Abstract
Purpose
Cardiac implantable electronic devices (CIEDs) have traditionally been a contraindication for magnetic resonance imaging (MRI). Recent studies suggest that MRI can be conducted safely in select patients with pacemakers (PPMs) and implantable cardioverter defibrillators (ICDs). We sought to determine the safety of MRI in patients with CIEDs, using a protocol for patient selection and device programming.
Methods
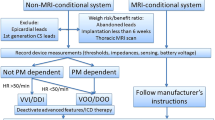
This is a prospective, single-center study. Patients with a PPM or ICD and a clinical indication for MRI were considered. Exclusion criteria included newly implanted devices (<4 weeks), PPMs manufactured before 1996 and ICDs before 2000, epicardial and abandoned leads, and pacemaker-dependent ICD patients. Pacemaker-dependent PPM patients were programmed to asynchronous pacing. Tachycardia detection/therapies were disabled for ICDs. Devices were interrogated pre- and post-scan, and at follow-up 1–6 weeks later. Defibrillation threshold (DFT) was not tested post-scan. Patients were followed to monitor device therapies.
Results
Two hundred twenty-seven patients underwent 293 scans. Devices included 170 (70.6%) PPMs and 71 (29.5%) ICDs. Thirteen (4.4%) scans were aborted mainly due to subjective complaints or artifact on scout cardiac imaging. Post-scan and follow-up interrogation demonstrated no changes in device parameters requiring reprogramming or revision. Over long-term follow-up (median, 354 days [IQR 65–629]), nine ICD patients had appropriate shocks (median, 3 [IQR 1–8]). One had four inappropriate shocks for atrial fibrillation. All tachyarrhythmias meeting criteria for defibrillation were successfully terminated.
Conclusions
MRI can be conducted safely in patients with CIEDs when done in a protocoled manner with appropriate supervision. DFT testing after MRI may not be necessary.

Similar content being viewed by others
References
Chang AE, Matory YL, Dwyer AJ, Hill SC, Girton ME, Steinberg SM, et al. Magnetic resonance imaging versus computed tomography in the evaluation of soft tissue tumors of the extremities. Ann Surg. 1987;205(4):340–8.
Ridgway JP. Cardiovascular magnetic resonance physics for clinicians: part I. J Cardiovasc Magn Reson Off J Soc Cardiovasc Magn Reson. 2010;12:71.
Levine GN, Gomes AS, Arai AE, Bluemke DA, Flamm SD, Kanal E, et al. Safety of magnetic resonance imaging in patients with cardiovascular devices: an American Heart Association scientific statement from the Committee on Diagnostic and Interventional Cardiac Catheterization, Council on Clinical Cardiology, and the Council on Cardiovascular Radiology and Intervention: endorsed by the American College of Cardiology Foundation, the North American Society for Cardiac Imaging, and the Society for Cardiovascular Magnetic Resonance. Circulation. 2007;116(24):2878–91.
Babouri A, Hedjeidj A. In vitro investigation of eddy current effect on pacemaker operation generated by low frequency magnetic field. Conf Proc Annu Int Conf IEEE Eng Med Biol Soc IEEE Eng Med Biol Soc Annu Conf. 2007;2007:5684–7.
Achenbach S, Moshage W, Diem B, Bieberle T, Schibgilla V, Bachmann K. Effects of magnetic resonance imaging on cardiac pacemakers and electrodes. Am Heart J. 1997;134(3):467–73.
Luechinger R, Duru F, Scheidegger MB, Boesiger P, Candinas R. Force and torque effects of a 1.5-tesla MRI scanner on cardiac pacemakers and ICDs. Pacing Clin Electrophysiol PACE. 2001;24(2):199–205.
Luechinger R, Zeijlemaker VA, Pedersen EM, Mortensen P, Falk E, Duru F, et al. In vivo heating of pacemaker leads during magnetic resonance imaging. Eur Heart J. 2005;26(4):376–83. discussion 325–7
Gimbel JR, Johnson D, Levine PA, Wilkoff BL. Safe performance of magnetic resonance imaging on five patients with permanent cardiac pacemakers. Pacing Clin Electrophysiol PACE. 1996;19(6):913–9.
Sommer T, Lauck G, Schimpf R, von Smekal A, Wolke S, Block W, et al. MRI in patients with cardiac pacemakers: in vitro and in vivo evaluation at 0.5 tesla. RöFo Fortschritte Auf Dem Geb Röntgenstrahlen Nukl. 1998;168(1):36–43.
Sommer T, Vahlhaus C, Lauck G, von Smekal A, Reinke M, Hofer U, et al. MR imaging and cardiac pacemakers: in-vitro evaluation and in-vivo studies in 51 patients at 0.5 T. Radiology. 2000;215(3):869–79.
Vahlhaus C, Sommer T, Lewalter T, Schimpf R, Schumacher B, Jung W, et al. Interference with cardiac pacemakers by magnetic resonance imaging: are there irreversible changes at 0.5 tesla? Pacing Clin Electrophysiol PACE. 2001;24(4 Pt 1):489–95.
Martin ET, Coman JA, Shellock FG, Pulling CC, Fair R, Jenkins K. Magnetic resonance imaging and cardiac pacemaker safety at 1.5-tesla. J Am Coll Cardiol. 2004;43(7):1315–24.
Del Ojo JL, Moya F, Villalba J, Sanz O, Pavón R, Garcia D, et al. Is magnetic resonance imaging safe in cardiac pacemaker recipients? Pacing Clin Electrophysiol PACE. 2005;28(4):274–8.
Gimbel JR, Kanal E, Schwartz KM, Wilkoff BL. Outcome of magnetic resonance imaging (MRI) in selected patients with implantable cardioverter defibrillators (ICDs). Pacing Clin Electrophysiol PACE. 2005;28(4):270–3.
