Abstract
Purpose
To evaluate the cytogenetic risk of assisted reproductive technology (ART) by comparing the incidence of de novo chromosomal abnormalities between fetuses conceived via in vitro fertilization/intracytoplasmic sperm injection (IVF/ICSI) and natural conception.
Materials and methods
Prenatal invasive diagnostic testing (amniocentesis and cytogenetic analysis) was performed on 1496 fetuses conceived via IVF/ICSI (IVF/ICSI group) and 1396 fetuses from natural conception (NC group). The incidence of de novo chromosomal abnormalities (including aneuploidy and chromosomal structure abnormalities) was used to evaluate the cytogenetic risk of ART. For statistical analysis, χ2-test was used for binary dependent variable. The significance level was P < 0.05 and confidence interval was 95%.
Result(s)
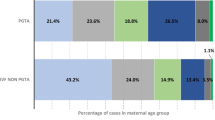
The IVF/ICSI group displayed a modest increase in the overall de novo chromosomal abnormality rate compared with that in the NC group but with no statistical significance (6.75% vs. 6.16%; χ2 = 0.42, P > 0.05). The incidence of abnormal karyotypes was also not significantly different between the IVF/ICSI and NC groups in different maternal ages, including ≥ 35 years group (7.55% vs. 9.60%, χ2 = 1.40, P > 0.05) and < 35 years group (6.20% vs. 4.54%, χ2 = 2.51, P > 0.05). Moreover, there was no difference in the proportion of aneuploid and structural abnormalities in detected karyotypes between the IVF/ICSI and NC groups. Logistic regression analysis showed no significant association between the method of pregnancy and de novo chromosomal abnormalities (odds ratio (OR) 1.03; 95% CI 0.71–1.50; P = 0.86) after adjusting for other confounding factors.
Conclusion(s).
Fetuses conceived via IVF/ICSI had a slight but not statistically significant increase in de novo abnormal karyotypes compared to those in naturally conceived fetuses. Our findings indicate no significant association between de novo fetal chromosomal abnormalities and the pregnancy method in high-risk pregnancies in the second trimester. For these pregnancies with a high risk but with a normal karyotype, further genetic testing is required for diagnosis.
Similar content being viewed by others
References
Steptoe PC, Edwards RG. Birth after the reimplantation of a human embryo. Lancet. 1978;2(8085):366.
Brison DR, Roberts SA, Kimber SJ. How should we assess the safety of IVF technologies? Reprod Biomed Online. 2013;27(6):710–21.
Wdowiak A, Wdowiak E, Bojar I. Improving the safety of the embryo and the patient during in vitro fertilization procedures. Wideochir Inne Tech Maloinwazyjne. 2016;11(3):137–43.
Belva F, De Schrijver F, Tournaye H, Liebaers I, Devroey P, Haentjens P, et al. Neonatal outcome of 724 children born after ICSI using non-ejaculated sperm. Hum Reprod. 2011;26(7):1752–8.
Stokes PJ, Hawkhead JA, Fawthrop RK, Picton HM, Sharma V, Leese HJ, et al. Metabolism of human embryos following cryopreservation: implications for the safety and selection of embryos for transfer in clinical IVF. Hum Reprod. 2007;22(3):829–35.
Angell RR, Aitken RJ, van Look PF, Lumsden MA, Templeton AA. Chromosome abnormalities in human embryos after in vitro fertilization. Nature. 1983;303(5915):336–8.
Jozwiak EA, Ulug U, Mesut A, Erden HF, Bahceci M. Prenatal karyotypes of fetuses conceived by intracytoplasmic sperm injection. Fertil Steril. 2004;82(3):628–33.
Westergaard HB, Johansen AM, Erb K, Andersen AN. Danish national in-vitro fertilization registry 1994 and 1995: a controlled study of births, malformations and cytogenetic findings. Hum Reprod. 1999;14(7):1896–902.
Zhang X, Wang Y, Zhao N, Liu P, Huang J. Variations in chromosomal aneuploidy rates in IVF blastocysts and early spontaneous abortion chorionic villi. J Assist Reprod Genet. 2020;37(3):527–37.
Kim JW, Lee WS, Yoon TK, Seok HH, Cho JH, Kim YS, et al. Chromosomal abnormalities in spontaneous abortion after assisted reproductive treatment. BMC Med Genet. 2010;11:153.
Lathi RB, Milki AA. Rate of aneuploidy in miscarriages following in vitro fertilization and intracytoplasmic sperm injection. Fertil Steril. 2004;81(5):1270–2.
Bingol B, Abike F, Gedikbasi A, Tapisiz OL, Gunenc Z. Comparison of chromosomal abnormality rates in ICSI for non-male factor and spontaneous conception. J Assist Reprod Genet. 2012;29(1):25–30.
Belva F, Bonduelle M, Buysse A, Van den Bogaert A, Hes F, Roelants M, et al. Chromosomal abnormalities after ICSI in relation to semen parameters: results in 1114 fetuses and 1391 neonates from a single center. Hum Reprod. 2020;35(9):2149–62.
Vicic A, Roje D, Strinic T, Stipoljev F. Trisomy 1 in an early pregnancy failure. Am J Med Genet A. 2008;146A(18):2439–41.
Findikli N, Kahraman S, Saglam Y, Beyazyurek C, Sertyel S, Karlikaya G, et al. Embryo aneuploidy screening for repeated implantation failure and unexplained recurrent miscarriage. Reprod Biomed Online. 2006;13(1):38–46.
Wolczynski S, Kulikowski M, Szamatowicz M. Triploidy as a cause of failure in human reproduction. Ginekol Pol. 1993;64(3):154–60.
