Abstract
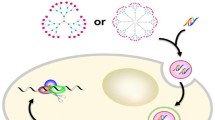
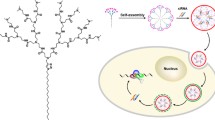
RNA interference (RNAi) is the mechanism of gene silencing-mediated messenger RNA degradation by small interference RNA (siRNA), which becomes a powerful tool for genetic analysis and novel gene therapy. However, one of the major obstacles for siRNA delivery is the difficulty to cross the biological membrane due to its hydrophilicity and high molecular weight. We evaluated the potential use of the starburst polyamidoamine dendrimer (generation 3) conjugate with α-cyclodextrin (α-CyD) having an average degree of substitution of 2.4 (α-CDE conjugate) as a siRNA carrier for RNAi. The ternary complex composed of pGL2 control vector (pDNA)/pGL2 siRNA/α-CDE conjugate showed higher pGL2 siRNA sequence-specific gene silencing effects without off-target effects than those of commercial transfection reagents such as Lipofectamine™2000 (LP), TransFast™ (TF) and Lipofectin™ (LF). These results suggest that α-CDE conjugate has the potential to be a novel carrier for siRNA.
Similar content being viewed by others
References
Gilmore L.R., Fox S.P., Hollins A.J., Sohail M., Akhtar A., J. Drug Target. 12: 315 (2004)
Simeoni F., Morris M.C., Heitz F., Divita G., Nucleic Acids Res. 31: 589 (2003)
Soutschek J., Akinc A., Bramlage B., Charisse K., Constien R., Donoghue M., Elbashir S., Geick A., Hadwiger P., Harborth J., John M., Kesavan V., Lavine G, Pandey R.K., Racie T., Rajeev K.G., Röhl I., Tooudjarska I., Wang G., Wuschko S., Bumcrot D., Koteliansky V., Limmer S., Manoharan M., Vornlocher H.P., Nature432: 173 (2004)
Elbashir S.M., Harborth J., Lendeckel W., Yalcin A., Weber K., Tuschl T., Nature 411: 494 (2001)
Qian Z.K., Xuan B.Q., Min T.S., Xu J.F., Li L., Huang W.D., World J. Gastroenterol. 11: 1297 (2005)
Spagnou S., Miller A.D., Keller M., Biochemistry43: 13348 (2004)
Gebhart C.L., Kabanov A.V., J. Control. Release 73: 401 (2001)
Matulis D., Rouzina I., Bloomfield V.A., J. Biol. Chem. 273: 7507 (1998)
Gebhart C.L., Kabanov A.V., J. Control. Release 73: 401 (2001)
Matulis D., Rouzina I., Bloomfield V.A., J. Mol. Biol. 296: 1053 (2000)
Arima H., Kihara F., Hirayama F., Uekama K., Bioconjug. Chem. 12: 476 (2001)
Kihara F., Arima H., Tsutsumi T., Hirayama F., Uekama K., Bioconjug. Chem. 13: 1211 (2002)
Kihara F., Arima H., Tsutsumi T., Hirayama F., Uekama K., Bioconjug. Chem. 14: 342 (2003)
Hamasaki K., Kogure K., Ohwada K., Toxicon 34: 490 (1996)
Carol A.S., Mechelle H., Mechael J.V., Robert H.S., Bryan R.G.W., Nat. Cell Biol. 5: 834(2003)
Karikó K., Bhuyan P., Capodici J., Weissman D., J. Immunol. 172: 6545 (2004)
Ryther R.C., Flynt A.S., Phillipis J.A., Patton J.G., Gene Ther. 12 5: (2005)
Spagnou S., Miller A.D., Keller M., Biochemistry43: 13348 (2004)
Yano J., Hirabayashi K., Nakagawa S., Yamaguchi T., Nogawa M., Kashimori I., Naito H., Kitagawa H., Ishiyama K., Ohgi T., Irimura T., Clin. Cancer Res. 10: 7721 (2004)
Acknowledgements
This work was supported by a Grant-in-Aid from Tokyo Biochemical Research Foundation, a Grant-in-Aid from the Research Foundation for Pharmaceutical Sciences and a Grant-in-Aid for Scientists Research from Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsutsumi, T., Arima, H., Hirayama, F. et al. Potential Use of Dendrimer/α-Cyclodextrin Conjugate as a Novel Carrier for Small Interfering RNA (siRNA). J Incl Phenom Macrocycl Chem 56, 81–84 (2006). https://doi.org/10.1007/s10847-006-9065-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-006-9065-6