Abstract
Purpose
Previous clinical studies have shown that ranolazine (RAN) added to amiodarone (AMIO) might accelerate the termination of recent-onset atrial fibrillation. This study was undertaken to delineate possible mechanisms that contribute to the enhancement of the antiarrhythmic efficacy of RAN-AMIO coadministration.
Methods
Ten rabbits were anesthetized and two monophasic action potential (MAP) catheters were sequentially inserted into the right atrium. One MAP electrode was used to pace and record; the other electrode was used only for recording MAP from an adjacent atrial region. Intraatrial conduction time (IACT), 2:1 intraatrial conduction block (IACB), and atrial post-repolarization refractoriness (aPRR) were consecutively determined by high-rate atrial burst pacing and programmed stimulation, respectively. All parameters were evaluated during baseline and following AMIO (3 mg/kg iv) or AMIO+RAN (2.4 mg/kg iv bolus +0.134 mg/kg/min maintenance infusion).
Results
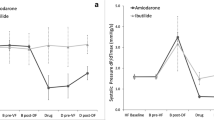
The IACT remained unchanged post AMIO compared with baseline (37.6 ± 3.8 vs 36.4 ± 2.4 ms), whereas the addition of RAN to AMIO significantly prolonged IACT (50.4 ± 3.6 ms, p < .001). The pacing cycle length producing 2:1 IACB was 101.2 ± 21.7 ms at baseline , 117.5 ± 15 ms after AMIO (p = 0.265), and 150 ± 14 ms after AMIO+RAN (p < .001). Baseline aPRR was longer following AMIO treatment (35 ± 5 vs 50 ± 9 ms, p < .01) but remarkably prolonged with RAN supplementation (105 ± 11 ms, p < .001).
Conclusions
RAN significantly prolonged the propagation time of rapid atrial depolarizations and potentiated the AMIO-induced moderate increases in aPRR. These mechanisms possibly contribute to the earlier termination of atrial fibrillation when RAN is co-administered with AMIO.







Similar content being viewed by others
References
Fragakis N, Koskinas KC, Katritsis DG, Pagourelias ED, Zografos T, Geleris P. Comparison of effectiveness of ranolazine plus amiodarone versus amiodarone alone for conversion of recent-onset atrial fibrillation. Am J Cardiol. 2012;110:673–7.
Koskinas KC, Fragakis N, Katritsis D, Skeberis V, Vassilikos V. Ranolazine enhances the efficacy of amiodarone for conversion of recent-onset atrial fibrillation. Europace. 2014;16:973–9.
Tsanaxidis N, Aidonidis I, Hatziefthimiou A, Daskalopoulou SS, Giamouzis G, Triposkiadis F, et al. Ranolazine added to amiodarone facilitates earlier conversion of atrial fibrillation compared to amiodarone-only therapy. Pacing Clin Electrophysiol. 2017;40:372–8.
Aidonidis I, Doulas K, Hatziefthimiou A, Tagarakis G, Simopoulos V, Rizos I, et al. Ranolazine-induced postrepolarization refractoriness suppresses induction of atrial flutter and fibrillation in anesthetized rabbits. J Cardiovasc Pharmacol Ther. 2013;18:94–101.
Bhimani AA, Yasuda T, Sadrpour SA, Khrestian CM, Lee S, Zeng D, et al. Ranolazine terminates atrial flutter and fibrillation in a canine model. Heart Rhythm. 2014;11:1592–9.
Antzelevitch C, Burashnikov A. Atrial-selective sodium channel block as a novel strategy for the management of atrial fibrillation. J Electrocardiol. 2009;42:543–8.
Camm AJ, Capucci A, Hohnloser SH, Torp-Pedersen C, Van Gelder IC, Mangal B, et al. A randomized active-controlled study comparing the efficacy and safety of vernakalant to amiodarone in recent-onset atrial fibrillation. J Am Coll Cardiol. 2011;57:313–21.