Heller JH. Human chromosome abnormalities as related to physical and mental dysfunction. J Hered. 1969;60(5):239–48.
Nucaro A, Pisano T, Chillotti I, Montaldo C, Pruna D. Chromosome 8p23.2-pter: a critical region for mental retardation, autism and epilepsy? Clin Genet. 2011;79(4):394–395; author reply 396.
Mariner R, Jackson AW 3rd, Levitas A, Hagerman RJ, Braden M, McBogg PM, et al. Autism, mental retardation, and chromosomal abnormalities. J Autism Dev Disord. 1986;16(4):425–40.
Driscoll DA, Gross S. Clinical practice. Prenatal screening for aneuploidy. N Engl J Med. 2009;360(24):2556–62.
Wurfel W, Haas-Andela H, Krusmann G, Rothenaicher M, Hirsch P, Kwapisz HK, et al. Prenatal diagnosis by amniocentesis in 82 pregnancies after in vitro fertilization. Eur J Obstet Gynecol Reprod Biol. 1992;44(1):47–52.
In’t Veld PA, van Opstal D, Van den Berg C, Van Ooijen M, Brandenburg H, Pijpers L, et al. Increased incidence of cytogenetic abnormalities in chorionic villus samples from pregnancies established by in vitro fertilization and embryo transfer (IVF-ET). Prenat Diagn. 1995;15(10):975–980.
Lam R, Ma S, Robinson WP, Chan T, Yuen BH. Cytogenetic investigation of fetuses and infants conceived through intracytoplasmic sperm injection. Fertil Steril. 2001;76(6):1272–5.
Gjerris AC, Loft A, Pinborg A, Christiansen M, Tabor A. Prenatal testing among women pregnant after assisted reproductive techniques in Denmark 1995–2000: a national cohort study. Hum Reprod. 2008;23(7):1545–52.
Bonduelle M, Van Assche E, Joris H, Keymolen K, Devroey P, Van Steirteghem A, et al. Prenatal testing in ICSI pregnancies: incidence of chromosomal anomalies in 1586 karyotypes and relation to sperm parameters. Hum Reprod. 2002;17(10):2600–14.
Xiao H, Yang YL, Zhang CY, Liao EJ, Zhao HR, Liao SX. Karyotype analysis with amniotic fluid in 12365 pregnant women with indications for genetic amniocentesis and strategies of prenatal diagnosis. J Obstet Gynaecol. 2016;36(3):293–6.
Meschede D, Horst J. Sex chromosomal anomalies in pregnancies conceived through intracytoplasmic sperm injection: a case for genetic counselling. Hum Reprod. 1997;12(6):1125–7.
Miryounesi M, Dianatpour M, Shadmani Z, Ghafouri-Fard S. Report of a case with trisomy 9 mosaicism. Iran J Med Sci. 2016;41(3):249–52.
Li M, Glass J, Du X, Dubbs H, Harr MH, Falk M, et al. Trisomy 9 mosaic syndrome: sixteen additional patients with new and/or less commonly reported features, literature review, and suggested clinical guidelines. Am J Med Genet A. 2021;185(8):2374–83.
Wang C, Chen Y, Zhao J, Xia Y. Prenatal diagnosis and genetic counseling of low-level trisomy 9 mosaicism with a favorable outcome. Taiwan J Obstet Gynecol. 2020;59(5):786–7.
Snijders RJ, Sebire NJ, Nicolaides KH. Maternal age and gestational age-specific risk for chromosomal defects. Fetal Diagn Ther. 1995;10(6):356–67.
Wu X, Li Y, Lin N, Xie X, Su L, Cai M, et al. Chromosomal microarray analysis for pregnancies with abnormal maternal serum screening who undergo invasive prenatal testing. J Cell Mol Med. 2021;25(13):6271–9.
Luo X, Zhu H, Wang L, Xiao B, Fan Y, Ye H, et al. Chromosomal microarray analysis in fetuses with high-risk prenatal indications: a retrospective study in China. Taiwan J Obstet Gynecol. 2021;60(2):299–304.
Petrovski S, Aggarwal V, Giordano JL, Stosic M, Wou K, Bier L, et al. , Anyane-Yeboa K, Miller R, Levy B, Goldstein DB, Wapner RJ. Whole-exome sequencing in the evaluation of fetal structural anomalies: a prospective cohort study. Lancet. 2019 23;393(10173):758–767.
Acknowledgements
We thank all members of the Prenatal Diagnosis Center of Reproductive and Genetic Hospital of CITIC–Xiangya for their invaluable efforts.
Funding
This work was supported by grants from the National Key Research & Developmental Program of China (2018YFC1004901), the National Natural Science Foundation of China (82171608 and 81971447), the Key Grant of Prevention and Treatment of Birth Defect from Hunan Province (2019SK1012), Hunan Provincial Grant for Innovative Province Construction (2019SK4012), the Research Grant of CITIC-Xiangya (YNXM-202004, YNXM-202006), and the Changsha Municipal Natural Science Foundation (YWXM-202209).
Author information
Authors and Affiliations
Contributions
SM.Y and LL.G conceived the study and drafted the manuscript; DH.C performed chromosome analysis; XR.L, H.H, and L.H designed the study and recruited the patients; GX.L, G.L, F.G, and YQ.T designed the study and revised the manuscript. YQ.T approved the final version of the manuscript for submission with input from all the authors.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yuan, S., Guo, L., Cheng, D. et al. The de novo aberration rate of prenatal karyotype was comparable between 1496 fetuses conceived via IVF/ICSI and 1396 fetuses from natural conception. J Assist Reprod Genet 39, 1683–1689 (2022). https://doi.org/10.1007/s10815-022-02500-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-022-02500-5