Abstract
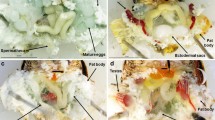
We investigated the effects of photoperiod and temperature on the development of the endangered butterfly Zizina emelina under laboratory conditions, and its seasonal occurrence in Osaka Prefecture, central Japan in 2009. At 20 °C most individuals reared under conditions of <13 h of light/day entered diapause during the larval stage, whereas those reared under ≥14 h of light/day developed into adults without any developmental arrest. At 25 °C, none of the individuals entered larval diapause irrespective of the photoperiod. The critical photoperiod for diapause induction at 20 °C was 12 h 40 min. Almost all larvae that had been in diapause for 2 months (with or without chilling at 10 °C) developed into adults, and the mean pupation period after transfer to 25 °C under 16 h of light/day differed little between the two groups, suggesting that chilling is not necessary to terminate larval diapause in Z. emelina. Adults were continuously found from late April to late October, with a low population density during spring. We estimate that this species has 5–6 generations in Osaka, according to the developmental threshold, effective accumulated temperature, and critical photoperiod data obtained from the rearing experiment. The estimated seasonal life cycle was similar to the seasonal prevalence observed in the field.






Similar content being viewed by others
References
Danilevskiĭ AS (1965) Photoperiodism and seasonal development of insects. Oliver & Boyd, Edinburgh
Danks HV (1987) Insect dormancy: an ecological perspective. Biological Survey of Canada (Terrestrial Arthropods), Ottawa
Denlinger DL (1985) Hormonal control of diapause. In: Kerkut GA, Gilbert LI (eds) Comprehensive insect physiology, biochemistry and pharmacology, vol 8. Pergamon Press, Oxford, pp 353–412
Denlinger DL (2001) Interrupted development: the impact of temperature on insect diapause. In: Arkinson D, Thorndyke M (eds) Environment and animal development: genes, life histories, and plasticity. Garland Science Publishing, New York, pp 235–250
Denlinger DL, Yocum GD, Rinehart JP (2011) Hormonal control of diapause. In: Gilbert LI (ed) Insect endocrinology. Academic Press, UK, pp 430–463
Fagerstrom T, Wiklund C (1982) Why do males emerge before females? Protandry as a mating strategy in male and female butterflies. Oecologia 52:164–166
Fukaya M, Mitsuhashi J (1957) The Hormonal control of larval diapause in the rice stem borer, Chilo suppressalis. I. Some factors in the head maintaining larval diapause. Jpn J Appl Entomol Zool 1:145–154
Fukaya M, Mitsuhashi J (1958) The Hormonal control of larval diapause in the rice stem borer, Chilo suppressalis. II. The activity of the corpora allata dureing the diapauseing period. Jpn J Appl Entomol Zool 2:223–226
Fukuda H, Hama E, Kuzuya T, Takahashi A, Takahashi M, Tanaka B, Tanaka H, Wakabayashi M, Watanabe Y (1984) The life histories of butterflies in Japan. III. Hoikusha, Osaka, Japan (in Japanese with English abstract)
Gelman DB, Woods CW (1983) Haemolymph ecdysteroid titers of diapause-and nondiapause-bound fifth instars and pupae of the European corn borer, Ostrinia nubilalis (Hübner). Comp Biochem Physiol A Physiol 76:367–375
Gelman DB, Thyagaraja BS, Kelly TJ, Masler EP, Bell RA, Borkovec AB (1992) Prothoracicotropic hormone levels in brains of the European corn borer, Ostrinia nubilalis: diapause versus the non-diapause state. J Insect Physiol 38:383–395
Ichinosé T (1974) Pupal diapause in some Japanese papilionid butterflies, with special reference to the difference in photoperiodic response between the diapausing pupae of Papilio maackii tutanus Fenton and P. xuthus Linnaeus. Kontyu 42:439–450
Ishii M (1980) Diapause and overwinter in Parnara guttata and Pelopedas mathias. Nat Insects 15:17–26 (in Japanese)
Ishii M (2009) Importance of the satoyama landscapes for conservation of biodiversity. In: Mano T, Fujii H (eds) Decline and conservation of butterflies and moths in Japan. VI. The Lepidopterological Society of Japan, Tokyo, pp 3–11
Ishii M, Hidaka T (1979) Seasonal polymorphism of the adult rice-plant skipper, Parnara guttata guttata (Lepidoptera: Hesperiidae) and its control. Japn Soc Appl Entomol Zool 14:173–184
Ishii M, Nakamura Y (2012) Development and future of insect conservation in Japan. In: New TR (ed) Insect conservation: past, present and prospects. Springer, Berlin, pp 339–357
Ishii M, Hirai N, Hirowatari T (2008) The occurrence of an endangered lycaenid, Zizina emelina (de l’Orza) (Lepidoptera, Lycaenidae), in Osaka International Airport, central Japan. Trans Lepidopterol Soc Jpn 59:78–82
Japan Meteorological Agency (2009) Weather, climate & earthquake information. Japan Meteorological Agency. http://www.jma.go.jp/jma/indexe.html. Accessed 21 Jan 2015
