Abstract
Hydration free energy calculations have become important tests of force fields. Alchemical free energy calculations based on molecular dynamics simulations provide a rigorous way to calculate these free energies for a particular force field, given sufficient sampling. Here, we report results of alchemical hydration free energy calculations for the set of small molecules comprising the 2011 Statistical Assessment of Modeling of Proteins and Ligands challenge. Our calculations are largely based on the Generalized Amber Force Field with several different charge models, and we achieved RMS errors in the 1.4–2.2 kcal/mol range depending on charge model, marginally higher than what we typically observed in previous studies (Mobley et al. in J Phys Chem B 111(9):2242–2254, 2007, J Chem Theory Comput 5(2):350–358, 2009, J Phys Chem B 115:1329–1332, 2011; Nicholls et al. in J Med Chem 51:769–779, 2008; Klimovich and Mobley in J Comput Aided Mol Design 24(4):307–316, 2010). The test set consists of ethane, biphenyl, and a dibenzyl dioxin, as well as a series of chlorinated derivatives of each. We found that, for this set, using high-quality partial charges from MP2/cc-PVTZ SCRF RESP fits provided marginally improved agreement with experiment over using AM1-BCC partial charges as we have more typically done, in keeping with our recent findings (Mobley et al. in J Phys Chem B 115:1329–1332, 2011). Switching to OPLS Lennard–Jones parameters with AM1-BCC charges also improves agreement with experiment. We also find a number of chemical trends within each molecular series which we can explain, but there are also some surprises, including some that are captured by the calculations and some that are not.



Similar content being viewed by others
Notes
These are perhaps not true predictions, as the data is typically available in the literature, but often in relatively obscure places, requiring individual attention from a skilled practitioner to extract hydration free energies and evaluate the accuracy of the data.
the FEP branch of git, commit 9e5241b9dfc8503f887d54640183bc8e397af261
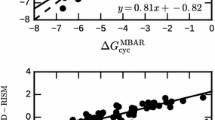
A full table of all computed hydration free energies is available in the Supporting Information (Table 2).
References
Mobley DL, Dumont É, Chodera JD, Dill K (2007) Comparison of charge models for fixed-charge force fields: small-molecule hydration free energies in explicit solvent. J Phys Chem B 111(9):2242–2254
Nicholls A, Mobley DL, Guthrie JP, Chodera JD, Bayly CI, Cooper MD, Pande VS (2008) Predicting small-molecule solvation free energies: an informal blind test for computational chemistry. J Med Chem 51:769–779
Mobley DL, Bayly CI, Cooper MD, Shirts MR, Dill KA (2009) Small molecule hydration free energies in explicit solvent: an extensive test of fixed-charge atomistic simulations. J Chem Theory Comput 5(2):350–358
Klimovich P, Mobley DL (2010) Predicting hydration free energies using all-atom molecular dynamics simulations and multiple starting conformations. J Comput Aided Mol Design 24(4):307–316
Mobley DL, Dumont É, Chodera JD, Dill KA (2011) Comparison of charge models for fixed-charge force fields: small molecule hydration free energies in explicit solvent. J Phys Chem B 115:1329–1332
Villa A, Mark AE (2002) Calculation of the free energy of solvation for neutral analogs of amino acid side chains. J Comput Chem 23(5):548–553
MacCallum JL, Tieleman DP (2003) Calculation of the water-cyclohexane transfer free energies of neutral amino acid side-chain analogs using the OPLS all-atom force field. J Comput Chem 24:1930–1935
Shirts MR, Pitera JW, Swope WC, Pande VS (2003) Extremely precise free energy calculations of amino acid side chain analogs: comparison of common molecular mechanics force fields for proteins. J Chem Phys 119(11):5740
