Abstract
Purpose
To evaluate the tumor-inducing ability of a few leukemic cells xenotransplanted inside an artificial ovary.
Methods
Ten and 100 BV-173 leukemic cells were embedded in a fibrin matrix along with 50,000 human ovarian stromal cells, and grafted to the peritoneal bursa of 5 and 5 SCID mice respectively. Four mice grafted with 3x106 leukemic cells in fibrin served as positive controls. At 20 weeks post-transplantation, the grafts, liver, spleen, blood and bone marrow were analyzed for the presence of leukemia by anti-CD79α IHC, flow cytometry (FC) and PCR.
Results
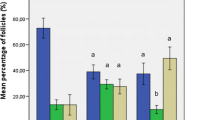
All mice grafted with 3x106 cells developed peritoneal masses 4 weeks after xenotransplantation, and systemic disease was confirmed by IHC, PCR and FC. Among mice grafted with 10 or 100 leukemic cells, none showed any sign of leukemia after 20 weeks, and IHC, FC and PCR on the different recovered tissues all proved negative.
Conclusion
This study investigates the tumor-inducing potential of a few leukemic cells grafted inside an artificial ovary. Transplantation of 100 leukemic cells appears to be insufficient to induce leukemia after 20 weeks. These results in an immunodeficient xenografting model are quite reassuring. However, for clinical application, follicle suspensions must be purged of leukemic cells before grafting, as even the slightest risk should be avoided.



Similar content being viewed by others
References
Edwards BK, Noone A-M, Mariotto AB, Simard EP, Boscoe FP, Henley SJ, et al. Annual Report to the Nation on the status of cancer, 1975–2010, featuring prevalence of comorbidity and impact on survival among persons with lung, colorectal, breast, or prostate cancer. Cancer. 2013:n/a-n/a.
Ward E, Desantis C, Robbins A, Kohler B, Jemal A. Childhood and adolescent cancer statistics, 2014. CA Cancer J Clin. 2014;64(2):83–103.
Meirow D, Nugent D. The effects of radiotherapy and chemotherapy on female reproduction. Hum Reprod Update. 2001;7(6):535–43.
Wallace WH, Anderson RA, Irvine DS. Fertility preservation for young patients with cancer: who is at risk and what can be offered? Lancet Oncol. 2005;6(4):209–18.
Donnez J, Martinez-Madrid B, Jadoul P, Van Langendonckt A, Demylle D, Dolmans MM. Ovarian tissue cryopreservation and transplantation: a review. Hum Reprod Update. 2006;12(5):519–35.
Byrne J, Fears TR, Gail MH, Pee D, Connelly RR, Austin DF, et al. Early menopause in long-term survivors of cancer during adolescence. Am J Obstet Gynecol. 1992;166(3):788–93.
Larsen EC, Muller J, Schmiegelow K, Rechnitzer C, Andersen AN. Reduced ovarian function in long-term survivors of radiation- and chemotherapy-treated childhood cancer. J Clin Endocrinol Metab. 2003;88(11):5307–14.
Green DM, Kawashima T, Stovall M, Leisenring W, Sklar CA, Mertens AC, et al. Fertility of female survivors of childhood cancer: a report from the childhood cancer survivor study. J Clin Oncol. 2009;27(16):2677–85.
Anderson RA, Wallace WH. Antimullerian hormone, the assessment of the ovarian reserve, and the reproductive outcome of the young patient with cancer. Fertil Steril. 2013;99(6):1469–75.
Jadoul P, Dolmans MM, Donnez J. Fertility preservation in girls during childhood: is it feasible, efficient and safe and to whom should it be proposed? Hum Reprod Update. 2010;16(6):617–30.
Cancer in Children and Adolescents. Belgian Cancer Registry; 2013.
Dolmans MM, Jadoul P, Gilliaux S, Amorim CA, Luyckx V, Squifflet J, et al. A review of 15 years of ovarian tissue bank activities. J Assist Reprod Genet. 2013;30(3):305–14.
Dolmans MM, Luyckx V, Donnez J, Andersen CY, Greve T. Risk of transferring malignant cells with transplanted frozen-thawed ovarian tissue. Fertil Steril. 2013;99(6):1514–22.
Donnez J, Dolmans MM. Fertility preservation in women. Nat Rev Endocrinol. 2013;9(12):735–49.
Cobo A, Garcia-Velasco JA, Domingo J, Remohi J, Pellicer A. Is vitrification of oocytes useful for fertility preservation for age-related fertility decline and in cancer patients? Fertil Steril. 2013;99(6):1485–95.
Cakmak H, Rosen MP. Ovarian stimulation in cancer patients. Fertil Steril. 2013;99(6):1476–84.
