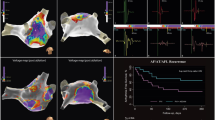
Abstract
Background
There are no standard mapping approaches for patients with persistent atrial fibrillation (PeAF), particularly after failed prior catheter ablation (CA). In this study, we assess the feasibility of using Electrogram Morphology Recurrence (EMR) to guide ablation.
Methods
Ten patients with recurrent PeAF after prior CA underwent detailed mapping of both atria during PeAF using the PentaRay (4 mm interelectrode spacing) and 3D mapping with CARTO. At each site, 15 s recordings were made. Custom software identified each electrogram and cross-correlation was used to identify the most recurrent electrogram morphology from which the % recurrence and cycle length of the most repeatable morphology (CLR) was calculated. Sites of shortest CLR and sites within 5 ms of shortest CLR with recurrence ≥ 80% were used to inform CA strategy.
Results
A mean of 342.9 ± 131.9 LA and 328.6 ± 91.5 RA sites were recorded per patient. Nine had PV reconnection. Shortest CLR sites guided ablation in 6/10 patients while 1 patient failed to fulfill shortest CLR criteria, and another 3 did not undergo CA guided by shortest CLR due to operator preference. On 12-month follow-up, all 4 patients without shortest CLR guided CA had recurrent PeAF. Of the 6 patients with shortest CLR guided CA, 5 patients did not have recurrent PeAF (p = 0.048), although 1 had paroxysmal AF and 2 had atypical atrial flutter.
Conclusion
EMR is a feasible, novel technique to guide CA in patients with PeAF. Further evaluation is needed to provide an electrogram-based method for mapping guided targeted ablation of key areas.



Similar content being viewed by others
Data Availability
The Data will not be made available. Any inquires regarding the data can be submitted to the corresponding author and it will be addressed accordingly.
Abbreviations
- AF:
-
Atrial fibrillation
- CA:
-
Catheter ablation
- CL:
-
Cycle length
- CLR :
-
Cycle length of the most recurrent morphology
- DF:
-
Dominant frequency
- EMR:
-
Electrogram morphology recurrence
- LA:
-
Left atrium
- PeAF:
-
Persistent atrial fibrillation
- PVI:
-
Pulmonary vein isolation
- RA:
-
Right atrium
- Rec%:
-
Frequency of most recurrent morphology
References
Oral H, Knight BP, Tada H, et al. Pulmonary vein isolation for paroxysmal and persistent atrial fibrillation. Circulation. 2002;105(9):1077–81. https://doi.org/10.1161/HC0902.104712.
Calkins H, Hindricks G, Cappato R, et al. 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation. Heart Rhythm. 2017;14(10):e275–444. https://doi.org/10.1016/j.hrthm.2017.05.012.
Marrouche NF, Wazni O, McGann C, et al. Effect of MRI-Guided Fibrosis Ablation vs Conventional Catheter Ablation on Atrial Arrhythmia Recurrence in Patients With Persistent Atrial Fibrillation: The DECAAF II Randomized Clinical Trial. JAMA. 2022;327(23):2296–305. https://doi.org/10.1001/JAMA.2022.8831.
Weng W, Birnie DH, Ramirez FD, et al. Outcomes of a comprehensive strategy during repeat atrial fibrillation ablation. J Interv Card Electrophysiol. 2022;65(2):391–9. https://doi.org/10.1007/S10840-022-01190-4.
Daimee UA, Akhtar T, Boyle TA, et al. Repeat catheter ablation for recurrent atrial fibrillation: Electrophysiologic findings and clinical outcomes. J Cardiovasc Electrophysiol. 2021;32(3):628–38. https://doi.org/10.1111/JCE.14867.
Tahir KS, Mounsey JP, Hummel JP. Posterior Wall Isolation in Atrial Fibrillation Ablation. J Innov Card Rhythm Manag. 2018;9(6):3186–94. https://doi.org/10.19102/ICRM.2018.090602.
Alturki A, Huynh T, Dawas A, et al. Left atrial appendage isolation in atrial fibrillation catheter ablation: A meta-analysis. J Arrhythm. 2018;34(5):478–84. https://doi.org/10.1002/JOA3.12095.
