Abstract
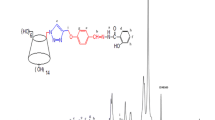
The complexation behavior of a series of electron donor–accepter (D–A) covalently linked compounds with cyclodextrins (CyDs) has been reviewed. The structure of these complexes showed rotaxane type conformations, which were investigated by means of 1 H NMR spectrometry. The complex formations induced a fixation of a conformation of guest D–A molecules, and D–A distances were enlarged. Thus, accurate photo-induced charge transfer behaviors can be discussed.
Similar content being viewed by others
References
GL Closs JR Miller (1988) Science 240 440
M.R. Wasielewski (1992) Chem Rev 92 435
D Gust TA Moore AL Moore (1993) Acc Chem Res 26 198
NJ Turro (1978) Modern Molecular Photochemistry Benjamin Cummings Menlo Park
GJ Kavarnos NJ Turro (1986) Chem Rev 86 401
UE Steiner T Ulrich (1989) Chem Rev 89 51
H Hayashi JF Rabek (1990) Photochemistry and Photophysics CRC Press Boca Raton 59
MCT Fyfe JF Stoddart (1997) Acc Chem Res 30 393
ML Bender M Komiyama (1978) Cyclodextrin Chemistry Springer-Verlag New York
G Wenz (1994) Angew Chem Int Ed Engl 33 803
A Harada J Li M Kamachi (1992) Nature 356 325
H. Ogino (1993) New J Chem 17 683
SA Nepogodiev JF Stoddart (1998) Chem Rev 98 1959
H Saito H Yonemura H Nakamura T Matsuo (1990) Chem. Lett. 535 ␣
H Yonemura H Saito S Matsushita H Nakamura T Matsuo (1989) Tetrahedron Lett 30 3143
H Yonemura M Kasahara H Saito H Nakamura T Matsuo (1992) J Phys Chem 92 5765
H Yonemura M Kasahara H Saito H Nakamura T Matsuo (1992) J Phys Chem 96 5765
T Ujiie T Morozumi T Kimura T Ito H Nakamura (2002) J Inclusion Phenom 42 301
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morozumi, T., Kimura, T., Ujiie, T. et al. Rotaxane Type Complexation Behavior of Cyclodextrins with a Series of Donor–Acceptor Covarently Linked Compounds. J Incl Phenom Macrocycl Chem 50, 105–110 (2004). https://doi.org/10.1007/s10847-003-8849-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10847-003-8849-1