Abstract
Purpose
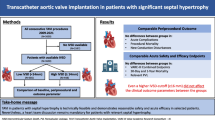
We aimed to study the characteristics of the membranous septum (MS) and its relationship with the aortic valve (AoV) and aortic annulus (AA) in patients who required PPM post-TAVR.
Methods
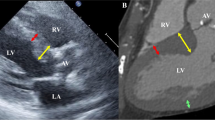
We performed a retrospective case–control study of 144 patients undergoing TAVR from 2016 to 2018. Thirty-four patients, requiring PPM implantation, were compared with 34 matched controls who did not require pacing. The total MS length, supra-annular MS (SA-MS) length, infra-annular MS (IA-MS) length, angle between the plane of the AA and MS (AA-MS), and degree of AoV calcifications (AVC) were obtained from preoperative CT. AoV prosthesis implantation depth was obtained from intra-operative fluoroscopy.
Results
There were no significant differences in valve type (self-expandable: 23 cases vs 25 controls, and balloon-expandable: 11 vs 9, p = 0.79), degree of AVC (0.65 cm3 vs 0.82 cm3, p = 0.62), or implantation depth (7.76 mm vs 7.28 mm, p = 0.83). Compared to controls, there was no difference in total MS length (6.68 mm vs 6.06 mm, p = 0.97), but the IA-MS was significantly shorter (3.64 mm vs 4.56 mm, p = 0.02) and the SA-MS was significantly longer (2.73 mm vs 1.67 mm, p = 0.02) in patients requiring PPM. Patients requiring PPM also had a larger AA-MS angle (103.5° vs 96.7°, p = 0.01).
Conclusion
The position of the MS with respect to the AA and MS distance below the annular plane were more closely associated with post-TAVR conduction abnormalities requiring PPM than the absolute length of the MS. Patients undergoing TAVR with such anatomy have a higher risk of requiring PPM and should be monitored for developing these complications.






Similar content being viewed by others
Availability of data and material
Raw data were generated at University of Maryland Medical Center. Derived data supporting findings of this study are available from the corresponding author (JN) on request.
This manuscript is not under consideration for publication elsewhere, its publication is approved by all authors and tacitly or explicitly by the responsible authorities where the work was carried out, and that, if accepted, it will not be published elsewhere in the same form, in English or in any other language, including electronically without the written consent of the copyright-holder. All authors have read and approved of the manuscript. No funding was required for this study.
References
Cribier A. The development of transcatheter aortic valve replacement (TAVR). Glob Cardiol Sci Pract. 2016 Dec 30;2016(4):e201632. https://doi.org/10.21542/gcsp.2016.32.
Spears J, Al-Saiegh Y, Goldberg D, Manthey S, Goldberg S. TAVR: a review of current practices and considerations in low-risk patients. J Interv Cardiol. 2020;2020:2582938. https://doi.org/10.1155/2020/2582938.
Baan J, Yong ZY, Koch KT, Henriques JP, Bouma BJ, Vis MM, Cocchieri R, Piek JJ, de Mol BA. Factors associated with cardiac conduction disorders and permanent pacemaker implantation after percutaneous aortic valve implantation with the CoreValve prosthesis. Am Heart J. 2010;159(3):497–503. https://doi.org/10.1016/j.ahj.2009.12.009.
Grover FL, Vemulapalli S, Carroll JD, Edwards FH, Mack MJ, Thourani VH, Brindis RG, Shahian DM, Ruiz CE, Jacobs JP, Hanzel G, Bavaria JE, Tuzcu EM, Peterson ED, Fitzgerald S, Kourtis M, Michaels J, Christensen B, Seward WF, Hewitt K, Holmes DR. 2016 annual report of the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry. J Am Coll Cardiol. 2017;69(10):1215–30. https://doi.org/10.1016/j.jacc.2016.11.033.
Popma JJ, Deeb GM, Yakubov SJ, et al. Transcatheter aortic-valve replacement with a self-expanding valve in low-risk patients. N Engl J Med. 2019;380(18):1706–15. https://doi.org/10.1056/NEJMoa1816885.
Cabrera JÁ, Anderson RH, Macías Y, Nevado-Medina J, Porta-Sánchez A, Rubio JM, Sánchez-Quintana D. Variable arrangement of the atrioventricular conduction axis within the triangle of Koch: implications for permanent his bundle pacing. JACC Clin Electrophysiol. 2020;6(4):362–77. https://doi.org/10.1016/j.jacep.2019.12.004.
