Abstract
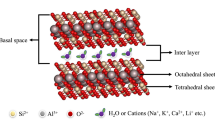
The preparations of hybrids based on montmorillonite as inorganic receiver and 8-hydroxyquinoline (8HQ) as organic host have been previously studied via solid–solid reaction at room temperature without studying the effect of physic-chemical parameters on the adsorption processes. In our present work, Na- and Zn-hybrids of 8HQ were elaborated by the extraction of 8HQ by Na(I)- and Zn(II)-montmorillonites from aqueous solution, at different pH and at different initial concentration of 8HQ. The modeling of adsorption isotherms showed that the adsorption of 8HQ follows the Langmuir model at pH 2 and the Freundlich model at pH 6 and 10. The characterization of hybrids showed firstly the swelling properties of montmorillonites and secondly the intercalation of 8HQ in the interlayer space of Na-montmorillonites (Na-Mont) and Zn-montmorillonites (Zn-Mont). The inclusion of 8HQ molecules in the inorganic receiver (clay) is assigned to its complexation with Na+ and Zn2+. The maximum quantities Qads of adsorption of 8HQ occur at pH 2 with Na-Mont and at pH 6 with Zn-Mont. The double Qads of 8HQ adsorbed by Zn-Mont, compared to that of Na-Mont, confirms the formation of complexes [Zn(8HQ)2]2+ in the interlayer spaces of Zn-Mont. The thermal analysis demonstrated the stability of both Na- and Zn-hybrids of 8HQ than the free complexes [Na(8HQ)]+ and [Zn(8HQ)2]2+. The luminescence properties of resulting hybrids showed the high emission of the hybrid 8HQ/Zn-Mont at pH 6, almost 100 times more intense than 8HQ/Na-Mont.







Similar content being viewed by others
References
Prachayasittikul, V., Prachayasittikul, S., Ruchirawat, S., Prachayasittikul, V.: 8-Hydroxyquinolines: a review of their metal chelating properties and medicinal applications. Drug Design Dev. Ther. 7, 1157–1178 (2013)
Khaorapapong, N., Ogawa, M.: Solid-state intercalation of 8-hydroxyquinoline into Li(I)-, Zn(II)- and Mn(II)-montmorillonites. Appl. Clay. Sci. 35, 31–38 (2007)
Khaorapapong, N., Ogawa, M.: Formation of mono(8-hydroxyquinoline) lithium(I) complex in smectites by solid–solid reactions. J. Phys. Chem. Solids 71, 1644–1650 (2010)
Erdem, B., Özcan, A., Özcan, A., Safa: Adsorption and solid phase extraction of 8-hydroxyquinoline from aqueous solutions by using natural bentonite. Appl. Surf. Sci. 256, 5422–5427 (2010)
Ogawa, M., Nagafusa, Y., Kuroda, K., Kato, C.: Solid-state intercalation of acrylamide into smectites and Na-taeniolite. Appl. Clay. Sci. 7(4), 291–302 (1992)
Yoshimoto, S., Ohashi, F., Kameyama, T.: X-ray diffraction studies of intercalation compounds prepared from aniline salts and montmorillonite by a mechanochemical processing. Solid State Commun 136, 251–256 (2005)
Khaorapapong, N.: Ogawa, M: In situ formation of bis(8-hydroxyquinoline) zinc(II) complex in the interlayer spaces of smectites by solid–solid reactions. J. Phys. Chem. Solids 69, 941–948 (2008)
Cölle, M., Gmeiner, J., Milius, W., Hillebrecht, H., Brütting, W.: Preparation and characterization of blue-luminescent tris(8-hydroxyquinoline)-aluminum (Alq3). Adv. Funct. Mater. 13(2), 108–112 (2003)
Huo, Y., Lu, J., Hu, S., Zhang, L., Zhao, F., Huang, H., Huang, B., Zhang, L.: Photoluminescence properties of new Zn(II) complexes with 8-hydroxyquinoline ligands: dependence on volume and electronic effect of substituents. J. Mol. Struct. 1083, 144–151 (2015)
