Abstract
Purpose
To evaluate the unknown genetic causes of teratozoospermia, and determine the pathogenicity of candidate variants.
Methods
A primary infertile patient and his family members were recruited in the West China Second University Hospital of Sichuan University. Whole-exome sequencing was performed to identify causative genes in a man with teratozoospermia. Immunofluorescence staining and western blotting were applied to assess the pathogenicity of the identified variant. Intracytoplasmic sperm injection (ICSI) was used to assist fertilization for the patient with teratozoospermia.
Results
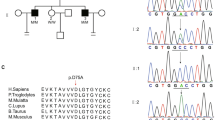
We performed whole-exome sequencing (WES) and detected a novel homozygous frameshift mutation of c.335_336del [p.E112Vfs*3] in DNAJB13 on a primary infertile male patient. Intriguingly, we identified abnormal sperm morphology in this patient, with recurrent respiratory infections and chronic cough. Furthermore, we confirmed that this mutation resulted in negative effects on DNAJB13 expression in the spermatozoa of the affected individual, causing ultrastructural defects in his sperm. Remarkably, our staining revealed that DNAJB13 was expressed in the cytoplasm of primary germ cells and in the flagella of spermatids during spermiogenesis in humans and mice. Finally, we are the first group to report a favorable prognosis using ICSI for a patient carrying this DNAJB13 mutation.
Conclusion
Our study revealed a novel homozygous frameshift mutation of c.335_336del [p.E112Vfs*3] in DNAJB13 involved in teratozoospermia phenotype. Our study greatly expands the spectrum of limited DNAJB13 mutations, and is expected to provide a better understanding of genetic counseling diagnoses and subsequent treatment of male infertility.



Similar content being viewed by others
Data availability
Data are available on request from the corresponding author.
References
Krausz C, Riera-Escamilla A. Genetics of male infertility. Nat Rev Urol. 2018;15(6):369–84. https://doi.org/10.1038/s41585-018-0003-3.
Coutton C, Escoffier J, Martinez G, Arnoult C, Ray PF. Teratozoospermia: spotlight on the main genetic actors in the human. Hum Reprod Update. 2015;21(4):455–85. https://doi.org/10.1093/humupd/dmv020.
Zhu F, Wang F, Yang X, Zhang J, Wu H, Zhang Z, et al. Biallelic SUN5 mutations cause autosomal-recessive acephalic spermatozoa syndrome. Am J Hum Genet. 2016;99(4):942–9. https://doi.org/10.1016/j.ajhg.2016.11.002.
Wang X, Jin H, Han F, Cui Y, Chen J, Yang C, et al. Homozygous DNAH1 frameshift mutation causes multiple morphological anomalies of the sperm flagella in Chinese. Clin Genet. 2017;91(2):313–21. https://doi.org/10.1111/cge.12857.
Tang S, Wang X, Li W, Yang X, Li Z, Liu W, et al. Biallelic mutations in CFAP43 and CFAP44 cause male infertility with multiple morphological abnormalities of the sperm flagella. Am J Hum Genet. 2017;100(6):854–64. https://doi.org/10.1016/j.ajhg.2017.04.012.
Liu C, Tu C, Wang L, Wu H, Houston BJ, Mastrorosa FK, et al. Deleterious variants in X-linked CFAP47 induce asthenoteratozoospermia and primary male infertility. Am J Hum Genet. 2021;108(2):309–23. https://doi.org/10.1016/j.ajhg.2021.01.002.
Li W, Wu H, Li F, Tian S, Kherraf ZE, Zhang J, et al. Biallelic mutations in CFAP65 cause male infertility with multiple morphological abnormalities of the sperm flagella in humans and mice. J Med Genet. 2020;57(2):89–95. https://doi.org/10.1136/jmedgenet-2019-106344.
He X, Liu C, Yang X, Lv M, Ni X, Li Q, et al. Bi-allelic loss-of-function variants in CFAP58 cause flagellar axoneme and mitochondrial sheath defects and asthenoteratozoospermia in humans and mice. Am J Hum Genet. 2020;107(3):514–26. https://doi.org/10.1016/j.ajhg.2020.07.010.
Ben Khelifa M, Zouari R, Harbuz R, Halouani L, Arnoult C, Lunardi J, et al. A new AURKC mutation causing macrozoospermia: implications for human spermatogenesis and clinical diagnosis. Mol Hum Reprod. 2011;17(12):762–8. https://doi.org/10.1093/molehr/gar050.
Elinati E, Kuentz P, Redin C, Jaber S, Vanden Meerschaut F, Makarian J, et al. Globozoospermia is mainly due to DPY19L2 deletion via non-allelic homologous recombination involving two recombination hotspots. Hum Mol Genet. 2012;21(16):3695–702. https://doi.org/10.1093/hmg/dds200.
Jha P, Laskar S, Dubey S, Bhattacharyya MK, Bhattacharyya S. Plasmodium Hsp40 and human Hsp70: a potential cochaperone-chaperone complex. Mol Biochem Parasitol. 2017;214:10–3. https://doi.org/10.1016/j.molbiopara.2017.03.003.
Guan J, Yuan L. A heat-shock protein 40, DNAJB13, is an axoneme-associated component in mouse spermatozoa. Mol Reprod Dev. 2008;75(9):1379–86. https://doi.org/10.1002/mrd.20874.
