Abstract
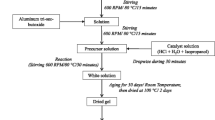
Powder granulation, Ti-doping and thermal post-treatment have beneficial effects on the electrochemical performance of 5 V LiNi0.5Mn1.5O4 (LNMO) spinel materials. It is shown that spray-drying combined with a post-doping process step is suitable to prepare Ti-doped 5 V materials with a defined and highly reproducible microstructure and chemical composition. Powder granulation via spray-drying and thermal post-treatment provides spherical LNMO granules with nano-crystalline primary particles and a 10 % higher specific discharge capacity compared to pristine LNMO materials due to the presence of the partially ordered P4 3 32 spinel phase and the reduced Mn3+ content. Powder granulation combined with a post-doping process step which utilizes titanium containing sol leads to spherical LiNi0.5Mn1.47Ti0.03O4 granules with uniform nano-crystalline primary particles and a homogenous Ti distribution. The second calcination process after spray-drying as well as the Ti-doping cause a reduced content of the Li x Ni1−x O impurity phase. This facile sol–gel processing leads to doped LiNi0.5Mn1.47Ti0.03O4 with an increased discharge capacity of 18 % compared to the original material. All the granulated materials show a good rate performance up to 10 C due to their particular microstructure.











Similar content being viewed by others
References
Liu GQ, Wen LY, Liu M (2010) J Solid State Electrochem 14:2191
Goodenough JB, Kim Y (2010) Chem Mater 22:587
Manthiram A (2011) J Phys Chem Lett 2:176
Xu B, Qian D, Wang Z, Meng YS (2012) Mater Sci Eng R Rep 73:51
Zhong Q, Bonakdarpur A, Zhang M, Gao Y, Dahn JR (1997) J Electrochem Soc 144:205
Borgel V, Markevich E, Aurbach D, Semrau G, Schmidt M (2009) J Power Sources 189:331
Yi T-F, Xie Y, Ye M-F, Jiang L-J, Zhu R-S, Zhu Y-R (2011) Ionics 17:383
Wu YP, Rahm E, Holze R (2002) Electrochim Acta 47:3491
Alcántara R, Jaraba M, Lavela P, Tirado JL (2003) Chem Mater 15:2376
Kum J-H, Myung S-T, Yoon CS, Oh I-H, Sun Y-K (2004) J Electrochem Soc 151(11):A1911
Santhanam R, Rambabu B (2010) J Power Sources 195:5442
Sun Y, Yang Y, Zhan H, Shao H, Zhou Y (2010) J Power Sources 195:4322
Hwang BJ, Wu YW, Venkateswarlu M, Cheng MY, Santhanam R (2009) J Power Sources 193:828
Li D, Ito A, Kobayakawa K, Noguchi H, Sato Y (2006) J Power Sources 161:1241
Hernán L, Morales J, Sánchez L, Santos J (1999) Solid State Ion 118:179
Noguchi T, Yamazaki I, Numata T, Shirakata MJ (2007) J Power Sources 174:359
Liu GQ, Yuan WS, Tian YW (2009) J Alloys Compd 484:567
Xiong L, Xu Y, Tao T, Goodenough JB (2012) J Power Sources 199:214
Livage J, Sanchez C, Babonneau F (1998) Molecular precursor routes to inorganic solids. In: Interrante LV, Hampden-Smith MJ (eds) Chemistry of advanced materials: an overview (Ch. 9). Wiley-VCH, New York
Schubert U, Hüsing N (2012) Synthesis of inorganic materials (Ch. 4). Wiley-VCH, Weinheim
Boyle TJ, Schwartz RW (1994) Comments Inorg Chem 16(5):243
Ceder G (2012) MRS Bull 35:693
Marom R, Amalraj SF, Leifer N, Jacob D, Aurbach D (2011) J Mater Chem 21:9938
Bach S, Pereira-Ramos JP, Baffier NJ (1998) Mater Chem 8(1):251
Le M-L-P, Strobel P, Colin CV, Pagnier T, Alloin F (2011) J Phys Chem Solids 72:124
TOPAS version 4.2. Bruker AXS, Karlsruhe
Parry KL, Shard AG, Short RD, White RG, Whittle JD, Wright A (2006) Surf Interface Anal 38:1497
Baddour-Hadjean R, Pereira-Ramos J-P (2010) Chem Rev 110:1278
Amdouni N, Zaghib K, Gendron F, Mauger A, Julien CM (2006) Ionics 12:117
Bhaskar A, Bramnik N, Senyshyn A, Fuess H, Ehrenberg HJ (2010) Electrochem Soc 157(6):A689
Biesinger MC, Lau LWM, Gerson AR, Smart RSC (2010) Appl Surf Sci 257:887
Kim J-H, Myung S-T, Yoon CS, Kang SG, Sun Y-K (2004) Chem Mater 16:906
Aklalouch M, Amarilla JM, Rojas RM, Saadoune I, Rojo JM (2010) Electrochem Commun 12:548
Shaju KM, Bruce PG (2008) Dalton Trans 40:5471
Lee H-W, Muralidharan P, Mari CM, Ruffo R, Kim DK (2011) J Power Sources 196:10712
Li D, Ito A, Kobayakawa K, Noguchi H, Sato Y (2007) Electrochim Acta 52:1919
Liu J, Manthiram A (2009) J Phys Chem C 113:15073
Xiao J, Chen X, Sushko PV, Sushko ML, Kovarik L, Feng J, Deng Z, Zheng J, Graff GL, Nie Z, Choi D, Liu J, Zhang J-G, Whittingham MS (2012) Adv Mater 24:2109
Bruce PG (ed) (1995) Chemistry of solid state materials, solid state electrochemistry (Ch. 3, 7, 8). Cambridge University Press, Cambridge
Acknowledgements
The authors would like to thank the Federal Ministry of Education and Research (BMBF) and the Ministry of Science, Research and Arts Baden-Württemberg (MWK) for financial support. The authors are grateful for the invaluable support of this work by Florian Stemme (IAM-WPT, SEM images of the spinel powders) and Dr. Thomas Bergfeldt (IAM-AWP, extensive ICP analysis to reveal the exact chemical composition of the spinel materials). Finally the authors would like to acknowledge the help provided by Dr. Stephen Doyle during the data collection at ANKA PDIFF.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Schroeder, M., Glatthaar, S., Geßwein, H. et al. Post-doping via spray-drying: a novel sol–gel process for the batch synthesis of doped LiNi0.5Mn1.5O4 spinel material. J Mater Sci 48, 3404–3414 (2013). https://doi.org/10.1007/s10853-012-7127-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-012-7127-2