Meirow D, Hardan I, Dor J, Fridman E, Elizur S, Ra’anani H, et al. Searching for evidence of disease and malignant cell contamination in ovarian tissue stored from hematologic cancer patients. Hum Reprod. 2008;23(5):1007–13.
Dolmans MM, Marinescu C, Saussoy P, Van Langendonckt A, Amorim C, Donnez J. Reimplantation of cryopreserved ovarian tissue from patients with acute lymphoblastic leukemia is potentially unsafe. Blood. 2010;116(16):2908–14.
Rosendahl M, Andersen MT, Ralfkiaer E, Kjeldsen L, Andersen MK, Andersen CY. Evidence of residual disease in cryopreserved ovarian cortex from female patients with leukemia. Fertil Steril. 2010;94(6):2186–90.
Amiot C, Angelot-Delettre F, Zver T, Alvergnas-Vieille M, Saas P, Garnache-Ottou F, et al. Minimal residual disease detection of leukemic cells in ovarian cortex by eight-color flow cytometry. Hum Reprod. 2013;28(8):2157–67.
Greve T, Clasen-Linde E, Andersen MT, Andersen MK, Sorensen SD, Rosendahl M, et al. Cryopreserved ovarian cortex from patients with leukemia in complete remission contains no apparent viable malignant cells. Blood. 2012;120(22):4311–6.
Bastings L, Beerendonk CC, Westphal JR, Massuger LF, Kaal SE, van Leeuwen FE, et al. Autotransplantation of cryopreserved ovarian tissue in cancer survivors and the risk of reintroducing malignancy: a systematic review. Hum Reprod Update. 2013;19(5):483–506.
Rosendahl M, Greve T, Andersen CY. The safety of transplanting cryopreserved ovarian tissue in cancer patients: a review of the literature. J Assist Reprod Genet. 2013;30(1):11–24.
Dolmans MM, Martinez-Madrid B, Gadisseux E, Guiot Y, Yuan WY, Torre A, et al. Short-term transplantation of isolated human ovarian follicles and cortical tissue into nude mice. Reproduction. 2007;134(2):253–62.
Dolmans MM, Yuan WY, Camboni A, Torre A, Van Langendonckt A, Martinez-Madrid B, et al. Development of antral follicles after xenografting of isolated small human preantral follicles. Reprod BioMed Online. 2008;16(5):705–11.
Vanacker J, Luyckx V, Dolmans MM, Des Rieux A, Jaeger J, Van Langendonckt A, et al. Transplantation of an alginate-matrigel matrix containing isolated ovarian cells: first step in developing a biodegradable scaffold to transplant isolated preantral follicles and ovarian cells. Biomaterials. 2012;33(26):6079–85.
Luyckx V, Dolmans MM, Vanacker J, Scalercio SR, Donnez J, Amorim CA. First step in developing a 3D biodegradable fibrin scaffold for an artificial ovary. J Ovarian Res. 2013;6(1):83.
Luyckx V, Dolmans MM, Vanacker J, Legat C, Fortuno Moya C, Donnez J, et al. A new step toward the artificial ovary: survival and proliferation of isolated murine follicles after autologous transplantation in a fibrin scaffold. Fertil Steril. 2014;101(4):1149–56.
Dath C, Dethy A, Van Langendonckt A, Van Eyck AS, Amorim CA, Luyckx V, et al. Endothelial cells are essential for ovarian stromal tissue restructuring after xenotransplantation of isolated ovarian stromal cells. Hum Reprod. 2011;26(6):1431–9.
Vanacker J, Camboni A, Dath C, Van Langendonckt A, Dolmans MM, Donnez J, et al. Enzymatic isolation of human primordial and primary ovarian follicles with Liberase DH: protocol for application in a clinical setting. Fertil Steril. 2011;96(2):379–83.e3.
Dolmans MM, Michaux N, Camboni A, Martinez-Madrid B, Van Langendonckt A, Nottola SA, et al. Evaluation of Liberase, a purified enzyme blend, for the isolation of human primordial and primary ovarian follicles. Hum Reprod. 2006;21(2):413–20.
Eppig JJ, O’Brien MJ. Development in vitro of mouse oocytes from primordial follicles. Biol Reprod. 1996;54(1):197–207.
O’Brien MJ, Pendola JK, Eppig JJ. A revised protocol for in vitro development of mouse oocytes from primordial follicles dramatically improves their developmental competence. Biol Reprod. 2003;68(5):1682–6.
Spears N, Boland NI, Murray AA, Gosden RG. Mouse oocytes derived from in vitro grown primary ovarian follicles are fertile. Hum Reprod. 1994;9(3):527–32.
