Abstract
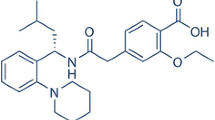
Telmisartan (TEL) is a BCS Class II drug having dissolution rate limited bioavailability. The aim of work was to enhance the solubility of TEL so that bioavailability problems are solved. β-Cyclodextrin (β-CD) based nanosponges (NSs) were formed by cross-linking β-CD with carbonate bonds, which were porous as well as nanosized. Drug was incorporated by solvent evaporation method. The effect of ternary component alkalizer (NaHCO3) on solubility of TEL was studied. In order to find out the solubilization efficiency of NS, phase solubility study was carried out. Saturation solubility and in vitro dissolution study of β-CD complex of TEL was compared with plain TEL and NS complexes of TEL. The NS and NS complexes of TEL were characterized by differential scanning calorimetry, powder X-ray diffraction, Fourier transform infrared spectroscopy, nuclear magnetic resonance and scanning electron microscope. It was found that solubility of TEL was increased by 8.53-fold in distilled water; 3.35-fold in 0.1 N HCl and 4.66-fold in phosphate buffer pH 6.8 by incorporating NaHCO3 in drug–NS complex than TEL. It was found that the NaHCO3 in NS based complex synergistically enhanced dissolution of TEL by modulating microenvironmental pH and by changing amorphization of the drug. The highest solubility and in vitro drug release was observed in inclusion complex prepared from NS and NaHCO3. An increase of 54.4 % in AUC was seen in case the ternary NS complex whereas β-CD ternary complex exhibited an increase of 79.65 %.












Similar content being viewed by others
References
Rossi, S., Ferrari, F., Bonferoni, M.C., Caramella, C.: Characterization of chitosan hydrochloride–mucin interaction by means of viscosimetric and turbidimetric measurements. Eur. J. Pharm. Sci. 10, 251–257 (2000)
Rekharsky, M.V., Inove, Y.: Complexation thermodynamics of cyclodextrins. Chem. Rev. 98, 1875–1918 (1998)
Connors, K.A.: The stability of cyclodextrin complexes in solution. Chem. Rev. 97, 1325–1358 (1997)
Szejtli, J.: Introduction and general overview of cyclodextrin chemistry. Chem. Rev. 98, 1743–1754 (1998)
Cavalli, R., Trotta, F., Tumiatti, W.: Cyclodextrin-based nanosponges for drug delivery. J. Incl. Phenom. Macrocycl. Chem. 56, 209–213 (2006)
Swaminathan, S., Pastero, L., Serpe, L., Trotta, F., Vavia, P., Aquilano, D., Trotta, M., Zara, G., Cavalli, R.: Cyclodextrin-based nanosponges encapsulating camptothecin: physicochemical characterization, stability and cytotoxicity. Eur. J. Pharm. Biopharm. 74, 193–201 (2010)
Velasquez, M.T.: Angiotensin II receptor blockers, a new class of antihypertensive drugs. Arch. Fam. Med. 5, 351–356 (1996)
Nakatani, M., Takeshi, S., Ohki, T., Toyoshima, K.: Solid TEL pharmaceutical formulations. US Patent 2004/0110813 A1, 2004
Phuong, H.L.T., Huyen, T.T., Beom-Jin, L.: Modulation of microenvironmental pH and crystallinity of ionizable TEL using alkalizers in solid dispersions for controlled release. J. Control. Release 129, 59–65 (2008)
Trotta, F., Tumiatti, W.: Cyclodextrin-based NS as a vehicle for antitumoral drugs. World Patent Number WO/2009/003656, 2009
Higuchi, T., Connors, K.: Phase solubility techniques. In: Reilly, C. (ed.) Advances in Analytical Chemistry and Instrumentation, pp. 117–212. Wiley–Interscience, New York (1965)
Swaminathan, S., Vavia, P.R., Trotta, F., Torne, S.: Formulation of betacyclodextrin based nanosponges of itraconazole. J. Incl. Phenom. Macrocycl. Chem. 57, 89–94 (2007)
Marques, M.: Dissolution media simulating fasted and fed state. Dissolution Technol. 11(2), 16 (2004)
Shimasaki, M., Yamashit, K.: Pharmacokinetics of 14C-TEL (2): plasma concentration and distribution of 14C-TEL after repeated oral administration to rats. Xenobiol. Metab. Dispos. 14, 432–436 (1999)
Brahmankar, D.M., Jaiswal, S.B.: Biopharmaceutics and Pharmacokinetics: A Treatise. Vallabh Prakashan, New Delhi (2005)
Chada, R., Kashid, N., Saini, A.: Account of analytical techniques employed for the determination of thermodynamics of inclusion complexation of drugs with cyclodextrins. J. Sci. Ind. Res. 63, 211–229 (2004)
Conflicts of interest
The author reports no conflicts of interest financial or otherwise.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rao, M., Bajaj, A., Khole, I. et al. In vitro and in vivo evaluation of β-cyclodextrin-based nanosponges of telmisartan. J Incl Phenom Macrocycl Chem 77, 135–145 (2013). https://doi.org/10.1007/s10847-012-0224-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-012-0224-7