Guan J, Kinoshita M, Yuan L. Spatiotemporal association of DNAJB13 with the annulus during mouse sperm flagellum development. BMC Dev Biol. 2009;9:23. https://doi.org/10.1186/1471-213X-9-23.
Li W, Liu G. DNAJB13, a type II HSP40 family member, localizes to the spermatids and spermatozoa during mouse spermatogenesis. BMC Dev Biol. 2014;14:38. https://doi.org/10.1186/s12861-014-0038-5.
Guan J, Ekwurtzel E, Kvist U, Hultenby K, Yuan L. DNAJB13 is a radial spoke protein of mouse ‘9+2’ axoneme. Reprod Domest Anim. 2010;45(6):992–6. https://doi.org/10.1111/j.1439-0531.2009.01473.x.
El Khouri E, Thomas L, Jeanson L, Bequignon E, Vallette B, Duquesnoy P, et al. Mutations in DNAJB13, encoding an HSP40 family member, cause primary ciliary dyskinesia and male infertility. Am J Hum Genet. 2016;99(2):489–500. https://doi.org/10.1016/j.ajhg.2016.06.022.
Shen Y, Zhang F, Li F, Jiang X, Yang Y, Li X, et al. Loss-of-function mutations in QRICH2 cause male infertility with multiple morphological abnormalities of the sperm flagella. Nat Commun. 2019;10(1):433 Published 2019 Jan 25. 10.1038/s41467-018-08182-x.
Liu G, Lu GX, Xing XW. Molecular cloning of TSARG6 gene related to apoptosis in human spermatogenic cells. Acta Biochim Biophys Sin (Shanghai). 2004;36(2):93–8. https://doi.org/10.1093/abbs/36.2.93.
Oji A, Noda T, Fujihara Y, Miyata H, Kim YJ, Muto M, et al. CRISPR/Cas9 mediated genome editing in ES cells and its application for chimeric analysis in mice. Sci Rep. 2016;6:31666. https://doi.org/10.1038/srep31666.
Li WN, Zhu L, Jia MM, Yin SL, Lu GX, Liu G. Missense mutation in DNAJB13 gene correlated with male fertility in asthenozoospermia. Andrology. 2020;8(2):299–306. https://doi.org/10.1111/andr.12685.
Omran H, Häffner K, Völkel A, Kuehr J, Ketelsen UP, Ross UH, et al. Homozygosity mapping of a gene locus for primary ciliary dyskinesia on chromosome 5p and identification of the heavy dynein chain DNAH5 as a candidate gene. Am J Respir Cell Mol Biol. 2000;23:696–702. https://doi.org/10.1165/ajrcmb.23.5.4257.
Olbrich H, Häffner K, Kispert A, Völkel A, Volz A, Sasmaz G, et al. Mutations in DNAH5 cause primary ciliary dyskinesia and randomization of left-right asymmetry. Nat Genet. 2002;30(2):143–4. https://doi.org/10.1038/ng817.
Guichard C, Harricane MC, Lafitte JJ, Godard P, Zaegel M, Tack V, et al. Loss-of-function mutations in a human gene related to Chlamydomonas reinhardtii dynein IC78 result in primary ciliary dyskinesia. Am J Hum Genet. 1999;65(6):1508–19. https://doi.org/10.1086/302683.
Acknowledgements
We thank the patient and his family for their interest and cooperation. The authors would like to thank Analytical and Testing Center of Sichuan University for the morphology characterization and the authors are grateful to Guiping Yuan for her help of TEM images and Yi He for his help of SEM images.
Author information
Authors and Affiliations
Contributions
Y.S. designed and supervised the study experiments. Y.Y. collected data and conducted the clinical evaluations. J.L. and C.J. performed TEM and SEM. M.L. wrote the first article draft; M.L., Y.Z., and Y.S. performed immunofluorescence staining and Minigene assay. All authors revised and approved the article.
Corresponding authors
Ethics declarations
Ethics approval
The study had been approved by the Ethics Committee of West China Second University Hospital, Sichuan University.
Consent to participate
Obtained
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Table S1
(XLSX 88 kb)
Fig. S1
Chest X-ray of the normal control and patient. The patient with the DNAJB13 mutant showed no situs -inversus phenotype but increased and disordered lung texture. (JPG 182 kb)

Fig. S2
Results of minigene assay. Pattern diagram showing no harmful splicing effects caused by the DNAJB13 mutation. a Agarose gel electrophoresis of RT-PCR products obtained from wild-type and mutant plasmids. The first channel is a marker, and the second is the empty vector. We observed no change in molecular weight in the mutant plasmid (channel 3) compared with the wildtype plasmid (channel 4). b Proposed model of results by the splice-site mutation. The primers SD6 and SA2 were used to amplify exons 4 and 5 of the DNAHB13 gene. c Sequence analysis of the RT-PCR product obtained from wild-type and mutant plasmid. (PNG 445 kb)
Rights and permissions
About this article
Cite this article
Liu, M., Li, J., Jiang, C. et al. A novel homozygous mutation in DNAJB13—a gene associated with the sperm axoneme—leads to teratozoospermia. J Assist Reprod Genet 39, 757–764 (2022). https://doi.org/10.1007/s10815-022-02431-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-022-02431-1