Abstract
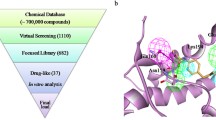
Conformational conversion of the normal cellular prion protein, PrPC, into the misfolded isoform, PrPSc, is considered to be a central event in the development of fatal neurodegenerative diseases. Stabilization of prion protein at the normal cellular form (PrPC) with small molecules is a rational and efficient strategy for treatment of prion related diseases. However, few compounds have been identified as potent prion inhibitors by binding to the normal conformation of prion. In this work, to rational screening of inhibitors capable of stabilizing cellular form of prion protein, multiple approaches combining docking-based virtual screening, steady-state fluorescence quenching, surface plasmon resonance and thioflavin T fluorescence assay were used to discover new compounds interrupting PrPC to PrPSc conversion. Compound 3253-0207 that can bind to PrPC with micromolar affinity and inhibit prion fibrillation was identified from small molecule databases. Molecular dynamics simulation indicated that compound 3253-0207 can bind to the hotspot residues in the binding pocket composed by β1, β2 and α2, which are significant structure moieties in conversion from PrPC to PrPSc.








Similar content being viewed by others
References
Collinge J, Sidle KC, Meads J, Ironside J, Hill AF (1996) Nature 383:685–690
Heinemann U, Krasnianski A, Meissner B, Varges D, Kallenberg K, Schulz-Schaeffer W, Steinhoff B, Grasbon-Frodl E, Kretzschmar H, Zerr I (2007) Brain 130(5):1350–1359
Prusiner SB (1998) Proc Natl Acad Sci USA 95(23):13363–13383
Collinge J (2001) Annu Rev Neurosci 24(1):519–550
Stahl N, Baldwin MA, Teplow DB, Hood L, Gibson BW, Burlingame AL, Prusiner SB (1993) Biochemistry 32(8):1991–2002
Sigurdson CJ, Nilsson KPR, Hornemann S, Heikenwalder M, Manco G, Schwarz P, Ott D, Rülicke T, Liberski PP, Julius C (2009) Proc Natl Acad Sci USA 106(1):304–309
Castilla J, Saá P, Hetz C, Soto C (2005) Cell 121(2):195–206
Pan K-M, Baldwin M, Nguyen J, Gasset M, Serban A, Groth D, Mehlhorn I, Huang Z, Fletterick RJ, Cohen FE (1993) Proc Natl Acad Sci USA 90(23):1096210966
Caughey BW, Dong A, Bhat KS, Ernst D, Hayes SF, Caughey WS (1991) Biochemistry 30(31):7672–7680
Prusiner SB (1991) Science 252(5012):1515–1522
Trevitt CR, Collinge J (2006) Brain 129(Pt 9):2241–2265
Poncet-Montange G, St Martin SJ, Bogatova OV, Prusiner SB, Shoichet BK, Ghaemmaghami S (2011) J Biol Chem 286(31):27718–27728
Ghaemmaghami S, May BC, Renslo AR, Prusiner SB (2010) J Virol 84(7):3408–3412
Bertsch U, Winklhofer KF, Hirschberger T, Bieschke J, Weber P, Hartl FU, Tavan P, Tatzelt J, Kretzschmar HA, Giese A (2005) J Virol 79(12):7785–7791
Giese A, Kretzschmar H (2001) Prion-induced neuronal damage—the mechanisms of neuronal destruction in the subacute spongiform encephalopathies. The mechanisms of neuronal damage in virus infections of the nervous system. Springer, Berlin, pp 203–217
Antonyuk SV, Trevitt CR, Strange RW, Jackson GS, Sangar D, Batchelor M, Cooper S, Fraser C, Jones S, Georgiou T, Khalili-Shirazi A, Clarke AR, Hasnain SS, Collinge J (2009) Proc Natl Acad Sci USA 106(8):25542558
Singh J, Udgaonkar JB (2015) Angew Chem Int Ed Engl 54(26):7529–7533
Ghaemmaghami S, Russo M, Renslo AR (2014) J Med Chem 57(16):6919–6929
Baral PK, Swayampakula M, Rout MK, Kav NN, Spyracopoulos L, Aguzzi A, James MN (2014) Structure 22(2):291–303
Singh J, Kumar H, Sabareesan AT, Udgaonkar JB (2014) J Am Chem Soc 136(48):16704–16707
Rester U (2008) Curr Opin Drug Discov Dev 11(4):559–568
Walters WP, Stahl MT, Murcko MA (1998) Drug Discov Today 3(4):160–178
Lakowicz JR (1983) Quenching of fluorescence. Principles of fluorescence spectroscopy. Springer, New York, pp 277–330
Mátyus L, Szöllősi J, Jenei A (2006) J Photochem Photobiol B 83(3):223–236
Rich RL, Myszka DG (2003) J Mol Recognit 16(6):351–382
Rich RL, Hoth LR, Geoghegan KF, Brown TA, LeMotte PK, Simons SP, Hensley P, Myszka DG (2002) Proc Natl Acad Sci USA 99(13):8562–8567
Navratilova I, Hopkins AL (2010) ACS Med Chem Lett 1(1):44–48