Kato Y (2000) Interpopulational variation in pupal diapause of the butterfly Atrophaneura alcinous (Klug) (Lepidoptera, Papilionidae) in the Kanto District, eastern Japan. Trans Lepidopterol Soc Jpn 51:233–242
Kim SH, Kim NJ, Hong SJ, Lee YB, Park HC, Je YH, Lee KP (2014) Environmental induction of larval diapause and life-history consequences of post-diapause development in the Large Copper butterfly, Lycaena dispar (Lepidoptera: Lycaenidae). J Insect Conserv 18:693–700
Kiritani K (2012) The low development threshold temperature and the thermal constant in insects and mites in Japan (2nd edition). Bull Natl Inst Agro Environ Sci 31:1–74
Mano T, Fujii H (2009) The red data lists of butterflies in 43 Prefectures, Japan. In: Mano T, Fujii H (eds) Decline and conservation of butterflies and moths in Japan. VI. The Lepidopterological Society of Japan, Tokyo, pp 107–265 (in Japanese)
Mikami H (2011) A note of Zizina emelina in Noto Peninsula. Tobu 209:1–6 (in Japanese)
Ministry of Environment, Japan (2012) Listed species of red data. http://www.biodic.go.jp/rdb/rdb_f.html. Accessed 21 Jan 2015
Minohara S, Morichi S, Hirai N, Ishii M (2007) Distribution and seasonal occurrence of the lycaenid, Zizina emelina (de l’Orza) (Lepidoptera, Lycaenidae), around the Osaka International Airport, central Japan. Trans Lepidopterol Soc Jpn 58:421–432
Mitsuhashi J, Fukaya M (1960) The Hormonal control of larval diapause in the rice stem borer, Chilo suppressalis. III. Histological studies on the neurosecretory cells of the brain and the secretory cells of the corpora allata during diapause and post diapause. Jpn J Appl Entomol Zool 4:127–134
Nakamura Y (2003) Current status and the future of butterfly conservation in Japan. In: Sunose T, Eda K (eds) Decline and conservation of butterflies in Japan. V. Lepidopterological Society of Japan, Tokyo, pp 171–176
Nylin S (2013) Induction of diapause and seasonal morphs in butterflies and other insects: knowns, unknowns and the challenge of integration. Physiol Entomol 38:96–104
Oliver CG (1969) Experiments on the diapause dynamics of Papilio polyxenes. J Insect Physiol 15:1579–1589
Rock GC, Shaffer PL (1983) Developmental rates of codling moth (Lepidoptera, Olethreutidae) reared on apple at 4 constant temperatures. Environ Entomol 12:831–834
Sakai T, Masaki S (1965) Photoperiod as a factorcausing seasonal forms in Lycaena phlaeas daimio Seitz (Lepidoptera: Lycaenidae). Kontyu 33:275–283
Sims SR, Shapiro AM (1983) Pupal diapause in Battus philenor (Lepidoptera, Papilionidae). Ann Entomol Soc Am 76:407–412
Singtripop T, Oda Y, Wanichacheewa S, Sakurai S (2002) Sensitivities to juvenile hormone and ecdysteroid in the diapause larvae of Omphisa fuscidentalis based on the hemolymph trehalose dynamics index. J Insect Physiol 48:817–824
Sunose T, Eda K (2003) The red data lists of butterflies in 43 Prefectures, Japan. In: Sunose T, Eda K (eds) Decline and conservation of butterflies in Japan. Lepidopterological Society of Japan, Tokyo, pp 1–169 (in Japanese)
Wiklund C, Fagerström T (1977) Why do males emerge before females? A hypothesis to explain the incidence of protandry in butterflies. Oecologia 31:153–158
Wiklund C, Lindfors V, Forsberg J (1996) Early male emergence and reproductive phenology of the adult overwintering butterfly Gonepteryx rhamni in Sweden. Oikos 75:227–240
Yago M, Hirai N, Kondo M, Tanikawa T, Ishii M, Wang M, Williams M, Ueshima R (2008) Molecular systematics and biogeography of the genus Zizina (Lepidoptera: Lycaenidae). Zootaxa 1746:15–38
Yoshio M, Ishii M (1998) Geographical variation of pupal diapause in the great mormon butterfly, Papilio memnon L. (Lepidoptera: Papilionidae), in Western Japan. Appl Entomol Zool 33:281–288
Acknowledgments
We are grateful to Dr. T. Hirowatari of the Entomological Laboratory of Kyushu University, Dr. M. Yago of The University Museum, The University of Tokyo, and Dr. H. Tsumuki, former Professor of Research Institute for Bioresources, Okayama University, for their kind advice and assistance, Mr. S. Morichi and Mr. S. Minohara of the Lepidopterological Society of Japan for collecting samples, and our laboratory colleagues for their cooperation and helpful comments. We also would like to express our sincere thanks to the members of New Kansai International Airport Company, Ltd., for giving us opportunities to conduct the survey around the airport. This study was supported in part by the Japan Society for the Promotion of Science (Grant-in-Aid for JSPS Fellows 11J10420 and Grant-in-Aid for Scientific Research 15K06934, 24510331, and 23510297).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakamoto, Y., Hirai, N. & Ishii, M. Effects of photoperiod and temperature on the development and diapause of the endangered butterfly Zizina emelina (Lepidoptera: Lycaenidae). J Insect Conserv 19, 639–645 (2015). https://doi.org/10.1007/s10841-015-9786-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-015-9786-1