Abstract
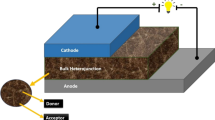
In this work, six dipyridine dipyrrole (DPDP) derivatives are reported as π-conjugated, electron donor molecules used in BHJ solar cells. Density functional theory (DFT) method with the Becke three-parameter, hybrid functional combined with the Lee, Yang, and Parr correlation functional (B3LYP) taking 6-311G(d) basis set supported by the Gaussian 09 program has been employed for the determination of the structural, geometrical and molecular electrostatic potential. Absorption properties have been examined employing the time-dependent density functional theory (TD-DFT) method with B3LYP functional and 6-311G(d) basis set. Two major photovoltaic parameters for predicting the efficiency of the bulk heterojunction (BHJ) solar cell for power conversion, namely, open-circuit voltages (Voc) and light-harvesting efficiencies (LHE), are studied. The π-conjugated molecules, due to their alternate π-bonds and lower bandgap energy (Egap), possess excellent photovoltaic properties, and large photocurrent generation is obtained upon light absorption. The reported molecules can be used as electron donor molecules in the active part of the BHJ devices to generate photoelectronic current based on their studied properties. The reported molecules can be easily synthesized and have properties like mechanical flexibility, non-toxicity, low-cost production platform, and biodegradability.









Similar content being viewed by others
References
Bourass, M., Amine, A., Hamidi, M., Bouachrine, M.: New organic dyes based on phenylenevinylene for solar cells: DFT and TD-DFT investigation. Karbala Int. J. Mod. Sci. 3, 75–82 (2017)
Raftani, M., Abram, T., Bennani, N., Bouachrine, M.: Theoretical study of new conjugated compounds with a low bandgap for bulk heterojunction solar cells: DFT and TD-DFT study. Results Chem. 2, 100040 (2020)
Bourass, M., Benjelloun, A.T., Benzakour, M., Mcharfi, M., Jhilal, F., Hamidi, M., Bouachrine, M.: The optoelectronic properties of organic materials based on triphenylamine that are relevant to organic solar photovoltaic cells. New J. Chem. 41, 13336–13346 (2017)
Scharber, M.C., Mühlbacher, D., Koppe, M., Denk, P., Waldauf, C., Heeger, A.J., Brabec, C.J.: Design rules for donors in bulk-heterojunction solar cells-Towards 10% energy-conversion efficiency. Adv. Mater. 18, 789–794 (2006)
Chapin, D.M., Fuller, C.S., Pearson, G.L.: A new silicon p-n junction photocell for converting solar radiation into electrical power. J. Appl. Phys. 25, 676 (1954)
Chidichimo, G., Filippelli, L.: Organic Solar Cells: Problems and Perspectives. Int. J. Photoenergy 2010, 123534 (2010)
Sampaio, P.G., González, M.O.: Photovoltaic solar energy: conceptual framework. Renew. Sustain. Energy Rev. 74, 590–601 (2017)
Khlyabich, P.P., Burkhart, B., Rudenko, A.E., Thompson, B.C.: Optimization and simplification of polymer–fullerene solar cells through polymer and active layer design. Polymer 54, 5267–5298 (2013)
Dennler, G., Scharber, M.C., Brabec, C.J.: Polymer-fullerene bulk-heterojunction solar cells. Adv. Mater. 21, 1323–1338 (2009)
Heeger, A.J.: 25th anniversary article: bulk heterojunction solar cells: understanding the mechanism of operation. Adv. Mater. 26, 10–28 (2014)
Schilinsky, P., Waldauf, C.: Recombination and loss analysis in polythiophene based bulk heterojunction photodetectors. Appl. Phys. Lett. 81, 3885 (2002)
Zhang, J., Tan, H.S., Guo, X., Facchetti, A., Yan, H.: Material insights and challenges for non-fullerene organic solar cells based on small molecular acceptors. Nat. Energy 3, 720–731 (2018)
Shaheen, S.E., Brabec, C.J.: Sariciftci, N.S.: 2.5% efficient organic plastic solar cells. Appl. Phys. Lett. 78, 841 (2001)
Yang, L., Yan, L., You, W.: Organic solar cells beyond one pair of donor-acceptor: ternary blends and more. J. Phys. Chem. Lett. 4, 1802–1810 (2013)
