Abstract
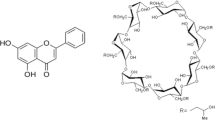
The objectives of the present investigation were to prepare and characterize solid inclusion complexes of Etodolac (ETD) with β-cyclodextrin (β-CD) in order to study the effect of complexation on the dissolution rate of ETD, a hydrophobic guest molecule. Phase solubility curve was classified as a typical AL-type for the cyclodextrins (CD’s), showing that soluble complex was formed. The inclusion complexes in the molar ratio of 1:1 and 1:2 (β-CD–ETD) were prepared by various methods such as kneading, co-evaporation and in molar ratio of 1:1 by spray dried technique respectively. The molecular behaviors of ETD in all samples were characterized by nuclear magnetic resonance (NMR) spectroscopy, fourier-transform infrared (FTIR) spectroscopy, differential scanning calorimetry (DSC) and powder X-ray diffraction (PXRD) studies and Scanning Electron microscopy (SEM) analysis. The results of these studies indicated that complexes prepared by kneading, co-evaporation and spray drying techniques showed inclusion of the ETD molecule into the CD’s cavities. The highest improvement in in vitro dissolution profiles was observed in complexes prepared with spray dried technique. Mean in vitro dissolution time indicated significant difference between the release profiles of ETD from complexes and physical mixtures and from pure ETD.







Similar content being viewed by others
References
Davis, M.E., Brewster, M.E.: Cyclodextrin-based pharmaceutics: past, present and future. Nat. Rev. Drug Discov. 3, 1023–1035 (2004)
Loftsson, T., Jarho, P., Masson, M., Jarvinen, T.: Cyclodextrins in drug delivery. Expert Opin. Drug Deliv. 2, 335–351 (2005)
Loftsson, T.: Cyclodextrins and biopharmaceutical classification system of drugs. J. Incl. Phenom. Macrocycl. Chem. 44, 63–67 (2002)
Inoue, K., Fujisawa, H., Sasaki, Y., Nishimura, T., Nishimura, I., Inoue, Y., Yokota, M., Masuda, T., Ueda, F., Shibata, Y., Kimura, K., Inoue, K., Komiya, Y., Nishioka, J.: Pharmacological properties of new non steroidal anti-infammatory agent etodolac. Arzneim-Forsch. 41, 228–234 (1991)
Jacob, G., Sanda, M., Mullane, J., Kemmedy, J., Barbetti, M., Suleski, P.: Long term evaluation of the efficacy and safety of etodolac in the treatment of rheumatoid arthritis. Adv. Ther. 2, 82–95 (1985)
Milic-Askrabic, J., Rajic, D.S., Tasic, Lj., Djuric, S., Kasa, P., Pintye-Hodi, K.: Etodolac and solid dispersion with β-cyclodextrin. Drug Dev. Ind. Pharm. 23, 1123–1129 (1997)
Martin, A.: Complexation and protein binding. In: Martin, A. (ed.) Physical Pharmacy: Physical Chemical Principles in the Pharmaceutical Sciences, pp. 251–283. Waverly International Publishers, Maryland (1995)
Li, P., Zhao, L., Yalkowsky, S.: Combined effect of cosolvent and cyclodextrin on solubilization of non-polar drugs. J. Pharm. Sci. 88, 1107–1111 (1999)
Higuchi, T., Connors, A.K.: Phase-solubility techniques. In: Reill, C.N. (ed.) Advances in Analytical Chemistry and Instrumentation, pp. 117–212. Wiley, New York (1965)
Uekama, K., Irie, T., Sunada, M., Otagiri, M., Arimatsu, Y., Nomura, S.: Alleviation of prochlorperazine-induced primary irritation of skin by cyclodextrin complexation. Chem. Pharm. Bull. 30, 3860–3862 (1982)
