Abstract
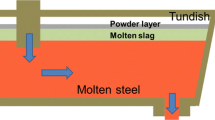
This study concerns the chemical reactions involved and the phases formed during penetration of slags of variable composition into porous plaster structure of tundish. Tundish plaster is mainly composed of MgO with minor amounts of SiO2 and impurities, with a grain size of less than 1 mm, inorganic or organic fibers in order to decrease density and provide porosity for insulation, plasticizers and stiffening agents and some other additions. Tundish slag analysis for different grades of steel (7176D and 1191D, according to DIN standard) at different sequences, indicated a very variable composition of CaO (11–42%), SiO2 (28–46%), Al2O3 (6–12%), MgO (11–20%), MnO (0–13%) and some minor variations of Fe, FeO and TiO2. Experimental work indicated that slag when penetrated into pores of plaster, develop the phases of Monticellite (CaO · MgO · SiO2) and Merwinite (CaO1.5 · MgO0.5 · SiO2) around MgO particles and decrease the liquidus temperature from 2,800 °C to about 1,500 °C and provide dissolution of MgO grains in steel making process. Calculation based on two kinetic equations developed for diffusion controlled dissolution, indicated that the dissolution of MgO in tundish plaster is not a diffusion controlled process and is affected by turbulent flow parameters. Phase diagram of CaO–SiO2–MgO indicates that decreasing SiO2 to below 20% and increasing CaO content to as high as possible, increases the liquidus temperature to above 2,000 °C. Sources of SiO2 in the process are the rice husk addition, which is used as an insulating material on top of the melt, and the slag flux addition. These sources should be reduced to as low a level as possible. This fact does not affect the fluidity of slag which is required for inclusion removal. Fluidity of slag comes from low melting point eutectics in CaO–Fe2O3 and CaO–FeO (about 1,200 °C) due to iron oxide on top of the steel melt.







Similar content being viewed by others
References
Kataoka SI (1993) Refractories of steel making in Japan, Unitcer 93 conf proc pp 1–27
Connor G (2002) Chemically bonded tundish lining system and associated near tundish refractories, Foseco Steel International report
Stendera JW (2002) Refract Appl News 7(6):26
Mazumdar D, Guthrie RIL (1991) ISIJ Int 39:524
Leasky LA, Mchale AE (1989) Ceram Trans 4:453
Matsui T, Hiragushi K, Ikemoto T, Sawano K (2002) Taikabutsu 22(4):302
Dody JA (1997) Lightweight sprayable tundish lining composition, US patent 5 602 063, 11 Feb. 1997
Cheng JA (1991) Lightweight tundish refractory composition, US patent 5 073 525, 17 Dec. 1991
Barker BJ (2001) Basic tundish flux composition for steelmaking, US patent 6 179 895 B1, 30 Jan. 2001
Levine EM, Mc Murdie HF, Hall FP (1956) Phase diagram for ceramists. The American Ceramic Society, Inc., p 115
Liu J, Guo M, Jones PT, Verhaeghe F, Blanpain B, Wollants P (2006) J Eur Ceram Soc
Monshi A (1998) Inter Ceram 47(3):155
Alexander LE, Klug HP (1948) Anal Chem 20:886
Monshi A, Messer PF (1989) Proc. 42 of British Ceramic Society, pp 201–212
Monshi A, Messer PF (1991) J Mater Sci 26:3623
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moshtaghioun, B.M., Monshi, A. Hot corrosion mechanism of tundish plaster with steel slags in continuous casting. J Mater Sci 42, 6720–6728 (2007). https://doi.org/10.1007/s10853-006-1482-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-006-1482-9