Abstract
Background
Direct oral anticoagulants (DOACs) are contraindicated in patients with atrial fibrillation (AF) and mechanical cardiac valves. However, safety and efficacy are controversial in patients with biological cardiac valves.
Objective
We report the safety and feasibility of periprocedural and long-term treatment with DOACs in patients with biological valves undergoing ablation for AF.
Methods
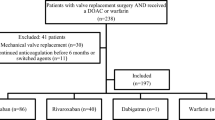
A total of 127 patients with AF and biological cardiac valve undergoing CA on uninterrupted DOAC were matched by gender and age with 127 patients with AF and biological cardiac valves undergoing CA on uninterrupted warfarin. All patients were anticoagulated for at least 3–4 weeks prior to ablation with either rivaroxaban (70%) or apixaban (30%), which were continued for at least 3 months and subsequently based on CHA2DS2-VASc score.
Results
Mean age of the study population was 63.0 ± 10.9 with 66% being male. The majority of patients on NOACs had aortic valve replacement (59%), while mitral valve was replaced in 41% of patients, which did not differ from the matched cohort on coumadin (aortic valve 57% and mitral valve 43%, (p = 0.8) (p = 0.8), respectively). The CHADS2 score was ≥ 2 in 90 patients (71.0%) on DOAC and 86 patients in (68%) the control (p = 0.6) group. Patients underwent ablation predominantly with uninterrupted rivaroxaban [89 (70%)], while the remaining 38 patients (30%) underwent ablation while on apixaban. Two groin hematomas were observed periprocedurally in both groups. No stroke/transient ischemic attack (TIA) was observed both periprocedurally and at long-term follow-up in either group.
Conclusion
Periprocedural and long-term administration of DOACs in patients with biological cardiac valves undergoing AF ablation appears as safe as warfarin therapy.


Similar content being viewed by others
References
January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC Jr, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association task force on practice guidelines and the Heart Rhythm Society. Circulation. 2014;130(23):e199–267. https://doi.org/10.1161/CIR.0000000000000041.
Benjamin EJ, Wolf PA, D'Agostino RB, Silbershatz H, Kannel WB, Levy D. Impact of atrial fibrillation on the risk of death: the Framingham heart study. Circulation. 1998;98(10):946–52.
De Caterina R, Camm AJ. What is ‘valvular’ atrial fibrillation? A reappraisal. Eur Heart J. 2014;35(47):3328–35. https://doi.org/10.1093/eurheartj/ehu352.
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP 3rd, Guyton RA, et al. 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association task force on practice guidelines. J Am Coll Cardiol. 2014;63(22):e57–185. https://doi.org/10.1016/j.jacc.2014.02.536.
Granger CB, Alexander JH, McMurray JJ, Lopes RD, Hylek EM, Hanna M, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365(11):981–92. https://doi.org/10.1056/NEJMoa1107039.
Giugliano RP, Ruff CT, Braunwald E, Murphy SA, Wiviott SD, Halperin JL, et al. Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2013;369(22):2093–104. https://doi.org/10.1056/NEJMoa1310907.
Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361(12):1139–51. https://doi.org/10.1056/NEJMoa0905561.
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365(10):883–91. https://doi.org/10.1056/NEJMoa1009638.
Di Biase L, Burkhardt JD, Santangeli P, Mohanty P, Sanchez JE, Horton R, et al. Periprocedural stroke and bleeding complications in patients undergoing catheter ablation of atrial fibrillation with different anticoagulation management: results from the role of coumadin in preventing thromboembolism in atrial fibrillation (AF) patients undergoing catheter ablation (COMPARE) randomized trial. Circulation. 2014;129(25):2638–44. https://doi.org/10.1161/CIRCULATIONAHA.113.006426.
Lakkireddy D, Reddy YM, Di Biase L, Vallakati A, Mansour MC, Santangeli P, et al. Feasibility and safety of uninterrupted rivaroxaban for periprocedural anticoagulation in patients undergoing radiofrequency ablation for atrial fibrillation: results from a multicenter prospective registry. J Am Coll Cardiol. 2014;63(10):982–8. https://doi.org/10.1016/j.jacc.2013.11.039.
Di Biase L, Lakkireddy D, Trivedi C, Deneke T, Martinek M, Mohanty S, et al. Feasibility and safety of uninterrupted peri-procedural apixaban administration in patients undergoing radiofrequency catheter ablation for atrial fibrillation: results from a multicenter study. Heart Rhythm. 2015;12:1162–8. https://doi.org/10.1016/j.hrthm.2015.02.028.
