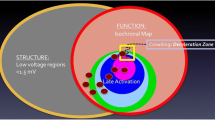
Abstract
Background
Reentrant atrial tachycardias (ATs) utilize critical isthmus (CI) for the maintenance of the circuit. The electrophysiological characteristics and clinical implications of the targeted CI regions of reentrant ATs during sinus rhythm (SR) were not clear. Therefore, our research aims at studying the electrical properties of the CI sites for scar-related reentrant ATs and the functional substrate mapping identified during SR.
Methods
Patients mapped with high-density catheters during SR and reentrant ATs were retrospectively analyzed. The CI regions of the reentrant ATs were confirmed by the combination of the activation map and the entrainment. The substrate mapping was analyzed for wavefront propagation, conduction velocity, and electrogram patterns.
Results
Twenty patients with 22 reentrant ATs that underwent high-density maps were analyzed at 2 hospitals. Mapping performed during SR identified a scar region of 23.0 ± 13.6% of the left atrium. Regions of the CI in SR were characterized by low voltage (0.3 ± 0.2 mV), conduction slowing (0.4 ± 0.2 m/s), and fractionated electrogram (duration 62.5 ± 13.9 ms). Substrate mapping during SR showed that the regions of the CI located with the low-voltage zone in 16 out of 22 CI (72.7%), the deceleration zone in 15 out of 22 CI (68.2%), and late atrial activation in 12 out of 22 CI (54.5%). Targeting regions of CI achieve 94% of termination or change of the reentrant circuit. At 6.2 ± 7.1 months, there was 75% freedom from atrial arrhythmia.
Conclusions
Novel high-density mapping can identify the functional substrates during SR and guide ablation. Low-voltage areas with conduction slowing are putative predictors of the CI for the maintenance of the reentrant ATs.




Similar content being viewed by others
Data availability
Data are available on reasonable request to the corresponding author.
References
Haïssaguerre M, Hocini M, Sanders P, Sacher F, Rotter M, Takahashi Y, et al. Catheter ablation of long-lasting persistent atrial fibrillation: clinical outcome and mechanisms of subsequent arrhythmias. J Cardiovasc Electrophysiol. 2005;16:1138–47. https://doi.org/10.1111/j.1540-8167.2005.00308.x.
Kobza R, Hindricks G, Tanner H, Schirdewahn P, Dorszewski A, Piorkowski C, et al. Late recurrent arrhythmias after ablation of atrial fibrillation: incidence, mechanisms, and treatment. Heart Rhythm. 2004;1:676–83. https://doi.org/10.1016/j.hrthm.2004.08.009.
Wasmer K, Mönnig G, Bittner A, Dechering D, Zellerhoff S, Milberg P, et al. Incidence, characteristics, and outcome of left atrial tachycardias after circumferential antral ablation of atrial fibrillation. Heart Rhythm. 2012;9:1660–6. https://doi.org/10.1016/j.hrthm.2012.06.007.
Frontera A, Mahajan R, Dallet C, Vlachos K, Kitamura T, Takigawa M, et al. Characterizing localized reentry with high-resolution mapping: evidence for multiple slow conducting isthmuses within the circuit. Heart Rhythm. 2019;16:679–85. https://doi.org/10.1016/j.hrthm.2018.11.027.
Vicera JJB, Lin YJ, Lee PT, Chang SL, Lo LW, Hu YF, et al. Identification of critical isthmus using coherent mapping in patients with scar-related atrial tachycardia. J Cardiovasc Electrophysiol. 2020;31:1436–47. https://doi.org/10.1111/jce.14457.
Laţcu DG, Bun SS, Viera F, Delassi T, El Jamili M, Al Amoura A, Saoudi N, et al. Selection of critical isthmus in scar-related atrial tachycardia using a new automated ultrahigh resolution mapping system. Circ Arrhythm Electrophysiol. 2017;10:e004510. https://doi.org/10.1161/CIRCEP.116.004510.
