Abstract
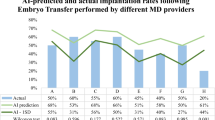
Staff competency is a crucial component of the in vitro fertilization (IVF) laboratory quality management system because it impacts clinical outcomes and informs the key performance indicators (KPIs) used to continuously monitor and assess culture conditions. Contemporary quality control and assurance in the IVF lab can be automated (collect, store, retrieve, and analyze), to elevate quality control and assurance beyond the cursory monthly review. Here we demonstrate that statistical KPI monitoring systems for individual embryologist performance and culture conditions can be detected by artificial intelligence systems to provide systemic, early detection of adverse outcomes, and identify clinically relevant shifts in pregnancy rates, providing critical validation for two statistical process controls proposed in the Vienna Consensus Document; intracytoplasmic sperm injection (ICSI) fertilization rate and day 3 embryo quality.



Similar content being viewed by others
Change history
14 May 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10815-021-02225-x
References
Olofsson JI, Banker MR, Sjoblom LP. Quality management systems for your in vitro fertilization clinic’s laboratory: why bother? J Hum Reprod Sci. 2013;6(1):3–8.
Cirillo F, Patrizio P, Baccini M, Morenghi E, Ronchetti C, Cafaro L, et al. The human factor: does the operator performing the embryo transfer significantly impact the cycle outcome? Hum Reprod. 2020;35(2):275–82.
Franco JG Jr, et al. Key performance indicators score (KPIs-score) based on clinical and laboratorial parameters can establish benchmarks for internal quality control in an ART program. JBRA Assist Reprod. 2017;21(2):61–6.
Castillo CM, et al. The impact of selected embryo culture conditions on ART treatment cycle outcomes: a UK national study. Hum Reprod Open. 2020;2020(1):hoz031.
Matson PL. Internal quality control and external quality assurance in the IVF laboratory. Hum Reprod. 1998;13(Suppl 4):156–65.
Strandell A, Bergh C, Lundin K. Selection of patients suitable for one-embryo transfer may reduce the rate of multiple births by half without impairment of overall birth rates. Hum Reprod. 2000;15(12):2520–5.
Curchoe CL, Bormann CL. Artificial intelligence and machine learning for human reproduction and embryology presented at ASRM and ESHRE 2018. J Assist Reprod Genet. 2019;36(4):591–600.
Kanakasabapathy MK, Thirumalaraju P, Bormann CL, Kandula H, Dimitriadis I, Souter I, et al. Development and evaluation of inexpensive automated deep learning-based imaging systems for embryology. Lab Chip. 2019;19(24):4139–45.
Khosravi P, Kazemi E, Zhan Q, Malmsten JE, Toschi M, Zisimopoulos P, et al. Deep learning enables robust assessment and selection of human blastocysts after in vitro fertilization. NPJ Digit Med. 2019;2:21.
Vogiatzi P, Pouliakis A, Siristatidis C. An artificial neural network for the prediction of assisted reproduction outcome. J Assist Reprod Genet. 2019;36(7):1441–8.
Tran D, Cooke S, Illingworth PJ, Gardner DK. Deep learning as a predictive tool for fetal heart pregnancy following time-lapse incubation and blastocyst transfer. Hum Reprod. 2019;34(6):1011–8.
Thirumalaraju P, Kanakasabapathy MK, Bormann CL, Gupta R, Pooniwala R Kandula H, Souter I, Dimitriadis I, Shafiee H. Evaluation of deep convolutional neural networks in classifying orphological quality. https://www.arxiv.org/abs/2005.10912
Behr B, Wang H. Effects of culture conditions on IVF outcome. Eur J Obstet Gynecol Reprod Biol. 2004;115(Suppl 1):S72–6. https://doi.org/10.1016/j.ejogrb.2004.01.016.
Kanakasabapathy, M.K., et al. Deep learning mediated single time-point image-based prediction of embryo developmental outcome at the cleavage stage. arXiv:2006.08346v1 [q-bio.TO]
Smith GD, Takayama S, Swain JE. Rethinking in vitro embryo culture: new developments in culture platforms and potential to improve assisted reproductive technologies. Biol Reprod. 2012;86(3):62.
Go KJ. ‘By the work, one knows the workman’: the practice and profession of the embryologist and its translation to quality in the embryology laboratory. Reprod BioMed Online. 2015;31(4):449–58.
Wikland M, Sjoblom C. The application of quality systems in ART programs. Mol Cell Endocrinol. 2000;166(1):3–7.
Embryology, E.S.I.G.o. and c.b.g.i. Alpha Scientists in Reproductive Medicine. Electronic address, The Vienna consensus: report of an expert meeting on the development of ART laboratory performance indicators. Reprod BioMed Online. 2017;35(5):494–510.
Hammond ER, Morbeck DE. Tracking quality: can embryology key performance indicators be used to identify clinically relevant shifts in pregnancy rate? Hum Reprod. 2019;34(1):37–43.
Alper MM, Brinsden PR, Fischer R, Wikland M. Is your IVF programme good? Hum Reprod. 2002;17(1):8–10.
Curchoe CL. Smartphone applications for reproduction: from rigorously validated and clinically useful to potentially harmful. EMJ Repro Health. 2020;6(1):85–91.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Charles L. Bormann should be listed as the first author for the article.
Rights and permissions
About this article
Cite this article
Bormann, C.L., Curchoe, C.L., Thirumalaraju, P. et al. Deep learning early warning system for embryo culture conditions and embryologist performance in the ART laboratory. J Assist Reprod Genet 38, 1641–1646 (2021). https://doi.org/10.1007/s10815-021-02198-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-021-02198-x