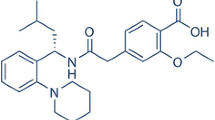
Abstract
In the present study influence of nature of selected cyclodextrins (CDs) and of methods of preparation of drug–CD complexes on the oral bioavailability, in vitro dissolution studies and pharmacodynamic activity of a sparingly water soluble drug rosuvastatin (RVS) was investigated. Phase solubility studies were conducted to find the interaction of RVS with β-CD and its derivatives, which indicated the formation of 1:1 stoichiometric inclusion complex. The apparent stability constant (K1:1) calculated from phase solubility diagram were in the rank order of β-CD < hydroxypropyl-β-cyclodextrin (HP-β-CD) < randomly methylated-β-cyclodextrin (RM-β-CD). Equimolar drug–CD solid complexes prepared by different methods were characterized by the Fourier transform infrared spectroscopy (FTIR), differential scanning calorimetry (DSC) and X-ray diffractometry (XRD). FTIR study demonstrated the presence of intermolecular hydrogen bonds and ordering of the molecule between RVS and CDs in inclusion complexes. DSC and XRD analysis confirmed formation of inclusion complex by freeze dried method with HP-β-CD and RM-β-CD. Aqueous solubility and dissolution studies indicated improved dissolution rates of prepared complexes in comparison with drug alone. Moreover, CD complexes demonstrated of significant improvement in reducing total cholesterol and triglycerides levels as compared to pure drug. However the in vivo results only partially agreed with those obtained from phase solubility studies.







Similar content being viewed by others
Abbreviations
- CDs:
-
Cyclodextrins
- RVS:
-
Rosuvastatin
- β-CD:
-
β-cyclodextrin
- HP-β-CD:
-
Hydroxypropyl-β-cyclodextrin
- RM-β-CD:
-
Randomly methylated-β-cyclodextrin
- KND:
-
Kneading
- PM:
-
Physical mixtures
- COEVP:
-
Coevaporation
- FZD:
-
Freeze dried
References
Sweetman, S.C.: Martindale: The Complete Drug Reference, 34th edn. Royal Pharmaceutical Society of Great Britain, London (2005)
Lennernäs, H., Fager, G.: Clinical pharmacokinetics and pharmacodynamics of HMG-CoA reductase inhibitors: similarities and dissimilarities. Clin. Pharmacokinet. 35, 403–425 (1997)
Chapman, M.J., McTaggart, F.: Optimizing the pharmacology of statins: characteristics of rosuvastatin. Atheroscler. Suppl. 2, 33–37 (2002)
Schachter, M.: Chemical, pharmacokinetic and pharmacodynamic properties of statins: an update. Fundam. Clin. Pharmacol. 19, 117–125 (2004)
Vyas, A., Saraf, S., Saraf, S.: Cyclodextrin based novel drug delivery systems. J. Incl. Phenom. Macrocycl. Chem. 62, 23–42 (2008)
Rekharsky, M.V., Inoue, Y.: Complexation thermodynamics of cyclodextrin. Chem. Rev. 98, 1875–1917 (1998)
Liu, L.X., Zhu, S.Y.: Preparation and characterization of inclusion complexes of prazosin hydrochloride with β-cyclodextrin and hydroxypropyl-β-cyclodextrin. J. Pharm. Biomed. Anal. 40(1), 122–127 (2006)
Liu, L., Zhu, S.: A study on the supramolecular structure of inclusion complex of β-cyclodextrin with prazosin hydrochloride. Carbohydr. Polym. 68(3), 472–476 (2007)
Szejtli, J.: Medicinal applications of cyclodextrins. Med. Res. Rev. 14, 364–368 (1994)
Bettinetti, G.P., Gazzaniga, A., Mura, P., Giordano, F., Setti, M.: Thermal behavior and dissolution properties of Naproxen in combinations with chemically modified β-cyclodextrins. Drug Dev. Ind. Pharm. 18, 39–53 (1992)
Higuchi, T., Connors, K.A.: Phase-solubility techniques. Adv. Anal. Chem. Instrum. 4, 117–212 (1965)
Karathanos, V.T., Mourtzinos, I., Yannakopoulou, K., Andrikopoulos, N.K.: Study of the solubility, antioxidant activity and structure of inclusion complex of vanillin with β-cyclodextrin. Food Chem. 101, 652–658 (2007)
Zingone, G., Rubessa, F.: Preformulation study of the inclusion complex warfarin–β-cyclodextrin. Int. J. Pharm. 29, 3–10 (2005)
