Abstract
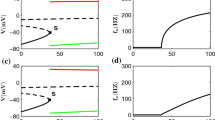
Adaptation of the spike-frequency response to constant stimulation, as observed on various timescales in many neurons, reflects high-pass filter properties of a neuron’s transfer function. Adaptation in general, however, is not sufficient to make a neuron’s response independent of the mean intensity of a sensory stimulus, since low frequency components of the stimulus are still transmitted, although with reduced gain. We here show, based on an analytically tractable model, that the response of a neuron is intensity invariant, if the fully adapted steady-state spike-frequency response to constant stimuli is independent of stimulus intensity. Electrophysiological recordings from the AN1, a primary auditory interneuron of crickets, show that for intensities above 60 dB SPL (sound pressure level) the AN1 adapted with a time-constant of ~40 ms to a steady-state firing rate of ~100 Hz. Using identical random amplitude-modulation stimuli we verified that the AN1’s spike-frequency response is indeed invariant to the stimulus’ mean intensity above 60 dB SPL. The transfer function of the AN1 is a band pass, resulting from a high-pass filter (cutoff frequency at 4 Hz) due to adaptation and a low-pass filter (100 Hz) determined by the steady-state spike frequency. Thus, fast spike-frequency adaptation can generate intensity invariance already at the first level of neural processing.
Similar content being viewed by others
References
Baccus, S. A., & Meister, M. (2002). Fast and slow contrast adaptation in retinal circuitry. Neuron, 36, 909–919.
Barlow, H. B. (1961). Possible principles underlying the transformation of sensory messages. In W. Rosenblith (Ed.), Sensory communication (pp. 217–234). Cambridge, MA: MIT Press.
Benda, J., & Herz, A. V. M. (2003). A universal model for spike-frequency adaptation. Neural Computation, 15, 2523–2564.
Benda, J., Longtin, A., & Maler, L. (2005). Spike-frequency adaptation separates transient communication signals from background oscillations. Journal of Neuroscience, 25, 2312–2321.
Brenner, N., Bialek, W., & de Ruyter van Steveninck, R. (2000). Adaptive rescaling maximizes information transfer. Neuron, 26, 695–702.
Cartling, B. (1996). A low-dimensional, time resolved and adapting model neuron. International Journal of Neural Systems, 7, 237–246.
Dean, I., Harper, N. S., & McAlpine, D. (2005). Neural population coding of sound level adapts to stimulus statistics. Nature Neuroscience, 8, 1684–1689.
Doolan, J. M., & Pollack, G. S. (1985). Phonotactic specifity of the cricket Teleogryllus oceanicus: Intensity-dependent selectivity for temporal parameters of the stimulus. Journal of Comparative Physiology, A, 157, 223–233.
Esch, H., Huber, F., & Wohlers, D. W. (1980). Primary auditory neurons in crickets: Physiology and central projections. Journal of Comparative Physiology, A, 137, 27–38.
Fairhall, A. L., Lewen, G. D., Bialek, W., & de Ruyter van Steveninck, R. R. (2001). Efficiency and ambiguity in an adaptive neural code. Nature, 412, 787–792.
Fourcaud-Trocmé, N., Hansel, D., van Vreeswijk, C., & Brunel, N. (2003). How spike generation mechanisms determine the neuronal response to fluctuating inputs. Journal of Neuroscience, 23, 11628–11640.
French, A. S., Höger, U., Sekizawa, S. I., & Torkkeli, P. H. (2001). Frequency response functions and information capacities of paired spider mechanoreceptor neurons. Biological Cybernetics, 85, 293–300.
French, A. S., & Torkkeli, P. H. (1994). The time course of sensory adaptation in the cockroach tactile spine. Neuroscience Letters, 178, 147–150.
Fuhrmann, G., Markram, H., & Tsodyks, M. (2002). Spike frequency adaptation and neocortical rhythms. Journal of Neurophysiology, 88, 761–770.
Gabbiani, F., & Krapp, H. G. (2006). Spike-frequency adaptation and intrinsic properties of an identified, looming-sensitive neuron. Journal of Neurophysiology, 96, 2951–2962.
Gabbiani, F., Krapp, H. G., Hatsopoulos, N., Mo, C. H., Koch, C., & Laurent, G. (2004). Multiplication and stimulus invariance in a looming-sensitive neuron. Journal of physiology Paris, 98, 19–34.
Hennig, R. M. (1988). Ascending auditory interneurons in the cricket Teleogryllus commodus (Walker): Comparative physiology and direct connections with afferents. Journal of Comparative Physiology, A, 163, 135–143.
Hennig, R. M. (2003). Acoustic feature extraction by cross-correlation in crickets? Journal of Comparative Physiology, A, 189, 589–598.
Hennig, R. M., & Weber, T. (1997). Filtering of temporal parameters of the calling song by cricket females of two closely related species: A behavioural analysis. Journal of Comparative Physiology A, 180, 621–630.
Horseman, G., & Huber, F. (1994a). Sound localisation in crickets I: Contralateral inhibition of an ascending auditory interneuron (AN1) in the cricket Gryllus bimaculatus. Journal of Comparative Physiology, A, 175, 389–398.
Horseman, G., & Huber, F. (1994b). Sound localisation in crickets II: Modelling the role of a simple neural network in the prothoracic ganglion. Journal of Comparative Physiology, A, 175, 399–413.
