Abstract
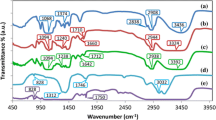
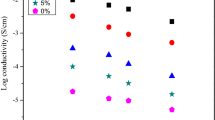
The degree of crystallinity in the matrix formed by cellulose acetate (CA) and lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) were reduced by embedding with deep eutectic solvent (DES) which is a type of ionic mixture synthesized from choline chloride and urea in a specific ratio. The fabrications of thin films were done by solution casting technique. The sample with composition of CA:LiTFSI:DES (28 wt%:12 wt%:60 wt%) appears as the highest conducting sample with the calculated value of 2.61 × 10−3 S cm−1 at ambient temperature. This high conducting sample possesses low relative viscosity so as to have high ion fluidity of 0.19. SEM micrographs were used to study the structural alternation that took place in the presence of DES at different ratio in the polymer electrolytes matrix. The dielectric loss tangent plot reveals the low relaxation time for high conducting sample which has immense movements of lithium conducting ion (Li+). The enhancement in the ionic conductivity of the DES plasticized samples with temperature obeys Arrhenius rule.





Similar content being viewed by others
References
Rajendran S, Sivakumar M, Subadevi R (2004) Mater Lett 58:641. doi:10.1016/S0167-577X(03)00585-8
Siva Kumar J, Subrahmanyam AR, Jaipal Reddy M, Subba Rao UV (2006) Mater Lett 60:3346. doi:10.1016/j.matlet.2006.03.015
Shriver DF, Papke BL, Ratner MA, Dupon R, Wong T, Brodwin M (1981) Solid State Ion 5:83. doi:10.1016/0167-2738(81)90199-5
Armand MB (1986) Annu Rev Mater Sci 16:245. doi:10.1146/annurev.ms.16.080186.001333
Ratner MA, Shriver DF (1988) Chem Rev 88:109. doi:10.1021/cr00083a006
Ramesh S, Winie T, Arof AK (2010) J Mater Sci 45:1283. doi:10.1007/s10853-009-4079-2
Reddy MJ, Sreekanth T, Chandrashekar M, Subba Rao UV (2000) J Mater Sci 35:2841. doi:10.1023/A:1004707521250
Vieira DF, Avellaneda CO, Pawlicka A (2007) Electrochim Acta 53:1404. doi:10.1016/j.electacta.2007.04.034
Xu YX, Miladinov V, Hanna MA (2004) Cereal Chem 81:735
Ning W, Xingxiang Z, Haihui L, Benqiao H (2009) Carbohyd Polym 76:482. doi:10.1016/j.carbpol.2008.11.005
Selvakumar M, Krishna Bhat D (2008) J Appl Polym Sci 110:594. doi:10.1002/app.28671
Subramaniam CK, Ramya CS, Ramya K (2008) J Appl Electrochem 41:197. doi:10.1007/s10800-010-0224-5
Ramesh S, Lu S-C (2008) J Power Sources 185:1439. doi:10.1016/j.jpowsour.2008.07.055
Webber AJ (1991) J Electrochem Soc 138:2586. doi:10.1149/1.2087287
Abbott AP, Capper G, Davies DL, Rasheed RK, Tambyrajah V (2003) Chem Commun 39:70. doi:10.1039/B210714G
Jhong H-R, Wong DS-H, Wan C-C, Wang Y-Y, Wei T-C (2009) Electrochem Commun 11:209. doi:10.1016/j.elecom.2008.11.001
Zhang J, Wu T, Chen S, Feng P, Bu X (2009) Angew Chem Int Ed Engl 48:3486. doi:10.1002/anie.200900134
Uma T, Mahalingam T, Stimming U (2003) Mater Chem Phys 82:478. doi:10.1016/S0254-0584(03)00277-3
Jana S, Zhong W-H (2008) J Mater Sci 43:4607. doi:10.1007/s10853-008-2677-z
Ahmad A, Rahman MYA, Low SP, Hamzah H (2011) ISRN Mater Sci 2011:1. doi:10.5402/2011/401280
Wu C, Wu F, Bai Y, Feng T, Pan C, Ye L, Feng Z (2009) J Chil Chem Soc 54:299. doi:10.4067/S0717-97072009000300020
Yahya MZA, Ali AMM, Mohammat MF, Hanafiah MAKM, Mustaffa M, Ibrahim SC, Darus ZM, Harun MK (2006) J Appl Sci 6:1287. doi:10.3923/jas.2006.1287.1291
Winie T, Ramesh S, Arof AK (2009) Phys B 404:4308. doi:10.1016/j.physb.2009.08.004
Anantha PS, Hariharan K (2005) Solid State Ion 176:155. doi:10.1016/j.ssi.2004.07.006
Sharma JP, Sekhon SS (2007) Solid State Ion 178:439. doi:10.1016/j.ssi.2007.01.017
Paradhan DK, Choundhary RNP, Samantaray BK (2008) Int J Electrochem Sci 3:597
Singh PK, Kim K-W, Rhee H-W (2008) Electrochem Commun 10:1769. doi:10.1016/j.elecom.2008.09.016
Muralidharan P, Venkateswarlu M, Satyanarayana N (2004) Solid State Ion 166:27. doi:10.1016/j.ssi.2003.10.011
Azizi Samir MAS, Alloin F, Sanchez J-Y, Gorecki W, Dufresne A (2004) J Phys Chem B 108:10845. doi:10.1021/jp0494483
Ramesh S, Yahaya AH, Arof AK (2002) Solid State Ion 152–153:291. doi:10.1016/S0167-2738(02)00311-9
Yahya MZA, Arof AK (2003) Euro Polym J 39:897. doi:10.1016/S0014-3057(02)00355-5
Ramesh S, Liew C–W, Ezra M, Durairaj R (2010) Thermochim Acta 511:140. doi:10.1016/j.tca.2010.08.005
Kumar GG, Kim P, Kim AR, Nahm KS, Elizabeth RN (2009) Mater Chem Phys 115:40. doi:10.1016/j.matchemphys.2008.11.023
Acknowledgement
This work was supported by the Fundamental Research Grant Scheme (FRGS) from Ministry of Higher Education, Malaysia (FP009/2010B) and Universiti Malaya Research Grant (UMRG: RG140-11AFR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ramesh, S., Shanti, R. & Morris, E. Discussion on the influence of DES content in CA-based polymer electrolytes. J Mater Sci 47, 1787–1793 (2012). https://doi.org/10.1007/s10853-011-5964-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-011-5964-z