Lakkireddy D, Reddy YM, Di Biase L, Vanga SR, Santangeli P, Swarup V, et al. Feasibility and safety of dabigatran versus warfarin for periprocedural anticoagulation in patients undergoing radiofrequency ablation for atrial fibrillation: results from a multicenter prospective registry. J Am Coll Cardiol. 2012;59(13):1168–74. https://doi.org/10.1016/j.jacc.2011.12.014.
Di Biase L, Burkhardt JD, Mohanty P, Sanchez J, Horton R, Gallinghouse GJ, et al. Periprocedural stroke and management of major bleeding complications in patients undergoing catheter ablation of atrial fibrillation: the impact of periprocedural therapeutic international normalized ratio. Circulation. 2010;121(23):2550–6. https://doi.org/10.1161/CIRCULATIONAHA.109.921320.
Di Biase L, Elayi CS, Fahmy TS, Martin DO, Ching CK, Barrett C, et al. Atrial fibrillation ablation strategies for paroxysmal patients: randomized comparison between different techniques. Circ Arrhythm Electrophysiol. 2009;2(2):113–9. https://doi.org/10.1161/CIRCEP.108.798447.
Di Biase L, Santangeli P, Natale A. How to ablate long-standing persistent atrial fibrillation? Curr Opin Cardiol. 2013;28(1):26–35. https://doi.org/10.1097/HCO.0b013e32835b59bb.
Di Biase L, Gaita F, Toso E, Santangeli P, Mohanty P, Rutledge N, et al. Does periprocedural anticoagulation management of atrial fibrillation affect the prevalence of silent thromboembolic lesion detected by diffusion cerebral magnetic resonance imaging in patients undergoing radiofrequency atrial fibrillation ablation with open irrigated catheters? Results from a prospective multicenter study. Heart Rhythm. 2014;11(5):791–8. https://doi.org/10.1016/j.hrthm.2014.03.003.
Di Biase L, Natale A. Apixaban is dear to me, but dearer still is warfarin. Pacing Clin Electrophysiol. 2015;38(2):153–4. https://doi.org/10.1111/pace.12570.
Whitlock RP, Sun JC, Fremes SE, Rubens FD, Teoh KH. Antithrombotic and thrombolytic therapy for valvular disease: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e576S–600S. https://doi.org/10.1378/chest.11-2305.
Romero J, Cerrud-Rodriguez RC, Diaz JC, Michaud GF, Taveras J, Alviz I, et al. Uninterrupted direct oral anticoagulants vs. uninterrupted vitamin K antagonists during catheter ablation of non-valvular atrial fibrillation: a systematic review and meta-analysis of randomized controlled trials. Europace. 2018;20(10):1612–20. https://doi.org/10.1093/europace/euy133.
Grunkemeier GL, Li HH, Naftel DC, Starr A, Rahimtoola SH. Long-term performance of heart valve prostheses. Curr Probl Cardiol. 2000;25(2):73–154.
January CT, Wann LS, Calkins H, Chen LY, Cigarroa JE, Cleveland JC Jr, et al. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2019;74:104–32. https://doi.org/10.1016/j.jacc.2019.01.011.
Kirchhof P, Haeusler KG, Blank B, De Bono J, Callans D, Elvan A, et al. Apixaban in patients at risk of stroke undergoing atrial fibrillation ablation. Eur Heart J. 2018;39:2942–55.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Di Biase is a consultant for Biosense Webster, Boston Scientific, Stereotaxis and Abbott Medical. Dr. Di Biase received speaker honoraria/travel support from Medtronic, Pfizer, Bristol Meyers Squibb and Biotronik. Dr. Natale is a consultant for Boston Scientific, Biosense Webster, St. Jude Medical, Biotronik and Medtronic. Dr. Burkhardt is a consultant for Biosense-Webster and Stereotaxis. The remaining authors have nothing to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Di Biase, L., Romero, J., Briceno, D. et al. Periprocedural and long-term safety and feasibility of direct oral anticoagulants in patients with biological valve undergoing radiofrequency catheter ablation for atrial fibrillation: a prospective multicenter study. J Interv Card Electrophysiol 61, 617–622 (2021). https://doi.org/10.1007/s10840-020-00833-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-020-00833-8