Hamdan A, Guetta V, Klempfner R, Konen E, Raanani E, Glikson M, Goitein O, Segev A, Barbash I, Fefer P, Spiegelstein D, Goldenberg I, Schwammenthal E. Inverse relationship between membranous septal length and the risk of atrioventricular block in patients undergoing transcatheter aortic valve implantation. JACC Cardiovasc Interv. 2015;8(9):1218–28. https://doi.org/10.1016/j.jcin.2015.05.010.
Piazza N, de Jaegere P, Schultz C, Becker AE, Serruys PW, Anderson RH. Anatomy of the aortic valvar complex and its implications for transcatheter implantation of the aortic valve. Circ Cardiovasc Interv. 2008;1(1):74–81. https://doi.org/10.1161/CIRCINTERVENTIONS.108.780858.
Achenbach S, Delgado V, Hausleiter J, Schoenhagen P, Min JK, Leipsic JA. SCCT expert consensus document on computed tomography imaging before transcatheter aortic valve implantation (TAVI)/transcatheter aortic valve replacement (TAVR). J Cardiovasc Comput Tomogr. 2012;6(6):366–80. https://doi.org/10.1016/j.jcct.2012.11.002.
Upadhyay GA. The variable arrangement of the atrioventricular conduction axis within the triangle of Koch: millimeters matter. JACC Clin Electrophysiol. 2020;6(4):378–81. https://doi.org/10.1016/j.jacep.2020.01.007.
Auffret V, Puri R, Urena M, Chamandi C, Rodriguez-Gabella T, Philippon F, Rodés-Cabau J. Conduction disturbances after transcatheter aortic valve replacement: current status and future perspectives. Circulation. 2017;136(11):1049–69. https://doi.org/10.1161/CIRCULATIONAHA.117.028352.
Moreno R, Dobarro D, López de Sá E, Prieto M, Morales C, Calvo Orbe L, Moreno-Gomez I, Filgueiras D, Sanchez-Recalde A, Galeote G, Jiménez-Valero S, Lopez-Sendon JL. Cause of complete atrioventricular block after percutaneous aortic valve implantation: insights from a necropsy study. Circulation. 2009 Aug 4;120(5):e29-30. https://doi.org/10.1161/CIRCULATIONAHA.109.849281.
Abdel-Wahab M, Mehilli J, Frerker C, et al. Comparison of balloon-expandable vs self-expandable valves in patients undergoing transcatheter aortic valve replacement. JAMA. 2014;311(15):1503–14. https://doi.org/10.1001/jama.2014.3316.
Nazif TM, Dizon JM, Hahn RT, Xu K, Babaliaros V, Douglas PS, El-Chami MF, Herrmann HC, Mack M, Makkar RR, Miller DC, Pichard A, Tuzcu EM, Szeto WY, Webb JG, Moses JW, Smith CR, Williams MR, Leon MB, Kodali SK; PARTNER Publications Office. Predictors and clinical outcomes of permanent pacemaker implantation after transcatheter aortic valve replacement: the PARTNER (Placement of AoRtic TraNscathetER Valves) trial and registry. JACC Cardiovasc Interv. 2015;8(1 Pt A):60–9. https://doi.org/10.1016/j.jcin.2014.07.022.
Yater WM, Cornell VH. Heart block due to calcareous lesions of the bundle of His: review and report of a case with detailed histopathologic study. Ann Intern Med. 1935;8:777–89.
Khawaja MZ, Rajani R, Cook A, Khavandi A, Moynagh A, Chowdhary S, Spence MS, Brown S, Khan SQ, Walker N, Trivedi U, Hutchinson N, De Belder AJ, Moat N, Blackman DJ, Levy RD, Manoharan G, Roberts D, Khogali SS, Crean P, Brecker SJ, Baumbach A, Mullen M, Laborde JC, Hildick-Smith D. Permanent pacemaker insertion after CoreValve transcatheter aortic valve implantation: incidence and contributing factors (the UK CoreValve Collaborative). Circulation. 2011;123(9):951–60. https://doi.org/10.1161/CIRCULATIONAHA.109.927152.
Tarantini G, Mojoli M, Purita P, Napodano M, D’Onofrio A, Frigo A, Covolo E, Facchin M, Isabella G, Gerosa G, Iliceto S. Unravelling the (arte)fact of increased pacemaker rate with the Edwards SAPIEN 3 valve. EuroIntervention : journal of EuroPCR in collaboration with the Working Group on Interventional Cardiology of the European Society of Cardiology. 2015;11(3):343–50. https://doi.org/10.4244/EIJY14M11_06.