Goyal R, Gracia E, Fan R. The Role of Superior Vena Cava Isolation in the Management of Atrial Fibrillation. J Innov Card Rhythm Manag. 2017;8(4):2674. https://doi.org/10.19102/ICRM.2017.080406.
Berenfeld O. Quantifying activation frequency in atrial fibrillation to establish underlying mechanisms and ablation guidance. Heart Rhythm. 2007;4(9):1225–34. https://doi.org/10.1016/j.hrthm.2007.05.004.
Sanders P, Berenfeld O, Hocini M, et al. Spectral Analysis Identifies Sites of High-Frequency Activity Maintaining Atrial Fibrillation in Humans. Circulation. 2005;112(6):789–97. https://doi.org/10.1161/CIRCULATIONAHA.104.517011.
Nademanee K, McKenzie J, Kosar E, et al. A new approach for catheter ablation of atrial fibrillation: mapping of the electrophysiologic substrate. J Am Coll Cardiol. 2004;43(11):2044–53. https://doi.org/10.1016/J.JACC.2003.12.054.
Kong MH, Piccini JP, Bahnson TD. Efficacy of adjunctive ablation of complex fractionated atrial electrograms and pulmonary vein isolation for the treatment of atrial fibrillation: a meta-analysis of randomized controlled trials. Europace. 2011;13(2):193–204. https://doi.org/10.1093/EUROPACE/EUQ384.
Li WJ, Bai YY, Zhang HY, et al. Additional Ablation of Complex Fractionated Atrial Electrograms After Pulmonary Vein Isolation in Patients With Atrial Fibrillation. Circ Arrhythm Electrophysiol. 2011;4(2):143–8. https://doi.org/10.1161/CIRCEP.110.958405.
Narayan SM, Krummen DE, Shivkumar K, Clopton P, Rappel WJ, Miller JM. Treatment of atrial fibrillation by the ablation of localized sources: CONFIRM (Conventional Ablation for Atrial Fibrillation With or Without Focal Impulse and Rotor Modulation) trial. J Am Coll Cardiol. 2012;60(7):628–36. https://doi.org/10.1016/J.JACC.2012.05.022.
Narayan SM, Patel J, Mulpuru S, Krummen DE. Focal impulse and rotor modulation ablation of sustaining rotors abruptly terminates persistent atrial fibrillation to sinus rhythm with elimination on follow-up: A video case study. Heart Rhythm. 2012;9(9):1436–9. https://doi.org/10.1016/j.hrthm.2012.03.055.
Estner HL, Hessling G, Ndrepepa G, et al. Electrogram-guided substrate ablation with or without pulmonary vein isolation in patients with persistent atrial fibrillation. Europace. 2008;10(11):1281–7. https://doi.org/10.1093/EUROPACE/EUN244.
Oral H, Chugh A, Good E, et al. Radiofrequency Catheter Ablation of Chronic Atrial Fibrillation Guided by Complex Electrograms. Circulation. 2007;115(20):2606–12. https://doi.org/10.1161/CIRCULATIONAHA.107.691386.
Verma A, Jiang C yang, Betts TR, et al. Approaches to Catheter Ablation for Persistent Atrial Fibrillation. N Engl J Med. 2015;372(19):1812–22. https://doi.org/10.1056/NEJMOA1408288/SUPPL_FILE/NEJMOA1408288_DISCLOSURES.PDF.
Wu S, Li H, Yi S, Yao J, Chen X. Comparing the efficacy of catheter ablation strategies for persistent atrial fibrillation: a Bayesian analysis of randomized controlled trials. J Interv Card Electrophysiol. 2022;66:757–70. https://doi.org/10.1007/s10840-022-01246-5.
Ayzenberg O, Swissa M, Shlezinger T, et al. Atrial fibrillation ablation success rate - a retrospective multicenter study. Curr Probl Cardiol. 2022;48(8):101161. https://doi.org/10.1016/J.CPCARDIOL.2022.101161.
Gordon D, Goldberger JJ, Arora R, Aistrup GL, Ng J. Searching for “order” in atrial fibrillation using electrogram morphology recurrence plots. Comput Biol Med. 2015;65:220–8. https://doi.org/10.1016/J.COMPBIOMED.2015.07.018.
