Abstract
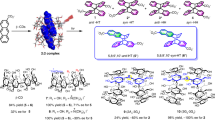
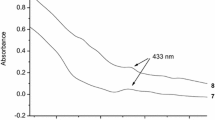
Two pyridinium groups were introduced into β-cyclodextrin (β-CD) at the A and E glucose units to make a molecular flask for controlling the stereo-selectivity of photodimerization of 2-anthracenecarboxylic acid. When the photodimerization of 2-anthracenecarboxylic acid was carried out in the presence of bispyridinio-appended β-CD, the relative yield of one of the configurational isomers was increased 1.5-fold compared to the corresponding yield in aqueous solution. The optical yields of the photodimerization reaction products also increased more than 10-fold by the addition of bispyridinio-appended β-CD.
Similar content being viewed by others
References
J Szejtli T Osa (Eds) (1996) Comprehensive Supramolecular Chemistry, Vol 3 Pergamon Oxford
J Szejtli (1998) Cyclodextrin Technology Kluwer Pergamon
G Wenz (1994) Angew Chem Int Ed Engl 33 803
A Ueno K Takahashi T Osa (1980) Chem Soc Chem Commun 921 ␣–␣
M Itoh Y Fujiwara (1984) Bull Chem Soc Jpn 57 2261
T Tamaki T Kokubu K Ichimura (1987) Tetrahedron 43 1485
A Nakamura Y Inoue (2003) Bull Chem Soc 125 966–972
K Fujita H Yamamura T Imoto T Fujioka K Mihashi (1988) J Org Chem 53 1943
M. Matsui M Fujie H Sakate (1988) Bull Chem Soc Jpn 61 3409
H Ikeda H-T Moon Y-Q Du F Toda (1993) Supramol Chem 1 337–342
H Ikeda Y Iidaka A Ueno (2003) Org Lett 5 1625–1627
K Fujita S Ejima T Imoto (1985) Chem Lett 11 ␣–␣
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is dedicated to the honor and memory of Professor Akihiko Ueno, who passed away on March 23, 2003
Rights and permissions
About this article
Cite this article
Ikeda, H., Nihei, T. & Ueno, A. Stereoselective Photodimerization of 2-Anthracenecarboxylic Acid Using a Cation-Charged PSMP12-Cyclodextrin Template. J Incl Phenom Macrocycl Chem 50, 63–66 (2004). https://doi.org/10.1007/s10847-003-8840-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10847-003-8840-x