Abstract
Purpose
The aim of this study was to investigate whether treatment of sperm from infertile patients would gluthatione could reduce sperm premature chromosome condensation (PCC). To reach the goals of this study, the frequency of sperm PCC formation in sperm of normal and sub-fertile men with/without glutathione treatment were compared and analyzed.
Methods
Hamster oocytes were retrieved after super ovulation by PMSG and HCG injection. Following treatment with hyaluronidase, zonae was removed by trypsin digestion. Sperm were classified into 3 groups according to morphology, movement and counts, treated with glutathione(10 μg/ml) and then processed by swim up method. After capacitation, zona-free oocytes were incubated with sperm then transferred to fresh media containing colcemid. Cells were fixed and slides prepared using Tarkowskie’s standard air drying technique and after staining in 5% Giemsa, oocytes were analyzed at high magnification.
Results
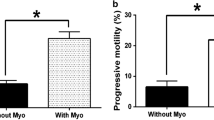
Sperm penetration rate was higher and the rate of intact sperm head and PCC was lower in GSH treated samples compared to non treated groups. Sperm penetration rate was significantly higher in treated astheno sperm samples compared to non-treated ones (66.4% and 50. 97% respectively) (P < 0.001). We observed a significantly higher PCC frequency in infertile patients (P < 0.001). In addition, there was a significantly lower rate of intact sperm head in treated astheno sperm samples (17.49%) compared to non treated ones (26.79%) (P < 0.001). Finally, a significantly lower rate of PCC in treated astheno sperm samples comparing to non treated ones was seen (51.06% and 72.96% respectively) (P < 0. 001).
Conclusions
Our results show that sperm PCC formation could be one of the major causes of failed fertilization in individuals with sperm abnormalities. Also sperm PCC formation may be involved in the etiology of some cases of idiopathic infertility. Given that the susceptibility of sperm to oxidative stress is significantly greater in idiopathic infertile men, our results show that treatment with glutathione could significantly reduce these stress factors and increase ART outcome.
Similar content being viewed by others
References
Nasr-Esfahani MH, Naghshizadian N, Imani H, Razavi S, Mardani M, Kazemi S, Shahvardi H. Can sperm protamine deficiency induce sperm premature chromosomal condensation? Andrologia. 2006;38:92–8.
Perreault SD, Barbee RR, Slott VL. Importance of glutathione in the acquisition and maintenance of sperm nuclear decondensing activity in maturing hamster oocytes’. Dev Biol. 1988;125:181–6.
Schmiady H, Sperling K, Kentenich H, Stauber M. Prematurely condensed human sperm chromosomes after in vitro fertilization (IVF). Hum Genet. 1986;74:441–3.
Ma S, Kalousek DK, Yuen BH, Gomel V, Katagiri S, Moon YS. Chromosome investigation in invitro fertilization failure. J Assist Reprod Genet. 1994;1:445–51.
Ma S, Yuen BH. Intra cytoplasmic sperm injection could minimize the incidence of prematurely condensed human sperm chromosomes. Fertil Steril. 2001;75:1095–101.
Luberda Z. The role of glutathione in mammalian gametes. Reprod Biol. 2005;5(1):5–17.
Finotti ACF, Silva RCP Costa e, Bordin BM, Silva CTX, Moura KKVO. Glutathione S-transferase M1 and T1 polymorphism in men with idiopathic infertility. Genet Mol Res. 2009;8(3):1093–8.
Huynh T, Mollard R, Trounson A. Selected genetic factors associated with male infertility. Hum Reprod Update. 2002;8:183–98.
Rosenbuch BE. Frequency and patterns of premature sperm chromosome condensation in oocytes failing to fertilize after intra cytoplasmic sperm injection. J Assist Reprod Genet. 2000;17:253–9.
Zuelke KA, Jeffay SC, Zucker RM, Perreault SD. Glutathione (GSH) concentrations vary with the Cell cycle in maturing hamster oocytes, zygotes, and pre-implantation stage embryos. Mol Rep Dev. 2003;64:106–12.
Mozdarani H, Mohseni, Meybodi A. Cytogenetic analysis of sperm nucleous components of Iranian normal and sub-fertile individuals using zona free hamster oocytes. J Assist Reprod Genet. 2004;21(11):409–14.
Tarkowski AK. An Air-drying method for chromosome preparation from mouse eggs. Cytogenetics. 1986;5:394–400.
Nasr-Esfahani MH, Razavi Sh, Mardani M, Shirazi R, Javanmardi S. Effects of failed oocyte activation and sperm protamine deficiency on fertilization post-ICSI. Reprod Biomed Online. 2007;14(4):422–9.
Perreault SD, Naish SJ. The timing of Hamster sperm nuclear decondensation and male pronucleus formation is related to sperm nuclear disulfide bond content. Biol Reprod. 1987;36:239–44.
Takeo T, Nakagata N. Reduced glutathione enhances fertility of frozen/thawed C57BL/6 mouse sperm after exposure to Methyl-Beta-Cyclodextrin. Biol Reprod. 2011;85(5):1066–72.
Bath ML. Inhibition of in vitro fertilizing capacity of cryopreserved mouse sperm by factors released by damaged sperm, and stimulation by glutathione. PLoS One. 2010;5(2):e9387.
Donnelly ET, McClure N, Lewis SE. Glutathione and hypotaurine in vitro: effects on human sperm motility, DNA integrity and production of reactive oxygen species. Mutagenesis. 2000;15:61–8.
Gadea J, Molla M, Selles E, Marco MA, Garcia-Vazquez FA, Gardon JC. Reduced glutathione content in human sperm is decreased after cryopreservation: effect of the addition of reduced glutathione to the freezing and thawing extenders. Cryobiology. 2011;62:40–6.
Aydemir B, Onaran I, Kiziler AR, Alici B, Akyolcu MC. Increased oxidative damage of sperm and seminal plasma in men with idiopathic infertility is higher in patients with gluthatione S-transferase Mu-1 null genotype. Asian J Androl. 2007;9(1):108–15.
Aitken RJ. The amoroso lecture: The human spermatozoon – a cell in crisis? J Reprod Fertil. 1999;115:1–7.
Sies H. Glutathione and its role in cellular functions. Free Radic Biol Med. 1999;27:916–21.
Fassio A, Sitia R. Formation, isomerisation and reduction of disulphide bonds during protein quality control in the endoplasmic reticulum. Histochem Cell Biol. 2002;117:151–7.
Boquest AC, Abeydeera LR, Wang WH, Day BN. Effect of adding reduced glutathione during insemination on the development of porcine embryos in vitro. Theriogenology. 1999;51:1311–9.
Acknowledgment
We are grateful to Dr. Mehrdad Mohseni for the help during the submission of this article. This study was funded by grants provided from Royan Institute.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule A clinical need exists for establishment of approaches which can be utilized to increase the ART success rate by treatment of the infertile patient’s sperms with gluthatione.
Rights and permissions
About this article
Cite this article
Meybodi, A.M., Mozdarani, H., Moradi, S.Z. et al. Importance of sperm gluthatione treatment in ART. J Assist Reprod Genet 29, 625–630 (2012). https://doi.org/10.1007/s10815-012-9755-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-012-9755-5