Green, M.A., Emery, K., Hishikawa, Y., Warta, W., Dunlop, E.D., Levi, D.H., Ho-Baillie, A.W.Y.: Solar cell efficiency tables (version 49). Prog. Photovolt. Res. Appl. 25, 3–13 (2017)
Krebs, F.C., Spanggaard, H.: Significant improvement of polymer solar cell stability. Chem. Mater. 17, 5235–5237 (2005)
Brabec, C.J., Zerza, G., Cerullo, G., De Silvestri, S., Luzzati, S., Hummelen, J.C., Sariciftci, S.: Tracing photoinduced electron transfer process in conjugated polymer/fullerene bulk heterojunctions in real time. Chem. Phys. Lett. 340, 232–236 (2001)
Schmidt-Mende, L., Bach, U., Humphry-Baker, R., Horiuchi, T., Miura, H., Ito, S., Uchida, S., Grätzel, M.: Organic dye for highly efficient solid-state dye-sensitized solar cells. Adv. Mater. 17, 813–815 (2005)
Bagher, A.M., Vahid, M.M., Mohsen, M.: Types of solar cells and application. Am. J. Opt. Photonics 3, 94–113 (2015)
Tabatchnik-Rebillon, A., Aub, C., Bakkali, H., Delaunay, T., Manh, G.T., Blot, V., Thobie-Gautier, C., Renault, E., Soulard, M., Planchat, A., Questel, J.-Y.L., Guvel, R.L., Guguen-Guillouzo, C., Kauffmann, B., Ferrand, Y., Huc, I., Urgin, K., Condon, S., Lonel, E., Evain, M., Lebreton, J., Jacquemin, D., Pipelier, M., Dubreuil, D.: Electrochemical synthesis and characterisation of alternating tripyridyl– dipyrrole molecular strands with multiple nitrogen-based donor-acceptor binding sites. Chem. Eur. J. 16, 11876–11889 (2010)
Smithen, D.A., Monro, S., Pinto, M., Roque, J., III., Diaz-Rodriguez, R.M., Yin, H., Cameron, C.G., Thompson, A., McFarland, S.A.: Bis[pyrrolyl Ru(ii)] triads: a new class of photosensitizers for metal–organic photodynamic therapy. Chem. Sci. 11, 12047–12069 (2020)
UrRehman, S., Alam, A., Bibi, S., Sadaf, S., Khan, S.R., Shoaib, M., Khan, A.Q., Khan, M., UrRehman, W.: The effect of different aromatic conjugated bridges on optoelectronic properties of diketopyrrolopyrrole-based donor materials for organic photovoltaics. J. Mol. Model. 26, 154 (2020)
Aicha, Y.A., Bouzzine, S.M., Zair, T., Bouachrine, M., Hamidi, M., Salgado-Morán, G., Tagle, R.R., Mendoza-Huizar, L.H.: Tuning the electronic, photophysical and charge transfer properties of small DA molecules based on Thienopyrazine-terthienyls by changing the donor fragment: a DFT study. J. Chil. Chem. Soc. 62, 3637–3646 (2017)
Alharbi, F.H., Rashkeev, S.N., El-Mellouhi, F., Lüthi, H.P., Tabet, N., Kais, S.: An efficient descriptor model for designing materials for solar cells. npj Comput. Mater. 1, 15003 (2015)
O’boyle, N.M., Tenderholt, A.L., Langner, K.M.: cclib: a library for package-independent computational chemistry algorithms. J. Comput. Chem. 29, 839–845 (2008)
El Alamy, A., Amine, A., Bouzzine, S.M., Lachgar, M., Hamidi, M., Elhamzi, A., Bouachrine, M.: DFT study of small compounds based on thiophene and benzo [1,2,5] thiadiazole for solar cells: correlation-structure/electronic properties. J. Mater. Environ. Sci. 8, 3897–3905 (2017)
El Assyry, A., Lamsayah, M., Warad, I., Touzani, R., Bentiss, F., Zarrouk, A.: Theoretical investigation using DFT of quinoxaline derivatives for electronic and photovoltaic effects. Heliyon 6, e03620 (2020)
Shao, S., Shi, J., Murtaza, I., Xu, P., He, Y., Ghosh, S., Zhu, X., Perepichka, I.F., Meng, H.: Exploring the electrochromic properties of poly(thieno[3,2-b]thiophene)s decorated with electron-deficient side groups. Polym. Chem. 8, 769–784 (2017)
Youssef, A.A., Bouzzine, S.M., Fahim, Z.M., Sıdır, İ, Hamidi, M., Bouachrine, M.: Designing donor-acceptor thienopyrazine derivatives for more efficient organic photovoltaic solar cell: a DFT study. Physica B Condens. Matter 560, 111–125 (2019)
Politzer, P., Murray, J.S.: The fundamental nature and role of the electrostatic potential in atoms and molecules. Theor. Chem. Acc. 108, 134–142 (2002)
Fei, E.T.L., Biswas, J., Datta, B., Kumar, D.: Computational studies of diindole-based molecules for organic bulk heterojunction solar devices using DFT and TD-DFT calculations. Struct. Chem. 32, 1973–1984 (2021)