Jaïs P, Matsuo S, Knecht S, Weerasooriya R, Hocini M, Sacher F, et al. A deductive mapping strategy for atrial tachycardia following atrial fibrillation ablation: importance of localized reentry. J Cardiovasc Electrophysiol. 2009;20:480–91. https://doi.org/10.1111/j.1540-8167.2008.01373.x.
Luther V, Sikkel M, Bennett N, Guerrero F, Leong K, Qureshi N, et al. Visualizing localized reentry with ultra-high density mapping in iatrogenic atrial tachycardia: beware pseudo-reentry. Circ Arrhythm Electrophysiol. 2017;10:e004724. https://doi.org/10.1161/CIRCEP.116.004724.
Aziz Z, Shatz D, Raiman M, Upadhyay GA, Beaser AD, Besser SA, et al. Targeted ablation of ventricular tachycardia guided by wavefront discontinuities During sinus rhythm: a new functional substrate mapping strategy. Circulation. 2019;140:1383–97. https://doi.org/10.1161/CIRCULATIONAHA.119.042423.
Raiman M, Tung R. Automated isochronal late activation mapping to identify deceleration zones: rationale and methodology of a practical electroanatomic mapping approach for ventricular tachycardia ablation. Comput Biol Med. 2018;102:336–40. https://doi.org/10.1016/j.compbiomed.2018.07.012.
Anter E, Kleber AG, Rottmann M, Leshem E, Barkagan M, Tschabrunn CM, et al. Infarct-related ventricular tachycardia: redefining the electrophysiological substrate of the isthmus During sinus rhythm. JACC Clin Electrophysiol. 2018;4:1033–48. https://doi.org/10.1016/j.jacep.2018.04.007.
De Ponti R, Marazzi R, Zoli L, Caravati F, Ghiringhelli S, Salerno-Uriarte JA. Electroanatomic mapping and ablation of macroreentrant atrial tachycardia: comparison between successfully and unsuccessfully treated cases. J Cardiovasc Electrophysiol. 2010;21:155–62. https://doi.org/10.1111/j.1540-8167.2009.01602.x.
Pascale P, Shah AJ, Roten L, Scherr D, Komatsu Y, Jadidi AS, et al. Pattern and timing of the coronary sinus activation to guide rapid diagnosis of atrial tachycardia after atrial fibrillation ablation. Circ Arrhythm Electrophysiol. 2013;6:481–90. https://doi.org/10.1161/CIRCEP.113.000182.
Rostock T, Drewitz I, Steven D, Hoffmann BA, Salukhe TV, Bock K, et al. Characterization, mapping, and catheter ablation of recurrent atrial tachycardias after stepwise ablation of long-lasting persistent atrial fibrillation. Circ Arrhythm Electrophysiol. 2010;3:160–9. https://doi.org/10.1161/CIRCEP.109.899021.
Chang SL, Tsao HM, Lin YJ, Lo LW, Hu YF, Tuan TC, et al. Differentiating macroreentrant from focal atrial tachycardias occurred after circumferential pulmonary vein isolation. J Cardiovasc Electrophysiol. 2011;22:748–55. https://doi.org/10.1111/j.1540-8167.2010.02002.x.
Takigawa M, Derval N, Frontera A, Martin R, Yamashita S, Cheniti G, et al. Revisiting anatomic macroreentrant tachycardia after atrial fibrillation ablation using ultrahigh-resolution mapping: implications for ablation. Heart Rhythm. 2018;15:326–33. https://doi.org/10.1016/j.hrthm.2017.10.029.
Hu YF, Higa S, Huang JL, Tai CT, Lin YJ, Chang SL, et al. Electrophysiologic characteristics and catheter ablation of focal atrial tachycardia with more than one focus. Heart Rhythm. 2009;6:198–203. https://doi.org/10.1016/j.hrthm.2008.10.041.
Higa S, Tai CT, Lin YJ, Liu TY, Lee PC, Huang JL, et al. Focal atrial tachycardia: new insight from noncontact mapping and catheter ablation. Circulation. 2004;109:84–91. https://doi.org/10.1161/01.CIR.0000109481.73788.2E.