Telfer EE, Zelinski MB. Ovarian follicle culture: advances and challenges for human and nonhuman primates. Fertil Steril. 2013;99(6):1523–33.
Hermann BP, Sukhwani M, Salati J, Sheng Y, Chu T, Orwig KE. Separating spermatogonia from cancer cells in contaminated prepubertal primate testis cell suspensions. Hum Reprod. 2011;26(12):3222–31.
Fujita K, Ohta H, Tsujimura A, Takao T, Miyagawa Y, Takada S, et al. Transplantation of spermatogonial stem cells isolated from leukemic mice restores fertility without inducing leukemia. J Clin Invest. 2005;115(7):1855–61.
Jahnukainen K, Morris I, Roe S, Salmi TT, Makipernaa A, Pollanen P. A rodent model for testicular involvement in acute lymphoblastic leukaemia. Br J Cancer. 1993;67(5):885–92.
Hou M, Andersson M, Zheng C, Sundblad A, Soder O, Jahnukainen K. Decontamination of leukemic cells and enrichment of germ cells from testicular samples from rats with Roser’s T-cell leukemia by flow cytometric sorting. Reproduction. 2007;134(6):767–79.
Fujita K, Tsujimura A, Hirai T, Ohta H, Matsuoka Y, Miyagawa Y, et al. Effect of human leukemia cells in testicular tissues grafted into immunodeficient mice. Int J Urol. 2008;15(8):733–8.
Donnez J, Jadoul P, Pirard C, Hutchings G, Demylle D, Squifflet J, et al. Live birth after transplantation of frozen-thawed ovarian tissue after bilateral oophorectomy for benign disease. Fertil Steril. 2012;98(3):720–5.
Donnez J, Dolmans MM, Pellicer A, Diaz-Garcia C, Sanchez Serrano M, Schmidt KT, et al. Restoration of ovarian activity and pregnancy after transplantation of cryopreserved ovarian tissue: a review of 60 cases of reimplantation. Fertil Steril. 2013;99(6):1503–13.
Fingert HJ, Chen Z, Mizrahi N, Gajewski WH, Bamberg MP, Kradin RL. Rapid growth of human cancer cells in a mouse model with fibrin clot subrenal capsule assay. Cancer Res. 1987;47(14):3824–9.
Jahnukainen K, Hou M, Petersen C, Setchell B, Soder O. Intratesticular transplantation of testicular cells from leukemic rats causes transmission of leukemia. Cancer Res. 2001;61(2):706–10.
Shankaran V, Ikeda H, Bruce AT, White JM, Swanson PE, Old LJ, et al. IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature. 2001;410(6832):1107–11.
Acknowledgments
The authors thank Mira Hryniuk for reviewing the English language of the manuscript and Olivier Van Kerk and Dolores Gonzalez for their technical assistance. Help from specialized technicians, namely Patricia Leveugle and Anne-Marie Mazzon for flow cytometry experiments and Béatrice Delepaut for PCR, was also greatly appreciated.
This work was supported by grants from the Fonds National de la Recherche Scientifique de Belgique (5/4/150/5 and 7.4518.12F), Fonds Spéciaux de Recherche, Fondation Saint Luc, Foundation Against Cancer, and donations from Mr. Pietro Ferrero, Baron Frère, and Viscount Philippe de Spoelberch.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule The tumor-inducing ability of a few BV-173 leukemic cells was evaluated in an artificial ovary by xenotransplantation. One hundred BV-173 cells proved insufficient to induce disease in mice after 20 weeks.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig 1
(JPEG 86 kb)
Supplementary Table 1
Flow cytometry results for leukemic cell detection expressed as the percentage of total events showing the BV-173 lineage phenotype. This table presents results obtained using panel 2 (CD44-FITC/CD10-PE/CD45-ECD/CD38-PC5/CD19-PC7), but percentages were similar with panel 1 (CD44-FITC/CD10-PE/CD45-ECD/CD38-PC5/CD19-PC7). Mice grafted with 10 or 100 leukemic cells showed no sign of leukemia in any of the different tissue samples by FC. Conversely, in all mice grafted with 3 million leukemic cells, the BV-173 cell phenotype was identified in peritoneal grafts and at least one other sample, showing the presence of systemic disease after 4 weeks of grafting. Neg = negative, Pos = positive, NP = analysis not possible due to small sample volume. (DOC 43 kb)
Rights and permissions
About this article
Cite this article
Soares, M., Saussoy, P., Sahrari, K. et al. Is transplantation of a few leukemic cells inside an artificial ovary able to induce leukemia in an experimental model?. J Assist Reprod Genet 32, 597–606 (2015). https://doi.org/10.1007/s10815-015-0438-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-015-0438-x