Zhou T, Xu L, Dey B, Hessell AJ, Van Ryk D, Xiang S-H, Yang X, Zhang M-Y, Zwick MB, Arthos J (2007) Nature 445(7129):732–737
Biancalana M, Makabe K, Koide A, Koide S (2009) J Mol Biol 385(4):1052–1063
Luchsinger JA, Tang M, Siddiqui M, Shea S, Mayeux R (2004) J Am Geriatr Soc 52(4):540–546
Xue W, Pan D, Yang Y, Liu H, Yao X (2012) Antiviral Res 93(1):126–137
Dodson GG, Lane DP, Verma CS (2008) EMBO Rep 9(2):144–150
Dror RO, Dirks RM, Grossman JP, Xu H, Shaw DE (2012) Annu Rev Biophys 41:429–452
Kaminski GA, Friesner RA, Tirado-Rives J, Jorgensen WL (2001) J Phys Chem B 105(28):6474–6487
Friesner RA, Banks JL, Murphy RB, Halgren TA, Klicic JJ, Mainz DT, Prpasky MP, Knoll EH, Sheelley M, Perry JK, Shaw DE, Francis P, Shenkin PS (2004) J Med Chem 47:1739–1749
Cross JB, Thompson DC, Rai BK, Baber JC, Fan KY, Hu Y, Humblet C (2009) J Chem Inf Model 49(6):1455–1474
Bjorndahl TC, Zhou GP, Liu X, Perez-Pineiro R, Semenchenko V, Saleem F, Acharya S, Bujold A, Sobsey CA, Wishart DS (2011) Biochemistry 50(7):1162–1173
Yin S-M, Zheng Y, Tien P (2003) Protein Expr Purif 32(1):104–109
Bradford MM (1976) Anal Biochem 72(1–2):248–254
Lakowicz JR, Weber G (1973) Biochemistry 12(21):4161–4170
Mehra J, Rechenberg H (2001) The historical development of quantum theory. Volume 1 part 1 the quantum theory of Planck, Einstein, Bohr and Sommerfeld 1900–1925: its foundation and the rise of its difficulties. Springer, New York, pp 1900–1925
Frostell-Karlsson Å, Remaeus A, Roos H, Andersson K, Borg P, Hämäläinen M, Karlsson R (2000) J Med Chem 43(10):1986–1992
Feltis B, Sexton B, Glenn F, Best M, Wilkins M, Davis T (2008) Biosens Bioelectron 23(7):1131–1136
Bocharova OV, Breydo L, Parfenov AS, Salnikov VV, Baskakov IV (2005) J Mol Biol 346(2):645–659
Baskakov IV (2004) J Biol Chem 279(9):7671–7677
Case DA, Cheatham TE, Darden T, Gohlke H, Luo R, Merz KM, Onufriev A, Simmerling C, Wang B, Woods RJ (2005) J Comput Chem 26(16):1668–1688
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JJA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09. Gaussian, Inc., Wallingford
Fox T, Kollman PA (1998) J Phys Chem B 102(41):8070–8079
Bayly CI, Cieplak P, Cornell W, Kollman PA (1993) J Phys Chem 97(40):10269–10280
Wang J, Wolf RM, Caldwell JW, Kollman PA, Case DA (2004) J Comput Chem 25(9):1157–1174
Lindorff-Larsen K, Piana S, Palmo K, Maragakis P, Klepeis JL, Dror RO, Shaw DE (2010) Proteins 78(8):1950–1958
Onufriev A, Bashford D, Case DA (2000) J Phys Chem B 104(15):3712–3720
Tsui V, Case DA (2000) Biopolymers 56(4):275–291
Massova I, Kollman PA (2000) Perspect Drug Discov 18(1):113–135
Sitkoff D, Sharp KA, Honig B (1994) J Phys Chem 98(7):1978–1988
Pearlman DA, Case DA, Caldwell JW, Ross WS, Cheatham TE, DeBolt S, Ferguson D, Seibel G, Kollman P (1995) Comput Phys Commun 91(1):1–41
Baell JB, Holloway GA (2010) J Med Chem 53(7):2719–2740
Rolinski OJ, Amaro M, Birch DJ (2010) Biosens Bioelectron 25(10):2249–2252
Zhou Z, Yan X, Pan K, Chen J, Xie ZS, Xiao GF, Yang FQ, Liang Y (2011) Biophys J 101(6):1483–1492
Zhou Z, Fan JB, Zhu HL, Shewmaker F, Yan X, Chen X, Chen J, Xiao GF, Guo L, Liang Y (2009) J Biol Chem 284(44):30148–30158
Caughey B, Raymond GJ (1993) J Virol 67(2):643–650
Raymond GJ, Olsen EA, Lee KS, Raymond LD, Bryant PK, Baron GS, Caughey WS, Kocisko DA, McHolland LE, Favara C, Langeveld J, Zijiderveld F, Mayer RT, Miller MW, Williams ES, Caughery B (2006) J Virol 80(2):596–604
Acknowledgements
This work was supported by the National Nature Science Foundation of China (Grant No. 21675070) and the Fundamental Research Funds for the Central Universities (Grant No. lzujbky-2016-146).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, L., Wei, W., Jia, WJ. et al. Discovery of small molecules binding to the normal conformation of prion by combining virtual screening and multiple biological activity evaluation methods. J Comput Aided Mol Des 31, 1053–1062 (2017). https://doi.org/10.1007/s10822-017-0086-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-017-0086-6