Bonnet G, Pernot M, Zaouter C, Peltan J, Seguy B, Klotz N, et al. Membranous septal length and valve IMPLANTATION depth OF TAVR: Predictors of new permanent Pacemaker implantation AFTER TAVR. Archives of Cardiovascular Diseases Supplements. 2019;11(1):70–1.
Rodés-Cabau J. Optimizing valve implantation depth to win the battle against conduction disturbances post-TAVR. JACC Cardiovasc Interv. 2019;12(18):1808–10. https://doi.org/10.1016/j.jcin.2019.06.039.
Kusumoto FM, Schoenfeld MH, Barrett C, Edgerton JR, Ellenbogen KA, Gold MR, Goldschlager NF, Hamilton RM, Joglar JA, Kim RJ, Lee R, Marine JE, McLeod CJ, Oken KR, Patton KK, Pellegrini CN, Selzman KA, Thompson A, Varosy PD. 2018 ACC/AHA/HRS guideline on the evaluation and management of patients with bradycardia and cardiac conduction delay: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2019;74(7):e51–156. https://doi.org/10.1016/j.jacc.2018.10.044.
Tovia-Brodie O, Michowitz Y, Belhassen B. Use of electrophysiological studies in transcatheter aortic valve implantation. Arrhythm Electrophysiol Rev. 2020;9(1):20–7. https://doi.org/10.15420/aer.2019.38.3.
Rivard L, Schram G, Asgar A, et al. Electrocardiographic and electrophysiological predictors of atrioventricular block after transcatheter aortic valve replacement. Heart Rhythm. 2015;12:321–9. https://doi.org/10.1016/j.hrthm.2014.10.023.
Maeno Y, Abramowitz Y, Kawamori H, Kazuno Y, Kubo S, Takahashi N, Mangat G, Okuyama K, Kashif M, Chakravarty T, Nakamura M, Cheng W, Friedman J, Berman D, Makkar RR, Jilaihawi H. A highly predictive risk model for pacemaker implantation after TAVR. JACC Cardiovasc Imaging. 2017;10(10 Pt A):1139–47. https://doi.org/10.1016/j.jcmg.2016.11.020.
Makki N, Dollery J, Jones D, Crestanello J, Lilly S. Conduction disturbances after TAVR: Electrophysiological studies and pacemaker dependency. Cardiovasc Revasc Med. 2017;18(5S1):S10–3. https://doi.org/10.1016/j.carrev.2017.03.009.
Badenco N, Chong-Nguyen C, Maupain C, et al. Respective role of surface electrocardiogram and His bundle recordings to assess the risk of atrioventricular block after transcatheter aortic valve replacement. Int J Cardiol. 2017;236:216–20. https://doi.org/10.1016/j.ijcard.2017.02.029.
Kostopoulou A, Karyofillis P, Livanis E, et al. Permanent pacing after transcatheter aortic valve implantation of a CoreValve prosthesis as determined by electrocardiographic and electrophysiological predictors: a single-centre experience. Europace. 2016;18:131–7. https://doi.org/10.1093/europace/euv137.
Acknowledgements
All authors made a substantial contribution to the study. The study was designed by Dr. Jonathan Na, Bryan Nixon, Dr. Jean Jeudy, and Dr. Alejandro Jimenez Restrepo. Radiographic CT data was analyzed by Bryan Nixon and Dr. Jean Jeudy. Demographic data was gathered by Dr. Jonathan Na, Dr. Paul Han, Dr. James Childress, Dr. Gregory Norcross, and Dr. Steven Liskov. Dr. Na, Bryan Nixon, and Dr. Jimenez Restrepo contributed to analysis and interpretation of data. Article was drafted by Dr. Na and Bryan Nixon and revisions were provided by Dr. Jimenez Restrepo. Dr. Jimenez Restrepo provided study oversight and final approval of article submitted. All authors have read the final article and have approved it for submission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Na, J., Nixon, B., Childress, J. et al. Anatomical characteristics of the membranous septum are predictive of pacemaker requirement in patients undergoing transcatheter aortic valve replacement. J Interv Card Electrophysiol 63, 449–459 (2022). https://doi.org/10.1007/s10840-021-01041-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-021-01041-8