Liu, W., Yang, S., Li, J., Su, G., Ren, J.C.: One molecule, two states: single molecular switch on metallic electrodes. WIREs Comput. Mol. Sci. 11, e1511 (2020)
Alipour, M., Mohajeri, A.: Molecular electrostatic potential as a tool for evaluating the etherification rate constant. J. Phys. Chem. A 114, 7417–7422 (2010)
Gadre, S.R., Suresh, C.H., Mohan, N.: Electrostatic potential topology for probing molecular structure, bonding and reactivity. Molecules 26, 3289 (2021)
El Azzouzi, M., Aouniti, A., Herrag, L., Chetouani, A., Elmsellem, H., Hammouti, B.: Investigation of isomers of hydroxyphenylamino propane nitrile as mild steel corrosion inhibitors in 1 M HCl. Der Pharma Chem. 7, 12–24 (2015)
Pearson, R.G.: Absolute electronegativity and hardness correlated with molecular orbital theory. Proc. Natl. Acad. Sci. U.S.A. 83, 8440–8441 (1986)
Zahllou, A., Abram, T., Boussaidi, S., Zgou, H., Bejjit, L., Bouachrine, M.: Theoretical investigation of new organic materials based on fluorene and thiophene for photovoltaic applications. Mor. J. Chem. 3, 861–871 (2015)
Chattaraj, P.K., Maiti, B.: HSAB principle applied to the time evolution of chemical reactions. J. Am. Chem. Soc. 125, 2705–2710 (2003)
Divya, V.V., Suresh, C.H.: Density functional theory study on the donating strength of donor systems in dye-sensitized solar cells. New J. Chem. 44, 7200–7209 (2020)
Divya, V.V., Suresh, C.H.: Tuning the donating strength of dye sensitizers using molecular electrostatic potential analysis. New J. Chem. 45, 2496–2507 (2021)
Anjali, B.A., Suresh, C.H.: Absorption and emission properties of 5-phenyl tris(8-hydroxyquinolinato) M(III) complexes (M = Al, Ga, In) and correlations with molecular electrostatic potential. J. Comput. Chem. 41, 1497–1508 (2020)
Divya, V.V., Sayyed, F.B., Suresh, C.H.: substituent effect transmission power of alkyl, alkenyl, alkynyl, phenyl, thiophenyl, and polyacene spacers. ChemPhysChem 20, 1752–1758 (2019)
Preat, J., Jacquemin, D., Perpete, E.A.: Towards new efficient dye-sensitized solar cells. Energy Environ. Sci. 3, 891–904 (2010)
Bourass, M., Benjelloun, A.T., Benzakour, M., Mcharfi, M., Hamidi, M., Bouzzine, S.M., Bouachrine, M.: DFT and TD-DFT calculation of new thienopyrazine-based small molecules for organic solar cells. Chem. Cent. J. 10, 67 (2016)
Durrant, J.R., Haque, S.A., Palomares, E.: Towards optimization of electron transfer processes in dye sensitized solar cells. Coord. Chem. Rev. 248, 1247–1257 (2004)
Dias, F.B., Bourdakos, K.N., Jankus, V., Moss, K.C., Kamtekar, K.T., Bhalla, V., Santos, J., Bryce, M.R., Monkman, A.P.: Triplet harvesting with 100% efficiency by way of thermally activated delayed fluorescence in charge transfer OLED emitters. Adv. Mater. 25, 3707–3714 (2013)
Acknowledgments
The authors S. Tamang, A. Thapa, K. Chettri and J. Biswas, would like to acknowledge the Department of Chemistry, Sikkim Manipal Institute of Technology, for help and support. The author B. Datta wishes to acknowledge the Department of Chemistry, Amity University, Kolkata, for help and support.
Funding
No funding was received.
Author information
Authors and Affiliations
Contributions
S. Tamang performed all computational studies and analyzed the data. A. Thapa and K. Chettri helped analyze the data. B. Datta was involved in the conceptualization, drafting and reviewing of the manuscript. J. Biswas was involved in the conceptualization, design and manuscript drafting.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tamang, S., Thapa, A., Chettri, K. et al. Analysis of dipyridine dipyrrole based molecules for solar cell application using computational approach. J Comput Electron 21, 94–105 (2022). https://doi.org/10.1007/s10825-021-01822-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10825-021-01822-4