Zaatari G, Mitrani R, Bohorquez J, et al. Electrogram morphology recurrence to characterize drivers of persistent atrial fibrillation: initial versus redo catheter ablation. JACC Clin Electrophysiol. 2023;9(4):526–40. https://doi.org/10.1016/j.jacep.2022.11.003.
Ng J, Gordon D, Passman RS, Knight BP, Arora R, Goldberger JJ. Electrogram morphology recurrence patterns during atrial fibrillation. Heart Rhythm. 2014;11(11):2027–34. https://doi.org/10.1016/j.hrthm.2014.08.002.
Ng J, Sehgal V, Ng JK, Gordon D, Goldberger JJ. Iterative method to detect atrial activations and measure cycle length from electrograms during atrial fibrillation. IEEE Trans Biomed Eng. 2014;61(2):273–8. https://doi.org/10.1109/TBME.2013.2290003.
Rillig A, Tilz RR, Lin T, et al. Unexpectedly high incidence of stroke and left atrial appendage thrombus formation after electrical isolation of the left atrial appendage for the treatment of atrial tachyarrhythmias. Circ Arrhythm Electrophysiol. 2016;9(5):e003461. https://doi.org/10.1161/CIRCEP.115.003461/-/DC1.
Nanthakumar K, Plumb VJ, Epstein AE, Veenhuyzen GD, Link D, Kay GN. Resumption of Electrical Conduction in Previously Isolated Pulmonary Veins. Circulation. 2004;109(10):1226–9. https://doi.org/10.1161/01.CIR.0000121423.78120.49.
Gerstenfeld EP, Callans DJ, Dixit S, Zado E, Marchlinski FE. Incidence and Location of Focal Atrial Fibrillation Triggers in Patients Undergoing Repeat Pulmonary Vein Isolation. J Cardiovasc Electrophysiol. 2003;14(7):685–90. https://doi.org/10.1046/J.1540-8167.2003.03013.X.
Lin D, Santangeli P, Zado ES, et al. Electrophysiologic Findings and Long-Term Outcomes in Patients Undergoing Third or More Catheter Ablation Procedures for Atrial Fibrillation. J Cardiovasc Electrophysiol. 2015;26(4):371–7. https://doi.org/10.1111/JCE.12603.
Kuck KH, Albenque JP, Chun KRJ, et al. Repeat ablation for atrial fibrillation recurrence post cryoballoon or radiofrequency ablation in the FIRE and ICE trial. Circ Arrhythm Electrophysiol. 2019;12(6):e007247. https://doi.org/10.1161/CIRCEP.119.007247.
Mol D, Mulder MJ, Veenstra R, et al. Strategies for repeat ablation for atrial fibrillation: A multicentre comparison of nonpulmonary vein versus pulmonary vein target ablation. J Cardiovasc Electrophysiol. 2022;33(5):885. https://doi.org/10.1111/JCE.15441.
Yoo S, Rottmann M, Ng J, et al. Regions of highly recurrent electrogram morphology with low cycle length reflect substrate for atrial fibrillation. JACC Basic Transl Sci. 2022;8(1):68–4. https://doi.org/10.1016/J.JACBTS.2022.07.011.
Romero J, di Biase L, Mohanty S, et al. Long-Term Outcomes of Left Atrial Appendage Electrical Isolation in Patients With Nonparoxysmal Atrial Fibrillation: A Propensity Score-Matched Analysis. Circ Arrhythm Electrophysiol. 2020;13(11):e008390. https://doi.org/10.1161/CIRCEP.120.008390.
Funding
This study was funded by grants 1R01HL125881 and 1R41HL127907 from the National Heart, Lung, and Blood Institute, National Institutes of Health. Drs. Mitrani and Goldberger receive funding from the Miami Heart Research Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. The patients were enrolled under an investigational device exemption (IDE) protocol approved by the Food and Drug Administration (FDA), and the University of Miami Institutional Review Board.
Informed consent
All patients provided written informed consent.
Competing interests
Drs. Jason Ng and Jeffrey J. Goldberger are co-inventors of the patent for electrogram morphology recurrence analysis.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mitrani, R.D., Zaatari, G., Bohorquez, J. et al. Electrogram morphology recurrence guided catheter ablation for repeat ablation of persistent atrial fibrillation. J Interv Card Electrophysiol 67, 183–191 (2024). https://doi.org/10.1007/s10840-023-01594-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-023-01594-w