Sommer T, Naehle CP, Yang A, Zeijlemaker V, Hackenbroch M, Schmiedel A, et al. Strategy for safe performance of extrathoracic magnetic resonance imaging at 1.5 tesla in the presence of cardiac pacemakers in non-pacemaker-dependent patients: a prospective study with 115 examinations. Circulation. 2006;114(12):1285–92.
Mollerus M, Albin G, Lipinski M, Lucca J. Cardiac biomarkers in patients with permanent pacemakers and implantable cardioverter-defibrillators undergoing an MRI scan. Pacing Clin Electrophysiol PACE. 2008;31(10):1241–5.
Naehle CP, Zeijlemaker V, Thomas D, Meyer C, Strach K, Fimmers R, et al. Evaluation of cumulative effects of MR imaging on pacemaker systems at 1.5 tesla. Pacing Clin Electrophysiol PACE. 2009;32(12):1526–35.
Naehle CP, Strach K, Thomas D, Meyer C, Linhart M, Bitaraf S, et al. Magnetic resonance imaging at 1.5-T in patients with implantable cardioverter-defibrillators. J Am Coll Cardiol. 2009;54(6):549–55.
Burke PT, Ghanbari H, Alexander PB, Shaw MK, Daccarett M, Machado C. A protocol for patients with cardiovascular implantable devices undergoing magnetic resonance imaging (MRI): should defibrillation threshold testing be performed post-(MRI). J Interv Card Electrophysiol Int J Arrhythm Pacing. 2010;28(1):59–66.
Halshtok O, Goitein O, Abu Sham’a R, Granit H, Glikson M, Konen E. Pacemakers and magnetic resonance imaging: no longer an absolute contraindication when scanned correctly. Isr Med Assoc J IMAJ. 2010;12(7):391–5.
Mollerus M, Albin G, Lipinski M, Lucca J. Magnetic resonance imaging of pacemakers and implantable cardioverter-defibrillators without specific absorption rate restrictions. Eur Eur Pacing Arrhythm Card Electrophysiol J Work Groups Card Pacing Arrhythm Card Cell Electrophysiol Eur Soc Cardiol. 2010;12(7):947–51.
Strach K, Naehle CP, Mühlsteffen A, Hinz M, Bernstein A, Thomas D, et al. Low-field magnetic resonance imaging: increased safety for pacemaker patients? Eur Eur Pacing Arrhythm Card Electrophysiol J Work Groups Card Pacing Arrhythm Card Cell Electrophysiol Eur Soc Cardiol. 2010;12(7):952–60.
Naehle CP, Kreuz J, Strach K, Schwab JO, Pingel S, Luechinger R, et al. Safety, feasibility, and diagnostic value of cardiac magnetic resonance imaging in patients with cardiac pacemakers and implantable cardioverters/defibrillators at 1.5 T. Am Heart J. 2011;161(6):1096–105.
Nazarian S, Hansford R, Roguin A, Goldsher D, Zviman MM, Lardo AC, et al. A prospective evaluation of a protocol for magnetic resonance imaging of patients with implanted cardiac devices. Ann Intern Med. 2011;155(7):415–24.
Russo RJ, Costa HS, Silva PD, Anderson JL, Arshad A, Biederman RWW, et al. Assessing the risks associated with MRI in patients with a pacemaker or defibrillator. N Engl J Med. 2017;376(8):755–64.
Langman DA, Goldberg IB, Finn JP, Ennis DB. Pacemaker lead tip heating in abandoned and pacemaker-attached leads at 1.5 tesla MRI. J Magn Reson Imaging JMRI. 2011;33(2):426–31.
Higgins JV, Gard JJ, Sheldon SH, Espinosa RE, Wood CP, Felmlee JP, et al. Safety and outcomes of magnetic resonance imaging in patients with abandoned pacemaker and defibrillator leads. Pacing Clin Electrophysiol PACE. 2014;37(10):1284–90.
Healey JS, Hohnloser SH, Glikson M, Neuzner J, Viñolas X, Mabo P, et al. The rationale and design of the Shockless IMPLant evaluation (SIMPLE) trial: a randomized, controlled trial of defibrillation testing at the time of defibrillator implantation. Am Heart J. 2012;164(2):146–52.
Sweeney MO. Antitachycardia pacing for ventricular tachycardia using implantable cardioverter defibrillators: pacing. Clin Electrophysiol PACE. 2004;27(9):1292–305.
Fisher JD, Mehra R, Furman S. Termination of ventricular tachycardia with bursts of rapid ventricular pacing. Am J Cardiol. 1978;41(1):94–102.
Bertomeu-González V, Moreno-Arribas J, Castillo-Castillo J, Martínez-Ferrer J, Viñolas X, Rodríguez A, et al. Etiology and programming effects on shock efficacy in ICD recipients. Pacing Clin Electrophysiol PACE. 2016;39(1):73–80.
Dewey M, Schink T, Dewey CF. Claustrophobia during magnetic resonance imaging: cohort study in over 55,000 patients. J Magn Reson Imaging JMRI. 2007;26(5):1322–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding sources
No external funding source.
Conflict of interest
Mrinal Yadava has received travel support from St. Jude and Biotronik. Charles A. Henrikson has received fellowship support from Boston Scientific, St. Jude, and Medtronic. Other authors declare that they have no conflict of interest.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Yadava, M., Nugent, M., Krebsbach, A. et al. Magnetic resonance imaging in patients with cardiac implantable electronic devices: a single-center prospective study. J Interv Card Electrophysiol 50, 95–104 (2017). https://doi.org/10.1007/s10840-017-0262-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-017-0262-6