Abstract
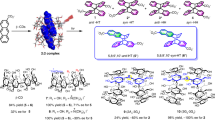
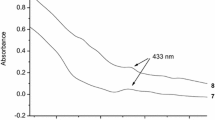
Two pyridinium groups were introduced into β-cyclodextrin (β-CD) at the A and E glucose units to make a molecular flask for controlling the stereo-selectivity of photodimerization of 2-anthracenecarboxylic acid. When the photodimerization of 2-anthracenecarboxylic acid was carried out in the presence of bispyridinio-appended β-CD, the relative yield of one of the configurational isomers was increased 1.5-fold compared to the corresponding yield in aqueous solution. The optical yields of the photodimerization reaction products also increased more than 10-fold by the addition of bispyridinio-appended β-CD.
Similar content being viewed by others
References
J.Szejtli and T.Osa,(eds.): Comprehensive Supramolecular Chemistry,Vol.3,Pergamon, Oxford (1996).
J.Szejtli: Cyclodextrin Technology,Kluwer, Dordrecht (1998).
G.Wenz: Angew.Chem.,Int.Ed.Engl. 33,803 (1994).
A.Ueno, K.Takahashi,and T.Osa: J.Chem.Soc.,Chem. Commun. 921 (1980).
M.Itoh and Y.Fujiwara: Bull.Chem.Soc.Jpn. 57, 2261 (1984).
T.Tamaki, T.Kokubu,and K.Ichimura: Tetrahedron 43,1485 (1987).
A.Nakamura and Y.Inoue: J.Am.Chem.Soc.125,966–972 (2003).
K.Fujita, H.Yamamura, T.Imoto, T.Fujioka,and K.Mihashi: J.Org.Chem. 53,1943 (1988).
M.Matsui, M.Fujie,and H.Sakate: Bull.Chem.Soc.Jpn. 61, 3409 (1988).
H.Ikeda, H.-T. Moon, Y.-Q. Du,and F.Toda, Supramol.Chem. 1,337–342 (1993).
H.Ikeda, Y.Iidaka,and A.Ueno: Org.Lett. 5,1625–1627 (2003).
K.Fujita, S.Ejima,and T.Imoto: Chem.Lett. 11 (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ikeda, H., Nihei, T. & Ueno, A. Stereoselective Photodimerization of 2-Anthracenecarboxylic Acid Using a Cation-Charged PSMP12-Cyclodextrin Template. Journal of Inclusion Phenomena 50, 63–66 (2004). https://doi.org/10.1007/s10847-004-8840-x
Issue Date:
DOI: https://doi.org/10.1007/s10847-004-8840-x