Kim, Y., Lee, J.-G.: Organometallic luminescent materials and organic electroluminescent device containing same. US Patent 6579633 B2 (2003)
Tsuboi, T., Nakai, Y., Torii, Y.: Photoluminescence of bis(8-hydroxyquinoline) zinc (Znq2) and magnesium (Mgq2). Cent. Eur. J. Phys. 10(2), 524–528 (2012)
Cölle, M., Brütting, W.: Thermal, structural and photophysical properties of the organic semiconductor Alq3. Phys. Status Solidi A 201(6), 1095–1115 (2004)
Sapochak, L.S., Benincasa, F.E., Schofield, R.S., Baker, J.L., Riccio, K.K.C., Fogarty, D., Kohlmann, H., Ferris, K.F., Burrows, P.E.: Electroluminescent zinc(II) bis(8-hydroxyquinoline): structural effects on electronic states and device performance. J. Am. Chem. Soc. 124(21), 6119–6125 (2002)
Bekri-Abbes, I., Srasra, E.: Synthesis of an organic–inorganic hybrid material by solid state intercalation of 2-mercaptopyridine into Na-, Al(III)- and Co(II)-montmorillonite. Bull. Mater. Sci. 29(3), 251–259 (2006)
Bergaya, F., Lagaly, G.: Chap. 1 general introduction: clays, clay minerals, and clay science. Dev. Clay Sci. 1, 1–18 (2006)
Filip, E.M., Humelnicu, I.V., Ghirvu, C.I.: Some Aspects of 8-hydroxyquinoline in solvents. Acta Chem. IASI 17, 85–96 (2009)
Ferreiro, E.A., Bussetti, S.G.D.E., Helmy, A.K.: Sorption of 8-hydroxyquinoline by some clays and oxides. Clays Clay Miner. 36(1), 61–67 (1988)
Langmuir, I.: The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 40(9), 1361–1403 (1918)
Hall, K.R., Eagleton, L.C., Acrivos, A., Vermeulen, T.: Pore- and solid-diffusion kinetics in fixed-bed adsorption under constant-pattern conditions. Ind. Eng. Chem. Fundam. 5(2), 212–223 (1966)
Freundlich, H.M.F.: Over the adsorption in solution. J. Phys. Chem. 57, 385–471 (1906)
Tsai, W.T., Lai, C.W., Hsien, K.J.: Effect of particle size of activated clay on the adsorption of paraquat from aqueous solution. J. Colloid Interface Sci. 263(1), 29–34 (2003)
Nakamoto, K., Ohkaku, N.: Metal isotope effect on metal-ligand vibrations. VI. Metal complexes of 8-hydroxyquinoline. Inorg. Chem. 10(4), 798–805 (1971)
Pastre, I.A., do Nascimento Oliveira, I., Moitinho, A.B.S., de Souza, G.R., Ionashiro, E.Y., Fertonani, F.L.: Thermal behavior of intercalated 8-hydroxyquinoline (oxine) in montmorillonite clay. J. Therm. Analy. Calom. 75, 661–669 (2004)
Khaorapapong, N., Kuroda, K., Ogawa, M.: Intercalation of 8-hydroxyquinoline into Al-montmorillonites by solid-solid reactions. Clays Clay Miner. 50, 428–434 (2002)
Hopkins, T.A., Meerholz, K., Shaheen, S., Anderson, M.L., Schmidt, A., Kippelen, B., Padias, A.B., Hall, H.K., Peyghambarian, N., Armstrong, N.R.: Substituted aluminum and zinc quinolates with blue-shifted absorbance/luminescence bands: synthesis and spectroscopic, photoluminescence, and electroluminescence characterization. Chem. Mater. 8, 344–351 (1996)
Acknowledgements
The author B. Mellah would like to congratulate Pr. K. Gloe for his 70th birthday. She is also gratefully acknowledge Pr. Yang Kim from Kumamoto University, Pr. Mauro Mocerino from Curtin University and Dr. Mohamed Triki from CNRSM for their helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mellah, B., Sall, D., Msaddak, I. et al. Intercalation of 8-hydroxyquinoline into Na(I)- and Zn(II)-Tunisian montmorillonites: characterization and luminescence properties of elaborated hybrids. J Incl Phenom Macrocycl Chem 94, 309–318 (2019). https://doi.org/10.1007/s10847-018-0826-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-018-0826-9