Ciaccio EJ, Ashikaga H, Coromilas J, Hopenfeld B, Cervantes DO, Wit AL, et al. Model of bipolar electrogram fractionation and conduction block associated with activation wavefront direction at infarct border zone lateral isthmus boundaries. Circ Arrhythm Electrophysiol. 2014;7:152–63. https://doi.org/10.1161/CIRCEP.113.000840.
Spach MS, Miller WT 3rd, Miller-Jones E, Warren RB, Barr RC. Extracellular potentials related to intracellular action potentials during impulse conduction in anisotropic canine cardiac muscle. Circ Res. 1979;45:188–204. https://doi.org/10.1161/01.res.45.2.188.
Kitamura T, Takigawa M, Derval N, Denis A, Martin R, Vlachos K, et al. Atrial tachycardia circuits include low voltage area from index atrial fibrillation ablation relationship between RF ablation lesion and AT. J Cardiovasc Electrophysiol. 2020;31:1640–8. https://doi.org/10.1111/jce.14576.
Laţcu DG, Bun SS, Casado Arroyo R, Wedn AM, Benaich FA, Hasni K, et al. Scar identification, quantification, and characterization in complex atrial tachycardia: a path to targeted ablation? Europace. 2019;21:i21–6. https://doi.org/10.1093/europace/euy182.
Valbom Mesquita D, Parreira L, Farinha J, Amador P, Esteves A, Fonseca M, et al. A new approach to atrial flutter ablation using functional substrate mapping with wavefront discontinuity during sinus rhythm. EP Europace. 2021:23.
Christopher E, Woods AAS, Nayak H, Stevens B, Kwasnik A, Shatz N, et al. Correlation between sinus rhythm deceleration zones and critical sites for localized reentrant atrial flutter: a retrospective multicenter analysis. Heart Rhythm. 2022;O2(29):279–87. https://doi.org/10.1016/j.hroo.2022.03.003.
Jadidi AS, Lehrmann H, Keyl C, Sorrel J, Markstein V, Minners J, et al. Ablation of persistent atrial fibrillation targeting low-voltage areas with selective activation characteristics. Circ Arrhythm Electrophysiol. 2016;9:e002962. https://doi.org/10.1161/CIRCEP.115.002962.
Kottkamp H, Berg J, Bender R, Rieger A, Schreiber D. Box isolation of fibrotic areas (BIFA): a patient-tailored substrate modification approach for ablation of atrial fibrillation. J Cardiovasc Electrophysiol. 2016;27:22–30. https://doi.org/10.1111/jce.12870.
Lo LW, Lin YJ, Chang SL, Hu YF, Chung FP, Chen SA. Pearls and pitfalls in catheter ablation of persistent atrial fibrillation. Circ J. 2016;80:306–13. https://doi.org/10.1253/circj.CJ-15-1366.
Author information
Authors and Affiliations
Contributions
Wen-Chin Tsai: concept, data analysis and collection, drafting the article; Shih-Lin Chang, Li-Wei Lo, Yu-Feng Hu, Tze-Fan Chao, Fa-Po Chung, Jo-Nan Liao, Ting-Yung Chang, Ta-Chuan Tuan, Ling Kuo, Cheng-I Wu, Chih-Min Liu, Shin-Huei Liu, Ming-Ren Kuo, MD: concept/design, clinical data collection and approval of the article; Yenn-Jiang Lin and Shih-Ann Chen: critical revision of the article, approval of the article.
Corresponding authors
Ethics declarations
Informed consent
Written informed consent was obtained from all patients and the study was approved by the institutional research committee.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tsai, WC., Lin, YJ., Chang, SL. et al. High-density characterization of the sinus rhythm: a new functional substrate map of scar-related atrial tachycardia. J Interv Card Electrophysiol 66, 1631–1639 (2023). https://doi.org/10.1007/s10840-023-01480-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-023-01480-5