Vosko SH, Jackson KA, Pederson MR, Singh DJ, Fiolhais C (1992) Atoms, molecules, solids, and surfaces: applications of the generalized gradient approximation for exchange and correlation. Phys Rev B 46:6671
Hay PJ, Wadt WR (1985) Ab initio effective core potentials for molecular calculations. Potentials for the transition metal atoms Sc to Hg. J Chem Phys 82:270–283
Hay PJ, Wadt WR (1985) Ab initio effective core potentials for molecular calculations. Potentials for K to Au including the outermost core orbitals. J Chem Phys 82:299–310
Xie H, Li X, Zhao L, Qin Z, Wu X, Tang Z, Xing X (2012) Photoelectron imaging and theoretical calculations of bimetallic clusters: AgCu−, AgCu (−)2 , and Ag2Cu−. J Phys Chem A 116:10365–10370
Zhou J, Li ZH, Wang WN, Fan KN (2006) Density functional study of the interaction of carbon monoxide with small neutral and charged silver clusters. J Phys Chem A 110:7167–7172
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell K, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2010) Gaussian 09, revision B.01. Gaussian Inc, Wallingford
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, Oxford
Bader RFW (1998) A bond path: a universal indicator of bonded interactions. J Phys Chem A 102:7314–7323
Bader RFW (1991) A quantum theory of molecular structure and its applications. Chem Rev 91:893
Keith TA (2013) AIMAll (version 13.02.26). TK Gristmill Software, Overland Park (aim.tkgristmill.com)
Li YF, Mao AJ, Li Y, Kuang XY (2012) Density functional study on size-dependent structures, stabilities, electronic and magnetic properties of AunM (M = Al and Si, n = 1–9) clusters: comparison with pure gold clusters. J Mol Model 18:3061–3072
Bhattacharjee D, Mishra BK, Deka RC (2014) A DFT study on structure, stabilities and electronic properties of double magnesium doped gold clusters. RSC Adv 4:56571–56581
Hakkinen H, Yoon B, Landman U, Li X, Zhai HJ, Wang LS (2003) On the electronic and atomic structures of small Au −N (N = 4–14) clusters: a photoelectron spectroscopy and density-functional study. J Phys Chem A 107:6168–6175
Cheeseman MA, Eyler JR (1992) Ionization potentials and reactivity of coinage metal clusters. J Phys Chem 96:1082–1087
Farrugia LJ, Evans C, Tegel MJ (2006) Chemical bonds without “Chemical Bonding”? A combined experimental and theoretical charge density study on an iron trimethylenemethane complex. J Phys Chem A 110:7952–7961
Cremer D, Kraka E (1984) Chemical bonds without bonding electron density-does the difference electron-density analysis suffice for a description of the chemical bond? Angew Chem Int Ed 23:627–628
Macchi P, Sironi A (2003) Chemical bonding in transition metal carbonyl clusters: complementary analysis of theoretical and experimental electron densities. Coord Chem Rev 238–239:383–412
Acknowledgements
The work is funded by the Department of Science and Technology, New Delhi in the form of a research project (SR/NM/NS-1023/2011-G). One of the authors, D.B. is thankful to CSIR, New Delhi for providing Senior Research fellowship.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bhattacharjee, D., Mishra, B.K. & Deka, R.C. Effect of double aluminium doping on the structure, stability and electronic properties of small gold clusters. J Mater Sci 50, 4586–4599 (2015). https://doi.org/10.1007/s10853-015-9007-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-015-9007-z