Abstract
Purpose
To identify the genetic causes of multiple morphological abnormalities in sperm flagella (MMAF) and male infertility in patients from two unrelated Han Chinese families.
Methods
Whole-exome sequencing was conducted using blood samples from the two individuals with MMAF and male infertility. Hematoxylin and eosin staining and scanning electron microscopy were performed to evaluate sperm morphology. Ultrastructural and immunostaining analyses of the spermatozoa were performed. The HEK293T cells were used to confirm the pathogenicity of the variants.
Results
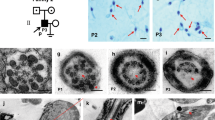
We identified two novel homozygous missense ARMC2 variants: c.314C > T: p.P105L and c.2227A > G: p.N743D. Both variants are absent or rare in the human population genome data and are predicted to be deleterious. In vitro experiments indicated that both ARMC2 variants caused a slightly increased protein expression. ARMC2-mutant spermatozoa showed multiple morphological abnormalities (bent, short, coiled, absent, and irregular) in the flagella. In addition, the spermatozoa of the patients revealed a frequent absence of the central pair complex and disrupted axonemal ultrastructure.
Conclusion
We identified two novel ARMC2 variants that caused male infertility and MMAF in Han Chinese patients. These findings expand the mutational spectrum of ARMC2 and provide insights into the complex causes and pathogenesis of MMAF.




Similar content being viewed by others
Data availability
The whole-exome sequencing data in this study have not been deposited in public repository because of privacy and ethical restrictions but are available from the corresponding author on reasonable request.
References
Agarwal A, Baskaran S, Parekh N, Cho C-L, Henkel R, Vij S, et al. Male infertility. The Lancet. 2021;397(10271):319–33. https://doi.org/10.1016/s0140-6736(20)32667-2.
Coutton C, Escoffier J, Martinez G, Arnoult C, Ray PF. Teratozoospermia: spotlight on the main genetic actors in the human. Hum Reprod Update. 2015;21(4):455–85. https://doi.org/10.1093/humupd/dmv020.
Ben Khelifa M, Coutton C, Zouari R, Karaouzene T, Rendu J, Bidart M, et al. Mutations in DNAH1, which encodes an inner arm heavy chain dynein, lead to male infertility from multiple morphological abnormalities of the sperm flagella. Am J Hum Genet. 2014;94(1):95–104. https://doi.org/10.1016/j.ajhg.2013.11.017.
Touré A, Martinez G, Kherraf Z-E, Cazin C, Beurois J, Arnoult C, et al. The genetic architecture of morphological abnormalities of the sperm tail. Hum Genet. 2020;140(1):21–42. https://doi.org/10.1007/s00439-020-02113-x.
Wang WL, Tu CF, Tan YQ. Insight on multiple morphological abnormalities of sperm flagella in male infertility: what is new? Asian journal of andrology. 2020;22(3):236–45. https://doi.org/10.4103/aja.aja_53_19.
Amiri-Yekta A, Coutton C, Kherraf Z-E, Karaouzène T, Le Tanno P, Sanati MH, et al. Whole-exome sequencing of familial cases of multiple morphological abnormalities of the sperm flagella (MMAF) reveals newDNAH1mutations. Hum Reprod. 2016;31(12):2872–80. https://doi.org/10.1093/humrep/dew262.
Coutton C, Vargas AS, Amiri-Yekta A, Kherraf Z-E, Ben Mustapha SF, Le Tanno P, et al. Mutations in CFAP43 and CFAP44 cause male infertility and flagellum defects in Trypanosoma and human. Nature Communications 2018;9(1) https://doi.org/10.1038/s41467-017-02792-7.
Wang W, Tu C, Nie H, Meng L, Li Y, Yuan S, et al. Biallelic mutations in CFAP65 lead to severe asthenoteratospermia due to acrosome hypoplasia and flagellum malformations. J Med Genet. 2019;56(11):750–7. https://doi.org/10.1136/jmedgenet-2019-106031.
Tu C, Cong J, Zhang Q, He X, Zheng R, Yang X, et al. Bi-allelic mutations of DNAH10 cause primary male infertility with asthenoteratozoospermia in humans and mice. Am J Hum Genet. 2021;108(8):1466–77. https://doi.org/10.1016/j.ajhg.2021.06.010.
Liu W, He X, Yang S, Zouari R, Wang J, Wu H, et al. Bi-allelic mutations in TTC21A induce asthenoteratospermia in humans and mice. Am J Hum Genet. 2019;104(4):738–48. https://doi.org/10.1016/j.ajhg.2019.02.020.
Lorès P, Dacheux D, Kherraf Z-E, Nsota Mbango J-F, Coutton C, Stouvenel L, et al. Mutations in TTC29, encoding an evolutionarily conserved axonemal protein, result in asthenozoospermia and male infertility. Am J Hum Genet. 2019;105(6):1148–67. https://doi.org/10.1016/j.ajhg.2019.10.007.
Coates JC. Armadillo repeat proteins: beyond the animal kingdom. Trends Cell Biol. 2003;13(9):463–71. https://doi.org/10.1016/s0962-8924(03)00167-3.
Tewari R, Bailes E, Bunting KA, Coates JC. Armadillo-repeat protein functions: questions for little creatures. Trends Cell Biol. 2010;20(8):470–81. https://doi.org/10.1016/j.tcb.2010.05.003.