Deng Y, Roux B (2004) Hydration of amino acid side chains: nonpolar and electrostatic contributions calculated from staged molecular dynamics free energy simulations with explicit water molecules. J Phys Chem B 108:16567–16576
Shirts MR, Pande VS (2005) Solvation free energies of amino acid side chain analogs for common molecular mechanics water models. J Chem Phys 122:134508
Hess B, van der Vegt NFA (2006) Hydration thermodynamic properties of amino acid analogues: a systematic comparison of biomolecular force fields and water models. J Phys Chem B 110:17616–17626
Mobley DL, Bayly CI, Cooper MD, Dill KA, Dill KA (2009) Predictions of hydration free energies from all-atom molecular dynamics simulations. J Phys Chem B 113:4533–4537
Shivakumar D, Deng Y, Roux B (2009) Computations of absolute solvation free energies of small molecules using explicit and implicit solvent model. J Chem Theory Comput 5(4):919–930
Shivakumar D, Williams J, Wu Y, Damm W, Shelley J, Sherman W (2010) Prediction of absolute solvation free energies using molecular dynamics free energy perturbation and the OPLS force field. J Chem Theory Comput 6:1509–1519
Knight JL, Brooks CL III (2011) Surveying implicit solvent models for estimating small molecule absolute hydration free energies. J Comput Chem 32:2909–2923
Deng Y, Roux B (2006) Calculation of standard binding free energies: aromatic molecules in the T4 lysozyme L99A mutant. J Chem Theory Comput 2:1255–1273
Mobley DL, Graves AP, Chodera JD, McReynolds A, Shoichet BK, Dill KA (2007) Predicting absolute ligand binding free energies to a simple model site. J Mol Biol 371:1118–1134
Boyce SE, Mobley DL, Rocklin GJ, Graves AP, Dill KA, Shoichet BK (2009) Predicting ligand binding affinity with alchemical free energy methods in a polar model binding site. J Mol Biol 394:747–763
Gallicchio E, Lapelosa M, Levy RM (2010) Binding energy distribution analysis method (BEDAM) for estimation of Protein−Ligand binding affinities. J Chem Theory Comput 6:2961–2977
Chodera JD, Mobley DL, Shirts MR, Dixon RW, Branson K, Pande VS (2011) Alchemical free energy methods for drug discovery: progress and challenges. Curr Opin Struct Biol 21:150–160
Li J, Zhu T, Hawkins GD, Winget P, Liotard DA, Truhlar DG (1999, June) Extension of the platform of applicability of SM5.42R universal solvation model
Guthrie JP (2009) A blind challenge for computational solvation free energies: introduction and overview. J Phys Chem B 113(14):4501–4507
Geballe MT, Skillman AG, Nicholls A, Guthrie JP, Taylor PJ (2010) The SAMPL2 blind prediction challenge: introduction and overview. J Comput Aided Mol Design 24:259–279
Wang J, Wolf R, Caldwell J, Kollman P, Case D (2004) Development and testing of a general amber force field. J Comput Chem 25(9):1157–1174
Wang J, Wang W, Kollman P, Case DA (2006) Automatic atom type and bond type perception in molecular mechanical calculations. J Mol Graph Model 25:247–260
Jakalian A, Bush B, Jack D, Bayly CI (2000) Fast, efficient generation of high-quality atomic charges. AM1-BCC model: I. Method. J Comput Chem 21(2):132–146
Jakalian A, Jack D, Bayly CI (2002) Fast, efficient generation of high-quality atomic charges. AM1-BCC model: II. Parameterization and validation. J Comput Chem 23(16):1623–1641
Bayly CI, Cieplak P, Cornell WD, Kollman P (1993) A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges: the RESP model. J Phys Chem 97(40):10269–10280
Dixon R (2011, March) OEAntechamber: assign and generate AMBER atom types and structural parameters. http://SimTk.org—OEAntechamber: assign and generate AMBER atom types and structural parameters
Jorgensen WL, Maxwell DS, Tirado-Rives J (1996) Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J Am Chem Soc 118:11225–11236
Shirts MR, Chodera JD (2008) Statistically optimal analysis of samples from multiple equilibrium states. J Chem Phys 129:124105. http://dx.doi.org/10.1063/1.2978177 See code at: http://simtk.org/home/pymbar