Suzuki T, Morishima M, Kato S, Ueda N, Honjo H, Kamiya K. Atrial selectivity in Na + channel blockade by acute amiodarone. Cardiovasc Res. 2013;98:136–44.
Koller BS, Karasik PE, Solomon AJ, Franz MR. Prolongation of conduction time during premature stimulation in the human atrium is primarily caused by local stimulus response latency. Eur Heart J. 1995;16:1920–4.
Li Z, Hertervig E, Yuan S, Yang Y, Lin Z, Olsson SB. Dispersion of atrial repolarization in patients with paroxysmal atrial fibrillation. Europace. 2001;3:285–91.
Simopoulos V, Tagarakis GI, Daskalopoulou SS, Daskalopoulos ME, Lenos A, Chryssagis K, et al. Ranolazine enhances the antiarrhythmic activity of amiodarone by accelerating conversion of new-onset atrial fibrillation after cardiac surgery. Angiology. 2014;65:294–7.
Simopoulos V, Hevas A, Hatziefthimiou A, Dipla K, Skoularigis I, Tsilimingas N, et al. Amiodarone plus ranolazine for conversion of post-cardiac surgery atrial fibrillation: enhanced effectiveness in reduced versus preserved ejection fraction patients. Cardiovasc Drugs Ther. 2018;32:559–65.
Frommeyer G, Schmidt M, Clauß C, Kaese S, Stypmann J, Pott C, et al. Further insights into the underlying electrophysiological mechanisms for reduction of atrial fibrillation by ranolazine in an experimental model of chronic heart failure. Eur J Heart Fail. 2012;14:1322–31.
Black-Maier EW, Pokorney SD, Barnett AS, Liu P, Shrader P, Ng J, et al. Ranolazine reduces atrial fibrillatory wave frequency. Europace. 2017;19:1096–100.
Ramirez RJ, Takemoto Y, Martins RP, Filgueiras-Rama D, Ennis SR, Mironov S, et al. Mechanisms by which ranolazine terminates paroxysmal but not persistent atrial fibrillation. Circ Arrhythm Electrophysiol. 2019;12(10):e005557.
Milberg P, Frommeyer G, Ghezelbash S, Rajamani S, Osada N, Razvan R, et al. Sodium channel block by ranolazine in an experimental model of stretch-related atrial fibrillation: prolongation of interatrial conduction time and increase in post-repolarization refractoriness. Europace. 2013;15:761–9.
Kirchhof P, Degen H, Franz MR, Eckardt L, Fabritz L, Milberg P, et al. Amiodarone-induced postrepolarization refractoriness suppresses induction of ventricular fibrillation. J Pharmacol Exp Ther. 2003;305:257–63.
Franz MR. Current status of monophasic action potential recording: theories, measurements and interpretations. Cardiovasc Res. 1999;41:25–40.
Burashnikov A, Belardinelli L, Antzelevitch C. Atrial-selective sodium channel block strategy to suppress atrial fibrillation: ranolazine versus propafenone. J Pharmacol Exp Ther. 2012;340:161–8.
Acknowledgments
We thank I. Makantasis for technical assistance during the experiments, and Dr Vet. Z. Arsenopoulou for her guidance regarding health status and anesthesia procedures of the animals.
Author information
Authors and Affiliations
Contributions
AI performed the conception, design, experimental preparation, and electrophysiologic data measurement. SV, SS, and SR performed the experimental preparation and data collection. DK, XA, and MP-A performed the statistical data analysis and English editorial of the manuscript.
Corresponding author
Ethics declarations
All experimental protocol procedures were approved by the Institutional Animal Care Committee. The study was conducted in accordance with the guidelines of the European Council on Animal Care.
Conflict of interests
The authors declare that they have no conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aidonidis, I., Simopoulos, V., Stravela, S. et al. Ranolazine depresses conduction of rapid atrial depolarizations in a beating rabbit heart model. J Interv Card Electrophysiol 62, 153–159 (2021). https://doi.org/10.1007/s10840-020-00865-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-020-00865-0