Abstract
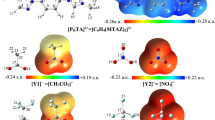
Complexation properties of dinaphthosulfide-substituted macrocyclic diamides 1 and 2 with some metal cations that have been obtained by conductometric method are described with quantum mechanics calculations. To do this, the most stable structures of ligands, Hg2+-ligand complexes, ligand-MeOH and ClO4 −–MeOH are optimized at HF/Lanl2dz level of theory and the most important interactions are analyzed by atoms in molecules (AIM) theory. These calculations predict the existence of strong interaction between Hg2+ cation and ligands 1 and 2, particularly, S–Hg2+ interaction. The different conductometric behaviors of complexation of ligands 1 and 2 with metal ions are interpreted on the basis of the calculated intramolecular hydrogen bonds in ligands and intermolecular hydrogen bonds between ligands and methanol as a solvent and perchlorate as a counter ion. In addition, binding energies between Hg2+ and ligands are also calculated by HF/Lanl2dz level of theory. Results show that all theoretical predictions are in line agreement with the experimental data.









Similar content being viewed by others
References
Fabbrizzi, L., Kaden, T.A., Perotti, A., Seghi, B., Siegfried, L.: Complexation of divalent and trivalent nickel and copper ions by rigid and flexible dioxo tetraaza macrocycles. Inorg. Chem. 25, 321–327 (1986)
Buseimaann, H.J.: The macrocyclic and cryptate effect. 7. Influence of structural changes on the complexation behavior of aza crown ethers and cryptands in different solvents. Inorg. Chim. Acta 120, 125–129 (1986)
Izatt, R.M., Pawlak, K., Bradshaw, J.S., Bmening, R.L.: Thermodynamic and kinetic data for macrocycle interactions with cations and anions. Chem. Rev. 91, 1721–2085 (1991)
Hourdakis, A., Popov, A.I.: Lithium-7, sodium-23, and cesium-133 NMR and far infrared study of alkali complexes with C222-dilactam in various solvents. J. Solut. Chem. 6, 299–307 (1977)
Gokel, G.W., Schall, O.F.: Crown ethers with amide lariat groups. In: Gokel, G.W. (ed.) Comprehensive supramolecular chemistry, pp. 97–152. Pergamon, Oxford (1996)
Mckervey, A., Schwing-Weill, M.J., Arnaud-Neu, F.: amide functionalized calixarenes. In: Gokel, G.W. (ed.) Comprehensive supramolcular chemistry, pp. 537–606. Pergamon, Oxford (1996)
Arnaud-Neu, F., Barboso, S., Casnati, A., Muzet, N., Pinallini, A., Ungaro, R., Schwing-Weill, M.J., Wipf, G.: Modulation of cation binding in calix[4]arene amides: synthesis, complexation and molecular modeling studies. J. Chem. Soc. Perkin Trans 2, 1727–1738 (1999)
lzatt, R.M., Bradshaw, J.S., Pawlak, K., Bmening, R.L., Tarbet, B.J.: Thermodynamic and kinetic data for macrocycle interaction with neutral molecules. Chem. Rev. 92, 1261–1354 (1992)
Goodman, M.S., Rose, S.D.: Photosensitized pyrimidine dimer splitting by a methoxyindole bound to a dimer-recognizing macrocycle. J. Org. Chem. 57, 3268–3270 (1992)
Kluger, R., Tsao, B.: Molecular reception catalysis of the decarboxylation of N-carboxyimidazolidinone. A model for activation by distortion of N-carboxybiotin. J. Am. Chem. Soc. 115, 2089–2090 (1993)
Shockravi, A., Shamsipur, M., Fattahi, H., Taghdiri, M., Heidaryan, D., Alizadeh, K., Rostami, E., Abbaszadeh, M., Yousefi, A.: Efficient synthesis and metal cations complexation of some novel dinaphthosulide-substituted macrocyclic diamides. J. Incl. Phenom. Macrocycl. Chem. 61, 153–160 (2008)
Eshghi, H., Seyedi, S.M., Sandarooss, R.: Synthesis of novel disulfide-bridged dilactam crown ethers. Chin. Chem. Lett. 18, 1439–1442 (2007)
Masrournia, M., Alizamani, H., Mohamadzadeh, H., Seyedi, S.M., Ganjali, M.R., Eshghi, H.: A silver(I) PVC-membrane sensor based on synthesized dilaktam crown ether. J. Chil. Chem. Soc. 53, 63–67 (2009)
Adams, D.M., Blandamer, M.J., Symons, M.C.R., Waddington, D.: Part 39. Infra-red and Raman studies of aqueous and non-aqueous solutions containing perchlorates. Trans. Faraday Soc. 67, 611–617 (1971)
Butler, R.N., Symons, M.C.R.: Part 26. Nuclear magnetic resonance studies of electrolyte solutions: cation and anion shifts in the hydroxylic proton resonance of methanol. Trans. Faraday Soc. 65, 945–949 (1969)
Butler, R.N., Symons, M.C.R.: Part 29. Nuclear magnetic resonance studies of electrolyte solutions: ion-solvent interactions in methanol. Trans. Faraday Soc. 65, 2559–2566 (1969)
Frisch, M.J., et al.: Gaussian 03 (Revision B.3). Gaussian, Inc., Pittsburgh (2003)
Biegler-König, F., Schönbohm, J., Bayles, D.: AIM2000-A program to analyze and visualize atoms in molecules. J. Comput. Chem. 22, 545–559 (2001)
Bader, R.F.W.: Atoms in molecules: a quantum theory. Oxford University Press, Oxford (1990)
Bader, R.F.W.: A quantum theory of molecular structure and its applications. Chem. Rev. 91, 893–928 (1991)
Bader, R.F.W., Essen, H.: The characterization of atomic interactions. J. Chem. Phys. 80, 1943–1960 (1984)
Popelier, P.: Atoms in molecules. An introduction, 1st edn. Prentice Hall, Pearson Educational Limited, Englewood Cliffs (2000)
Parthasarathi, R., Amutha, R., Subramanian, V., Nair, B.U., Ramasami, T.: Bader’s and reactivity descriptors’ analysis of DNA base pairs. J. Phys. Chem. A 108, 3817–3828 (2004)
Barbalace, K.: Periodic table of elements—sorted by ionic radius. http://EnvironmentalChemistry.com. 1995–2009. http://klbprouctions.com/. Accessed 17 Dec 2009
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Taghdiri, M., Behjatmanesh-Ardakani, R., Shockravi, A. et al. Study on the interaction between dinaphthosulfide-substituted macrocyclic diamides and some metal ions: experimental measurements vs. quantum mechanical calculations. J Incl Phenom Macrocycl Chem 73, 255–262 (2012). https://doi.org/10.1007/s10847-011-0050-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-011-0050-3