Abstract
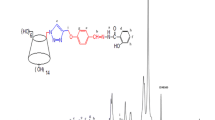
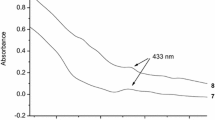
One or two coumarin units were incorporated to the primary face of γ-cyclodextrin (γ-CD), and the resultant coumarin derivatives were employed to harvest the chemical energy generated in the reaction of bis(trichlorophenyl)oxalate with hydrogen peroxide. In comparison with the coumarin without CD cavity for molecular recognition, the coumarin–CD conjugates demonstrated much higher chemiluminescence inetensity, indicating that the CD moiety remarkably improves the chemical energy transfer.





Similar content being viewed by others
References
Hadd, A.G., Lehmpuhl, D.W., Kuck, L.R., Birks, J.W., Mell, G.P.: Chemiluminescence demonstration illustrating principles of ester hydrolysis reactions. J. Chem. Educ. 76, 1237–1240 (1999)
Bortolus, P., Marconi, G., Monti, S., Mayer, B., Kohler, G., Grabner, G.: Interaction of 4-hydroxybiphenyl with cyclodextrins: effect of complex structure on spectroscopic and photophysical properties. Chem. Eur. J., 6, 1578–1591 (2000); Ramamurthy, V., Eaton, D. F.: Photochemistry and photophysics within cyclodextrin cavities. Acc. Chem. Res. 21, 300–309 (1988)
Lehn, J.-M.: Supramolecular Chemistry. VCH, Weinheim (1995); Balzani, V., Scandola, F.: Comprehensive supramolecular chemistry. In: Atwood, J.L.J., Davies, E.D., MacNicol, D.D., Votle, F., Lehn, J.-M., Reinhoudt, D.N. (eds.) Photochemical and Photophysical Devices, vol. 10, pp. 687–746. Pergamon, Oxford (1996)
Tamura, M., Gao, D., Ueno, A.: A polyrotaxane series containing a-cyclodextrin and naphthalene-modified α-cyclodextrin as a light-harvesting antenna system. Chem. Eur. J. 7, 1390–1397 (2001); Berberan-Santos, M.N., Choppinet, P., Fedorov, A., Jullien, L., Valeur, B.: Multichromophoric cyclodextrins. 8. Dynamics of homo- and heterotransfer of excitation energy in inclusion complexes with fluorescent dyes. J. Am. Chem. Soc. 122, 11876–11886 (2000); Takakusa, H., Kikuchi, K., Urano, Y., Higuchi, T., Nagano, T.: Intramolecular fluorescence resonance energy transfer system with coumarin donor included in β-cyclodextrin. Anal. Chem. 73, 939–942 (2001); Tabushi, I., Fujita, K., Yuan, L.C., Specific host-guest energy transfer by use of b-cyclodextrin rigidly capped with a benzophenone moiety. Tetrahedron Lett. 2503–2506 (1977)
Yuan, D.-Q., Kishikawa, N., Yang, C., Koga, K., Kuroda, K., Fujita, K.: Fluorophore-capped cyclodextrins as efficient chemical-to-light energy converters. Chem. Commun. 416–417 (2003)
Yuan, D.-Q., Tahara, T., Chen, W.-H., Okabe, Y., Yang, C., Yagi, Y., Nogami, Y., Fukudome, M., Fujita, K.: Functionalization of cyclodextrins via reactions of 2,3-anhydrocyclodextrins. J. Org. Chem. 68, 9456–9466 (2003), and references therein
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yuan, DQ., Zhao, Y., Fukudome, M. et al. Coumarin-conjugated cyclodextrins: remarkable enhancement of the chemical-to-light energy transfer efficiency. J Incl Phenom Macrocycl Chem 57, 125–129 (2007). https://doi.org/10.1007/s10847-006-9207-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-006-9207-x