Huber, F., Moore, T. E., & Loher, W. (1989). Cricket behavior and neurobiology. Ithaca, NY: Cornell University Press.
Kim, K. J., & Rieke, F. (2003). Slow Na+ inactivation and variance adaptation in salamander retinal ganglion cells. Journal of Neuroscience, 23, 1506–1516.
Knight, B. W. (1972). Dynamics of encoding in a population of neurons. Journal of General Physiology, 59, 734–766.
Kvale, M. N., & Schreiner, C. E. (2004). Short-term adaptation of auditory receptive fields to dynamic stimuli. Journal of Neurophysiology, 91, 604–612.
Laughlin, S. B. (1989). The role of sensory adaptation in the retina. Journal of Experimental Biology, 146, 39–62.
Maravall, M., Petersen, R. S., Fairhall, A. L., Arabzadeh, E., & Diamond, M. E. (2007). Shifts in coding properties and maintenance of information transmission during adaptation in barrel cortex. PLoS Biology, 5(2), e19.
Marsat, G., & Pollack, G. S. (2005). Effect of the temporal pattern of contralateral inhibition on sound localization cues. Journal of Neuroscience, 25, 6137–6144.
Nelken, I., Fishbach, A., Las, L., Ulanovsky, N., & Farkas, D. (2003). Primary auditory cortex of cats: Feature detection or something else? Biological Cybernetics, 89, 397–406.
Nelson, M. E., Xu, Z., & Payne, J. R. (1997). Characterization and modeling of P-type electrosensory afferent responses to amplitude modulations in a wave-type electric fish. Journal of Comparative Physiology, A, 181, 532–544.
Park, T. J., Klug, A., Holinstat, M., & Grothe, B. (2004). Interaural level difference processing in the lateral superior olive and the inferior colliculus. Journal of Neurophysiology, 92, 289–301.
Pollack, G. S. (1988). Selective attentiion in an insect auditory neuron. Journal of Neuroscience, 8, 2635–2639.
Pollack, G. S., & El-Feghaly, E. (1993). Calling song recognition in the cricket Teleogryllus oceanicus: Comparison of the effects of stimulus intensity and sound spectrum on selectivity for temporal patterns. Journal of Comparative Physiology, A, 171, 759–765.
Press, W. H., Teukolsky, S. A., Vetterling, W. T., & Flannery, B. P. (1992). Numerical recipes in C (2nd edn.). Cambridge: Cambridge University Press.
Reinagel, P. (2001). Neurobiology: The many faces of adaptation. Nature, 412, 776–777.
Römer, H., & Krusch, M. (2000). A gain-control mechanism for processing of chorus sounds in the afferent auditory pathway of the bushcricket Tettigonia viridissima (Orthoptera; Tettigoniidae). Journal of Comparative Physiology, A, 186, 181–191.
Sanchez-Vives, M. V., Nowak, L. G., & McCormick, D. A. (2000). Membrane mechanisms underlying contrast adaptation in cat area 17 in vivo. Journal of Neuroscience, 20, 4267–4285.
Schildberger, K. (1984). Temporal selectivity of identified auditory neurons in the cricket brain. Journal of Comparative Physiology A, 155, 171–185.
Schildberger, K., & Hörner, M. (1988). The function of auditory neurons in cricket phonotaxis. Journal of Comparative Physiology, A, 163, 621–631.
Smirnakis, S. M., Berry, M. J., Warland, D. K., Bialek, W., & Meister, M. (1997). Adaptation of retinal processing to image contrast and spatial scale. Nature, 386, 69–73.
Sobel, E. C., & Tank, D. W. (1994). In vivo Ca2+ dynamics in a cricket auditory neuron: An example of chemical computation. Science, 263, 823–826.
Solomon, S. G., Peirce, J. W., Dhruv, N. T., & Lennie, P. (2004). Profound contrast adaptation early in the visual pathway. Neuron, 42, 155–162.
Ulanovsky, N., Las, L., Farkas, D., & Nelken, I. (2004). Multiple time scales of adaptation in auditory cortex neurons. Journal of Neuroscience, 24, 10440–10453.
Wang, X. J. (1998). Calcium coding and adaptive temporal computation in cortical pyramidal neurons. Journal of Neurophysiology, 79, 1549–1566.
Weber, T., Thorson, J., & Huber, F. (1981). Auditory behaviour of the cricket I dynamics of compensated walking and discrimination paradigms on the kramer treadmill. Journal of Comparative Physiology A, 141, 215–232.
Wiskott, L. (2003). Slow feature analysis: A theoretical analysis of optimal free responses. Neural Computation, 15, 2147–2177.
Wohlers, D. W., & Huber, F. (1982). Processing of sound signals by six types of neurons in the prothoracic ganglion of the cricket, Gryllus campestrisL. Journal of Comparative Physiology A, 146, 161–173.
Xu, Z., Payne, J. R., & Nelson, M. E. (1996). Logarithmic time course of sensory adaptation in electrosensory afferent nerve fibers in a weakly electric fish. Journal of Neurophysiology, 76, 2020–2032.
Author information
Authors and Affiliations
Corresponding author
Additional information
Action Editor: Israel Nelken
Rights and permissions
About this article
Cite this article
Benda, J., Hennig, R.M. Spike-frequency adaptation generates intensity invariance in a primary auditory interneuron. J Comput Neurosci 24, 113–136 (2008). https://doi.org/10.1007/s10827-007-0044-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-007-0044-8