Jorgensen WL, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) Comparison of simple potential functions for simulating liquid water. J Chem Phys 79(2):926
Shirts MR, Mobley DL, Chodera JD, Pande VS (2007) Accurate and efficient corrections for missing dispersion interactions in molecular simulations. J Phys Chem B 111:13052–13063
Hess B, Bekker H, Berendsen HJC, Fraaije JGEM (1997) LINCS: A linear constraint solver for molecular simulations. J Comput Chem 18:1463–1472
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kuden KN, Burant JC, Milliam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomberts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng Cy, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004, September) Gaussian03, c.02 edn. Gaussian, Inc., Wallingford
Cancès E, Mennucci B, Tomasi J (1997) A new integral equation formalism for the polarizable continuum model: theoretical background and applications to isotropic and anisotropic dielectrics. J Chem Phys 107(8):3032
Mennucci B, Cancès E, Tomasi J (1997) Evaluation of solvent effects in isotropic and anisotropic dielectrics and in ionic solutions with a unified integral equation method: theoretical bases, computational implementation, and numerical applications. J Phys Chem B 101:10506–10517
Horn HW, Swope WC, Pitera JW, Madura JD, Dick TJ, Hura GL, Head-Gordon T (2004) Development of an improved four-site water model for biomolecular simulations: TIP4P-Ew. J Chem Phys 120(20):9665
Karamertzanis PG, Raiteri P, Galindo A (2010) The use of anisotropic potentials in modeling water and free energies of hydration. J Chem Theory Comput 6:1590–1607
Kemp DD, Gordon MS (2008) An interpretation of the enhancement of the water dipole moment due to the presence of other water molecules. J Phys Chem A 112:4885–4894
Wang J, Tingjun H (2011) Application of molecular dynamics simulations in molecular property prediction I: density and heat of vaporization. J Chem Theory Comput 7:2151–2165
Kehoe C (2011, July) Personal communication. Discussion of computed hydration free energies via Skype and e-mail
Kehoe CW, Fennell CJ, Dill KA (2011) Testing the semi-explicit assembly solvation model in the SAMPL3 community blind test. J Comput Aided Mol Design. doi:10.1007/s10822-011-9536-8
Geballe M (2011, August) Overview of SAMPL hydration challenge. In: SAMPL 2011, Stanford
Case DA, Cheatham TE, Darden T, Gohlke H, Luo R, Merz KM, Onufriev A, Simmerling C, Wang B, Woods RJ (2005) The amber biomolecular simulation programs. J Comput Chem 26:1668–1688
Guthrie JP (2011, August) Overview of experimental data for hydration. In: SAMPL 2011, Stanford
Cerutti DS, Baker NA, McCammon JA (2007) Solvent reaction field potential inside an uncharged globular protein: a bridge between implicit and explicit solvent models?. J Chem Phys 127(15):155101
Acknowledgments
DLM and SL acknowledge the Louisiana Board of Regents Research Competitiveness and Research Enhancement Subprograms as well as the Louisiana Optical Network Initiative (supported by the Louisiana Board of Regents Post-Katrina Support Fund Initiative grant LEQSF(2007-12)- ENH-PKSFI-PRS-01), and the National Science Foundation under NSF EPSCoR Cooperative Agreement No. EPS-1003897 with additional support from the Louisiana Board of Regents. DSC acknowledges the help of David A. Case (Rutgers) in developing the solvent reaction field QM calculations, and the support of NIH grants RR12255 and RR05969 (to Dr. Case).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mobley, D.L., Liu, S., Cerutti, D.S. et al. Alchemical prediction of hydration free energies for SAMPL. J Comput Aided Mol Des 26, 551–562 (2012). https://doi.org/10.1007/s10822-011-9528-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-011-9528-8