Trapani, G., Latrofa, A., Franco, M., Pantaleo, M.R., Sanna, E., Massa, F., Tuveri, F., Liso, G.: Complexation of zolpidem with 2-hydroxypropyl-beta-, methyl-beta-, and 2-hydroxypropyl-gamma-cyclodextrin: effect on aqueous solubility, dissolution rate, and ataxic activity in rat. J. Pharm. Sci. 89, 1443–1451 (2000)
Koester, L.S., Guterres, S.S., Le Roch, M., Eifler-Lima, V.L., Zuanazzi, J.A., Bassani, V.L.: Ofloxacin/beta-cyclodextrin complexation. Drug Dev. Ind. Pharm. 27, 533–540 (2001)
Connors, K.A.: The stability of cyclodextrin complexes in solution. Chem. Rev. 97, 1325–1357 (1997)
Nishijo, J., Nagai, M.: Inclusion complex of 8-anilinonaphthalene-1-sulfonate with β-cyclodextrin. J. Pharm. Sci. 80, 58–62 (1991)
Damarco, P.V., Thakkar, A.L.: Cycloheptaamylose inclusion complexes of barbiturates: correlation between proton magnetic resonance and solubility studies. J. Pharm. Sci. 60, 652–653 (1971)
Uekama, K., Otagiri, M., Kane, Y., Tanaka, S., Ikeda, K.: Inclusion complexes of cinnamic acids with cyclodextrins: mode of inclusion in aqueous solution. Chem. Pharm. Bull. 23, 1421–1430 (1975)
Veiga, M.D., Diaz, P.J., Ahsan, F.: Interactions of griseofulvin with cyclodextrins in solid binary systems. J. Pharm. Sci. 55, 891–900 (1998)
Mura, P., Faucci, M.T., Parrini, P.L., Furlanetto, S., Pinzauti, S.: Influence of the preparation method on physicochemical properties of ketoprofen-cyclodextrin binary systems. Int. J. Pharm. 179, 117–128 (1999)
Moyano, J.R., Arias Blanco, M.J.A., Gines, J.M., Perez-Martinez, J.I., Rabasco, A.M.: Dissolution behavior of oxazepam in the presense of cyclodextrins: Evaluation of oxazepam Dimeb binary system. Drug Dev. Ind. Pharm. 23, 379–385 (1997)
Esclusa-Diaz, M.T., Gayo-Otero, M., Perez-Marcos, M.B., Vila-Jato, J.L., Torres Labandeira, J.J.: Preparation and evaluation of ketoconazole-beta-cyclodextrin multicomponent complexes. Int. J. Pharm. 142, 183–187 (1996)
Mura, P., Adragna, E., Rabasco, A.M., Moyano, J.R., Perez-Martinez, J.I., Arias, M.J., Gines, J.M.: Effects of the host cavity size and the preparation method on the physicochemical properties of ibuproxam-cyclodextrin systems. Drug Dev. Ind. Pharm. 25, 279–287 (1999)
Sinha, V.R., Anitha, R., Ghosh, S., Nanda, A., Kumria, R.: Complexation of celecoxib with beta-cyclodextrin: characterization of the interaction in solution and in solid state. J. Pharm. Sci. 94, 676–687 (2005)
Becirevic-Lacan, M., Filipovic-Grcic, J., Skalko, N., Jalsenjak, J.: Dissolution characteristics of nifedipine complexes with beta-cyclodextrins. Drug Dev. Ind. Pharm. 22, 1213–1236 (1996)
Yamada, T., Sait, N., Imai, T., Otagiri, M.: Effect of grinding with hydropropyl cellulose in the dissolution and particle size of a poorly water-soluble drug. Chem. Pharm. Bull. 47, 1311–1313 (1999)
Acknowledgements
We would like to thank Ranbaxy Pvt. Ltd. for donating us the gift sample of ETD. We are grateful to S.A Chemicals Pvt. Ltd. for providing us the formulation excipients.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sinha, V.R., Amita, Chadha, R. et al. Enhancing the dissolution of hydrophobic guests using solid state inclusion complexation: characterization and in vitro evaluation. J Incl Phenom Macrocycl Chem 66, 381–392 (2010). https://doi.org/10.1007/s10847-009-9655-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-009-9655-1