Buchi, N.N., Chowdary, K.P.R., Murthy, K.V.R., Satyanarayana, V., Hayman, A.R., Becket, G.: Physicochemical characterization and dissolution properties of meloxicam–cyclodextrin binary systems. J. Pharm. Biomed. Anal. 35, 75–86 (2004)
Khan, K.A.: The concept of dissolution efficiency. J. Pharm. Pharmacol. 27, 48–49 (1975)
Schurr, P.E., Schultz, J.R., Parkinson, T.M.: Triton induced hyperlipidemia in rats as an animal model for screening hypolipidemic drugs. Lipids 7, 69–72 (1972)
Mukne, A.P., Nagarsenker, M.S.: Triamterene–β-cyclodextrin systems: preparation, characterization and in vivo evaluation. AAPS PharmSciTech. 5, E19 (2004)
Kurozumi, M., Nambu, N., Nagai, T.: Inclusion compounds of nonsteroidal antiinflammatory and other slightly water soluble drugs with alpha- and beta-cyclodextrins in powdered form. Chem. Pharm. Bull. 23, 3062–3068 (1975)
Winters, C.S., York, P., Timmins, P.: Solid state examination of a gliclazide:beta-cyclodextrin complex. Eur. J. Pharm. Sci. 5, 209–214 (1997)
Marques, H.C., Hadgraft, J., Kellaway, I.: Studies of cyclodextrin inclusion complexes. I. The salbutamol–cyclodextrin complex as studied by phase solubility and DSC. Int. J. Pharm. 63, 259–266 (1990)
Kim, K.H., Frank, M.J., Henderson, N.L.: Applications of differential scanning calorimetry to the study of solid drug dispersions. J. Pharm. Sci. 74, 283–289 (1985)
Mura, P., Adragna, E., Rabasco, A., Moyano, J., Pérez-Martìnez, J.I., Arias, M.J., Ginés, J.M.: Effects of the host cavity size and the preparation method on the physicochemical properties of ibuproxam–cyclodextrin. Drug Dev. Ind. Pharm. 25, 279–287 (1996)
Esclusa-Diaz, M., Torres-Labandeira, J., Kata, M., Vila-Jato, J.: Inclusion complexation of glibenclamide with 2-hydroxypropyl-β-cyclodextrin in solution and in solid state. Eur. J. Pharm. Sci. 1, 291–296 (1994)
Loftsson, T., Brewster, M.E.: Pharmaceutical applications of cyclodextrins. 1. Drug solubilization and stabilization. J. Pharm. Sci. 85, 1017–1025 (1996)
Damian, F., Blaton, N., Naesens, L., Balzarini, J., Kinget, R., Van den Mooter, G.: Physicochemical characterization of solid dispersion of the antiviral agent UC-781 with polyethylene glycol 6000 and Gelucire 44/14. Eur. J. Pharm. Sci. 10, 311–322 (2000)
Veiga, F., Fernandes, C., Maincent, P.: Influence of the preparation method on the physicochemical properties of tolbutamide cyclodextrin binary system. Drug Dev. Ind. Pharm. 27, 523–532 (2001)
Mura, P., Faucci, M.T., Parrini, P.L., Furlanetto, S., Pinzauti, S.: Influence of the preparation method on the physicochemical properties of ketoprofen cyclodextrin binary systems. Int. J. Pharm. 179, 117–128 (1999)
Mosher, G., Thompson, D.O.: In: Swarbrick, J., Boylan, J.C. (eds.) Encyclopedia of Pharmaceutical Technology, p. 531. Marcel Dekker, New York (2002)
Acknowledgment
The authors are thankful to UGC SAP, New Delhi for financial assistance. Authors are grateful to Ranbaxy, India for providing pure drug, Dr. Denis Simon and Emilie Van Oudendycke of Roquette, Lestrem, France and Signet Chemical Corpn. Pvt. Ltd. Mumbai, India for providing β-cyclodextrin, hydroxypropyl-beta-cyclodextrin, randomly methylated-β-cyclodextrin. Authors also extend their gratitude to Head, University Institute of Pharmacy, Pt. Ravishankar Shukla University, Raipur, Chhattisgarh, India for providing facilities to carry out research work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vyas, A. Preparation, characterization and pharmacodynamic activity of supramolecular and colloidal systems of rosuvastatin–cyclodextrin complexes. J Incl Phenom Macrocycl Chem 76, 37–46 (2013). https://doi.org/10.1007/s10847-012-0170-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-012-0170-4