Abstract
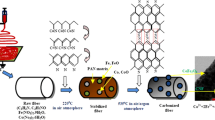
Among the reported nanostructural shapes, nanofibers have special interest due to the long axial ratio which has a distinct impact on many chemical and physical properties. In this study, synthesis of the desirable maghemite iron oxide (γ-Fe2O3) nanofibers is introduced. Calcination of electrospun mats composed of ferrous acetate and poly(vinyl alcohol) in argon atmosphere resulted in producing maghemite nanofibers. Detailed characterization affirmed that the obtained γ-Fe2O3 nanofibers are free of other iron oxides. Due to the axial ratio impact, the synthesized nanofibers which have an average diameter of ~70 nm do have magnetic properties resemble γ-Fe2O3 nanoparticles having an average diameter of ~5 nm. Accordingly, the produced nanofibers are considerable candidate for biomagnetic separation of the biomaterials. The prepared γ-Fe2O3 nanofibers can be easily handled as they were obtained in the form of strong mats. Electrical properties study indicated that the introduced nanofibers behave as a semiconducting material. Moreover, the synthesized γ-Fe2O3 nanofibers have band gap energy of ~4.2 eV. Based on the simplicity, effectiveness, high-yield, and low-cost features of the utilized preparation process and the studied physiochemical properties of the obtained product, the synthesized γ-Fe2O3 nanofibers might have considerable application fields.











Similar content being viewed by others
References
Babes L, Denizot B, Tanguy G, Le Jeune JJ, Jallet P (1999) J Colloid Interface Sci 212:474
Hu J, Ouyang MY, Lieber CM (1999) Nature 399:48
Xu DS, Guo GL, Gui LL, Tang YQ, Shi EJ, Jin EX (1999) Appl Phys Lett 75:481
Ovidiu C, Ioan DVT, Alexandru S (2012) J Mater Sci 47:2322. doi:10.1007/s10853-011-6047-x
Liang L, Liu J, Exarhos GJ, Lin Y (2002) Angew Chem Int Ed Engl 41:3665
Martin CR (1994) Science 266:1961
Wei Z, Zhang Z, Wan M (2002) Langmuir 18:917
Ramya C, Lifeng Z, Jane YH, Nyle EH, Yan Z et al (2009) J Mater Sci 44:1198. doi:10.1007/s10853-008-3201-1
Zhang X, Goux WJ, Manohar SK (2004) J Am Chem Soc 126:4502
Zhang XY, Manohar SK (2004) J Am Chem Soc 126:2714
Li W, Wang HL (2004) J Am Chem Soc 126:2278
Tejada J, Zhang XX, Balcells LI (1993) J Appl Phys 73:6709
Raphaeël J, Daniel G (2002) J Alloy Compd 333:302
Vollath D, Szabo DV, Taylor RD, Willis JO (1997) J Mater Res 12:2175
Lee SJ, Jeong JR, Shin SC, Kim JC, Chang YH, Chang YM, Kim JD (2004) J Magn Magn Mater 282:3432
Oscar BM, Lena M, Alexandra N, Sabino VV (2008) Chem Mater 20:591
Taeghwan H, Su SL, Jongnam P, Yunhee C, Hyon BN (2001) J Am Chem Soc 123:12798
Zemin W, Yong L, Xiaoyan Z (2006) Powder Technol 161:65
Rong CB, Zhang Y, Poudyal N, Szlufarska I, Hebert RJ, Kramer MJ, Liu JP (2011) J Mater Sci 46:6065. doi:10.1007/s10853-011-5568-7
Park J, Kang E, Son SU, Park HM, Lee MK, Kim J, Kim KW, Noh HJ, Park JH, Bae CJ, Park J, Hyeon T (2005) Adv Mater 17:429 (references therein)
Fraune M, Rüdiger U, Güntherodt G (2000) Appl Phys Lett 77:3815
Dennis CL, Borges RB, Buda LD, Ebels U, Gregg JF, Hehn M, Jouguelet E, Ounadjela K, Petej I, Prejbeanu IL, Thorntorn MJ (2002) J Phys Condens Matter 14:R1175
Henry Y, Ounadjela K, Piraux L, Dubois S, George JM, Duvail JL (2001) Eur Phys J B 20:35
Thurn-Albrecht T et al (2000) Science 290:2126
Zhou J, Cui YF, Liu HS, Wang W, Peng K, Xiao YD (2011) J Mater Sci 46:7567. doi:10.1007/s10853-011-5731-1
Whitney TW, Searson PC, Jiang JS, Chien CL (1993) Science 261:1316
Allwood DA, Xiong G, Cooke MD, Faulkner CC, Atkinson D, Vernier N, Cowburn RP (2002) Science 296:2003
Bentley AK, Ellis AB, Lisensky GC, Crone WC (2005) Nanotechnology 16:2193
Graeser M, Bognitzki M, Massa W, Pietzonka C, Greiner A, Wendorff JH (2007) Adv Mater 19:4244
Wu H, Zhang R, Liu X, Lin D, Pan W (2007) Chem Mater 19:3506
Barakat NAM, Woo KD, Kanjwal MA, Kyung EC, Khil MS, Kim HY (2008) Langmuir 24:11982
Barakat NAM, Hamza AM, Al-Deyabd SS, Qurashie A, Kim HY (2012) Mater Sci Eng B 177:34
Barakat NAM, Kim B, Kim HY (2009) J Phys Chem C 113:531
Barakat NAM, Kim B, Park SJ, Jo Y, Jung MH, Kim HY (2009) J Mater Chem 19:7371
Formhals A (1940) US Patent, 2,187, 306
Barakat NAM, Khil MS, Sheikh FA, Kim HY (2008) J Phys Chem C 112:12225
Cornell RM, Schertman U (2003) The iron oxides: structure, properties, reactions, occurrences, uses, 2nd edn. Wiley-VCH Gmbh & Co. KGaA, Weinheim, 141 pp
Poling GW (1969) J Electrochem Soc 116:958
Lili W, Gao L (2010) J Colloid Interface Sci 349:519
Jewur SS, Kuriacose JC (1977) Thermochem Acta 19:195
Coey MD (1971) Phys Rev Lett 27:1140
Fiorani D, Testa AM, Lucari F, D’Orazio F, Romero H (2002) Physica B 320:122
Dutta P, Manivannan A, Seehra MS, Shah N, Huffman GP (2004) Phys Rev B 70:174428-1-174428-7
Fernando GS, Jéssica AM, José CP, Geiza EO, Cezar MR, Luis MTR (2010) J Mater Sci 45:5012. doi:10.1007/s10853-010-4321-y
Franzreb M, Siemann-Herzberg M, Hobley TJ, Thomas ORT (2006) Appl Microbiol Biotechnol 70:505
Barakat NAM, Abadir MF, Nam KT, Hamza A, Al-Deyab S, Beak W, Kim HY (2011) J Mater Chem 21:10957
Hu XJ, Jiang QZ, Liao XZ, Shangguan WF, Ma ZF (2011) J New Mater Electrochem Syst 4:247
Acknowledgements
We thank Mr. T. S. Bae and Mr. J. C. Lim, KBSI, Jeonju branch, and Mr. Jong-Gyun Kang, Centre for University Research Facility, for taking high-quality FESEM and TEM images, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barakat, N.A.M. Synthesis and characterization of maghemite iron oxide (γ-Fe2O3) nanofibers: novel semiconductor with magnetic feature. J Mater Sci 47, 6237–6245 (2012). https://doi.org/10.1007/s10853-012-6543-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-012-6543-7