Peifer M, Berg S, Reynolds AB. A repeating amino acid motif shared by proteins with diverse cellular roles. Cell. 1994;76(5):789–91. https://doi.org/10.1016/0092-8674(94)90353-0.
Coutton C, Martinez G, Kherraf Z-E, Amiri-Yekta A, Boguenet M, Saut A, et al. Bi-allelic mutations in ARMC2 lead to severe astheno-teratozoospermia due to sperm flagellum malformations in humans and mice. Am J Hum Genet. 2019;104(2):331–40. https://doi.org/10.1016/j.ajhg.2018.12.013.
Khan I, Dil S, Zhang H, Zhang B, Khan T, Zeb A, et al. A novel stop-gain mutation in ARMC2 is associated with multiple morphological abnormalities of the sperm flagella. Reprod Biomed Online. 2021;43(5):913–9. https://doi.org/10.1016/j.rbmo.2021.07.021.
Wang J, Liu X, Zhang C, Xu Y, Wang W, Li H, et al. Patient with multiple morphological abnormalities of sperm flagella caused by a novel ARMC2 mutation has a favorable pregnancy outcome from intracytoplasmic sperm injection. J Assist Reprod Genet. 2022;39(7):1673–81. https://doi.org/10.1007/s10815-022-02516-x.
Martinez G, Kherraf ZE, Zouari R, Fourati Ben Mustapha S, Saut A, Pernet-Gallay K, et al. Whole-exome sequencing identifies mutations in FSIP2 as a recurrent cause of multiple morphological abnormalities of the sperm flagella. Hum reprod 2018;33(10):1973–84. https://doi.org/10.1093/humrep/dey264.
Wang Y, Yang J, Jia Y, Xiong C, Meng T, Guan H, et al. Variability in the morphologic assessment of human sperm: use of the strict criteria recommended by the World Health Organization in 2010. Fertil Steril. 2014;101(4):945–9. https://doi.org/10.1016/j.fertnstert.2013.12.047.
Liu C, Tu C, Wang L, Wu H, Houston BJ, Mastrorosa FK, et al. Deleterious variants in X-linked CFAP47 induce asthenoteratozoospermia and primary male infertility. Am J Hum Genet. 2021;108(2):309–23. https://doi.org/10.1016/j.ajhg.2021.01.002.
Tu C, Cong J, Zhang Q, He X, Zheng R, Yang X, et al. Bi-allelic mutations of DNAH10 cause primary male infertility with asthenoteratozoospermia in humans and mice. Am J Hum Genet. 2021;108(8):1466–77. https://doi.org/10.1016/j.ajhg.2021.06.010.
Zhang G, Li D, Tu C, Meng L, Tan Y, Ji Z, et al. Loss-of-function missense variant of AKAP4 induced male infertility through reduced interaction with QRICH2 during sperm flagella development. Hum Mol Genet. 2021;31(2):219–31. https://doi.org/10.1093/hmg/ddab234.
UniProt Consortium T. UniProt: the universal protein knowledgebase. Nucleic Acids Res. 2018;46(5):2699. https://doi.org/10.1093/nar/gky092.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med: J Am Coll Med Genet. 2015;17(5):405–24. https://doi.org/10.1038/gim.2015.30.
Gerstein MB, Kundaje A, Hariharan M, Landt SG, Yan KK, Cheng C, et al. Architecture of the human regulatory network derived from ENCODE data. Nature. 2012;489(7414):91–100. https://doi.org/10.1038/nature11245.
Lizio M, Harshbarger J, Abugessaisa I, Noguchi S, Kondo A, Severin J, et al. Update of the FANTOM web resource: high resolution transcriptome of diverse cell types in mammals. Nucleic Acids Res. 2017;45(D1):D737–43. https://doi.org/10.1093/nar/gkw995.
Zhang Z, Sapiro R, Kapfhamer D, Bucan M, Bray J, Chennathukuzhi V, et al. A sperm-associated WD repeat protein orthologous to Chlamydomonas PF20 associates with Spag6, the mammalian orthologue of Chlamydomonas PF16. Mol Cell Biol. 2002;22(22):7993–8004. https://doi.org/10.1128/MCB.22.22.7993-8004.2002.
Tan C, Meng L, Lv M, He X, Sha Y, Tang D, et al. Bi-allelic variants in DNHD1 cause flagellar axoneme defects and asthenoteratozoospermia in humans and mice. Am J Hum Genet. 2022;109(1):157–71. https://doi.org/10.1016/j.ajhg.2021.11.022.
Acknowledgements
The authors wish to sincerely thank all the families and individuals who participated in this study. The support from the technical platform and clinicians involved at the Jiangxi Maternal and Child Health Hospital and the Reproductive and Genetic Hospital of CITIC-Xiangya is gratefully acknowledged.
Funding
This work was supported by the National Natural Science Foundation of China (82101961 to CF.T.) and the outstanding Youth Foundation of Hunan Provincial Natural Science Foundation of China (2023JJ20080 to CF.T.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, S., Liu, Q., Su, L. et al. Identification of novel homozygous asthenoteratospermia-causing ARMC2 mutations associated with multiple morphological abnormalities of the sperm flagella. J Assist Reprod Genet 41, 1297–1306 (2024). https://doi.org/10.1007/s10815-024-03087-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-024-03087-9